Abstract
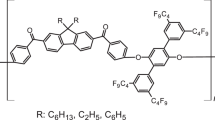
A series of amorphous and semicrystalline poly(aryl ether-bisketone)s have been synthesized from bisphenols and 3,3′-(4-fluorobenzoyl)biphenyl via nucleophilic aromatic substitution reactions. Model compound studies were carried out with a variety of substituted phenols, 3,3′-(4-fluorobenzoyl)biphenyl and 3,3′-(4-chlorobenzoyl)biphenyl. The bishalide monomers were prepared by the reaction of 3,3′-biphenyl dicarboxylic acid with thionyl chloride followed by Friedel–Crafts reaction with the appropriate aryl halide. Potassium carbonate mediated reaction of these monomers with a variety of bisphenols in dimethylacetamide or diphenylsulfone gave moderate to high molecular weight polymers in excellent yield. The high molecular weight polymers afforded tough films by compression molding. The glass transition temperatures, melting points and the thermal behaviors of the polymers were compared with the corresponding polymers derived from 4,4′-(4-fluorobenzoyl)biphenyl.
Similar content being viewed by others
Article PDF
References
C. P. Smith, Chemtech, 290 (1988).
S. Maiti and B. K. Mandal, Progr. Polym. Sci., 12, 111 (1986).
J. E. Harris and R. N. Johnson, “Encyclopedia of Polymer Science and Engineering,” Vol. 13, 2nd ed, H. F. Mark, N. M. Bikales, C. G. Overberger, and G. Manges, Ed, New York, N.Y., 1988, p 196.
F. Parodi, In “Comprehensive Polymer Science,” Vol. 5, G. Allen and J. C. Bevington, Ed., Pergamon, Oxford, 1989, p 561.
R. N. Johnson, A. G. Farnham, R. A. Callendinning, W. F. Hale, and C. N. J. Merian, J. Polym. Sci., Polym. Chem. Ed., 5, 2375 (1967).
R. Viswanathan, B. C. Johnson, and J. E. McGrath, Polymer, 25, 1827 (1984).
B. C. Auman, V. Percec, H. A. Scneider, W. Jishan, and H. J. Cantow, Polymer, 28, 119 (1987).
J. L. Hedrick and J. W. Labadie, Macromolecules, 23, 1561 (1990).
W. Risse and D. Y. Sogah, Macromolecules, 23, 4029 (1990).
T. E. Atwood, D. A. Barr, T. A. King, B. Newton, and J. B. Rose, Polymer, 18, 359 (1977).
J. Miller, “Aromatic Nucleophilic Substitution,” Elsevier, Amsterdam, The Netherlands, 1968, pp 139—148.
T. E. Atwood, P. C. Dawson, J. L. Freeman, L. R. J. Hoy, J. B. Rose, and P. A. Stanliland, Polymer, 22, 1096 (1981).
P. C. Dawson and D. Bundell, Polymer, 21, 577 (1980).
R. Rigby, Adv. Polym. Technol., 2, 163 (1982).
C. M. Chan and S. Venkatraman, Polym. Mater. Sci. Eng., 54, 37 (1986).
D. K. Mohanty, S. D. Wu, and J. E. McGrath, Polym. Prepr., Am. Chem. Soc., Div. Poly. Chem., 29, 352 (1988).
S. D. Wu, J. L. Hedrick, D. K. Mohanty, B. Carter, G. L. Wilkes, and J. E. McGrath, Int. SAMPE Symp., 31, 933 (1986).
D. K. Mohanty and J. E. McGrath, in “Advances in Polymer Synthesis,” 31, B. M. Cullbertson and J. E. McGrath, Ed., Plenum Press, New York and London, 1985, p 113.
J. L. Hedrick, D. K. Mohanty, B. C. Johnson, R. Viswanathan, J. A. Hinkley, and J. E. McGrath, J. Polym. Sci., Polym. Chem. Ed., 24, 287 (1986).
P. M. Hergenrother and B. J. Jensen, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 26, 174 (1985).
P. M. Hergenrother, N. T. Wakelyn, and S. J. Havens, J. Polym. Sci., Polym. Chem. Ed., 25, 1093 (1987).
H. R. Kricheldorf, U. Delius, and K. U. Tonnes, New Polymeric Materials, 1, 127 (1988).
J. M. DeSimone, S. Stompel, and E. T. Samulski, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 32, 172 (1991).
M. Winkler, P. Itteman, and G. Heinz, Ger. Offen, DE 3738749, 24 May 1989, BASF-AG.
V. Janson, S. Moore, and J. B. Mazzanti, Eur. Patent Appl. Ep 298771, Jan. 1989, Raychem Corp.
I. Colon and G. T. Kwiatkowski, J. Polym. Sci., Polym. Chem. Ed., 28, 367 (1990).
D. C. Cummings, R. S. Mani, P. B. Balanda, B. A. Howell, and D. K. Mohanty, J. Macromol. Sci., Polym. Chem. Ed., 28, 793 (1991).
A. Bhatnagar, R. S. Mani, B. R. Weeks, and D. K. Mohanty, J. Mater. Chem., 6, 977 (1991).
H. R. Snyder, C. Weaver, and T. E. Marshall, J. Am. Chem. Soc., 71, 289 (1949).
P. E. Resevear and W. H. F. Sasse, J. Hetero. Chem., 483 (1971).
J. W. Connel and P. M. Hergenrother, High Perform. Polym., 2, 211 (1990).
R. S. Mani and D. K. Mohanty, Polym. Mater. Sci. Eng., 65, 247 (1991).
J. F. Bunnet, Ann. Rev. Phys. Chem., 14, 271 (1963).
R. G. Pearson and J. Songstad, J. Am. Chem. Soc., 89, 1827 (1967).
R. G. Pearson, H. Sobel, and J. Songstad, J. Am. Chem. Soc., 90, 319 (1968).
P. L. Block and G. M. Whiteside, J. Am. Chem. Soc., 96, 2826 (1974).
M. Fujio, R. T. Mclver, Jr., and R. W. Taft, J. Am. Chem., 103, 4017 (1981).
R. S. Mani, B. Zimmernran, and D. K. Mohanty, Polymer, 34, 171 (1993).
V. Percec, R. S. Clough, P. L. Rinaldi, and V. E. Litman, Macromolecules, 24, 5889 (1991).
A. Bhatnagar, B. King, R. S. Mani, and D. K. Mohanty, Polymer (in press).
G. C. Levy, R. L. Lichter, and G. L. Nelson, in “Carbon-13 Nuclear Magnetic Spectroscopy,” 2nd Ed., John Wiley and Sons, New York, N.Y., 1980, p 111.
K. Tanaka, H. Kita, K. Okamoto, A. Nakamura, and Y. Kusuki, Polym. J., 22, 381 (1990).
L. M. Robeson, A. G. Farnham, and J. E. McGrath, in “Molecular Basis of Transitions and Relaxation,” D. J. Meier, Ed., Gordon and Breach, New York, N.Y., 1978, p 405.
G. Allen, J. McAinsh, and G. M. Jetts, Polymer, 18, 85 (1971).
J. Heigboer, Br. Polym. J., 1, 3 (1969).
J. J. Dumais, A. L. Cholli, L. W. Jelenski, J. L. Hedrick, and J. E. McGrath, Macromolecules, 19, 1884 (1986).
J. R. Fried and H. Kalkanoglu, J. Polym. Sci., Polym. Lett. Ed., 20, 381 (1982).
D. W. van Krevelen, Polymer, 16, 516 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mani, R., Weeks, B., Bhatnagar, A. et al. Synthesis and Characterization of Isomeric Biphenyl Containing Poly(aryl ether-bisketone)s II. Polymers Derived from 3,3′-(4-Fluorobenzoyl)biphenyl and Bisphenols. Polym J 26, 403–415 (1994). https://doi.org/10.1295/polymj.26.403
Issue Date:
DOI: https://doi.org/10.1295/polymj.26.403