Abstract
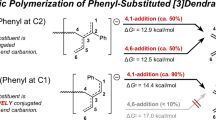
The reactivity of model anions of active species in the anionic polymerization of phenylbutadienes was investigated by reactions with several electrophiles. The anions employed are generated by one-to-one addition of 1-phenyl-1,3-butadiene with t-BuLi, lithiation of 1-phenyl-2-butene, one-to-one addition of 2-phenyl-1,3-butadiene with t-BuLi, and lithiation of 3-phenyl-1-butene. The former two anions are classified as π-benzyl type anions and the latter two as π-allylphenyl type anions according to negative charge distributions. In the reactions of these anions with methanol (protonation), difference in negative charge distribution does not affect the product distribution, and the reactivity of the α-position (terminal of the anion) is high. When methyl iodide is reacted, α-products (products reacted at the α-position) are selectively obtained (96—98%) in the case of π-benzyl type anions. On the other hand, the amount of α-products decreased (45—85%) in the case of π-allylphenyl type anions. These findings can be attributed to the differences in negative charge distribution. However, in reactions with trimethylchlorosilane or trimethylchlorotin, steric effect at the α- or γ-carbon controls the regioselectivity of the products. In reactions with acetone or ethylene oxide, α-products are obtained predominantly in the case of π-benzyl type anions, while γ-products are obtained mainly in the case of π-allylphenyl type anions.
Similar content being viewed by others
Article PDF
References
W. H. Glaze, J. E. Hanicak, M. L. Moore, and J. Chaudhuri, J. Organometal. Chem., 44, 39 (1972).
W. H. Glaze, J. E. Hanicak, D. J. Berry, and D. P. Duncan, J. Organometal. Chem., 44, 49 (1972).
W. H. Glaze, J. E. Hanicak, J. Chandhuri, M. L. Moore, and D. P. Duncan, J. Organometal. Chem., 51, 13 (1973).
W. H. Glaze, D. J. Berry, and D. P. Duncan, J. Organometal. Chem., 52, 233 (1973).
J. Sledz, F. Schué, B. K. Kaempf, and S. Libs, Eur. Polym. J., 10, 1207 (1974).
S. Brownstein, S. Bywater, and D. J. Worsfold, Macromolecules, 6, 715 (1973).
S. Bywater, D. J. Worsfold, and G. Hollingsworth, Macromolecules, 5, 389 (1972).
F. Schué, D. J. Worthfold, and S. Bywater, Macromolecules, 3, 509 (1970).
M. Morton, R. D. Sanderson, R. Sakata, and L. A. Falvo, Macromolecules, 6, 186 (1973).
M. Morton, R. D. Sanderson, and R. Sakata, J. Polym. Sci., Polym. Lett. Ed., 9, 61 (1971).
M. Morton, R. D. Sanderson, and R. Sakata, Macromolecules, 6, 181 (1973).
R. Salle and J. Gole, J. Polym. Sci., Polym. Chem. Ed., 15, 1199 (1977).
S. Bywater and D. J. Worsfold, J. Organometal. Chem., 159, 229 (1978).
S. Bywater, P. Lachance, and D. J. Worsfold, J. Phys. Chem., 79, 2148 (1978).
P. Miginiac, Bull. Soc. Chim. Fr., 1077 (1970).
T. Suzuki, Y. Tsuji, and Y. Takegami, Macromolecules, 11, 639 (1978).
T. Suzuki, Y. Tsuji, Y. Takegami, and H. J. Harwood, Macromolecules, 12, 234 (1979).
T. Suzuki, Y. Tsuji, Y. Watanabe, and Y. Takegami, Polym J., 11, 651 (1979).
T. Suzuki, Y. Tsuji, Y. Watanabe, and Y. Takegami, Polym. J., 11, 937 (1979).
G. Courtois and L. Miginiac, J. Organometal. Chem., 69, 1 (1974).
M. Gaudemar, Bull. Soc. Chim. Fr., 974 (1962).
L. Miginiac, Ph. Miginiac, and Ch. Prevost, Bull. Soc. Chim. Fr., 3560 (1965).
H. Felkin and C. Frajerman, Tetrahedron Lett., 1045 (1970).
M. Cherest, H. Felkin, and C. Frajerman, Tetrahedron Lett., 379 (1971).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsuji, Y., Suzuki, T., Watanabe, Y. et al. Active Species in Anionic Polymerization of Phenylbutadienes —Reactivity of Model Anions—. Polym J 13, 1099–1110 (1981). https://doi.org/10.1295/polymj.13.1099
Issue Date:
DOI: https://doi.org/10.1295/polymj.13.1099