Abstract
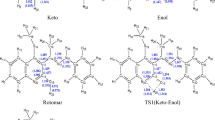
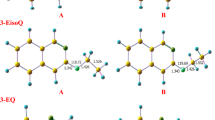
Proton and carbon-13 nuclear magnetic resonance as well as energy minimization and molecular dynamics calculations were all used to study chemical structures and single chain contributions of non-oxidized, completely oxidized and completely reduced polymerization products of p-benzoquinone. Polyquinones are shown using NMR techniques to have been transformed into polybenzoquinone upon oxidation. The negatively charged oxygen atoms of the polybenzoquinone are thus responsible for the increased torsion of the benzoquinone nuclei about connecting bonds. This should lead to lesser energetic dynamics with respect to the bending of its nuclei. Polyhydroquinone are completely stabilized by the presence of the hydrogen bonding not available to polybenzoquinone. Nuclei bending in this case is greatly influenced by the more restricted torsion behavior. Polyquinones, a mixture of both, shows inbetween behavior but more on the polyhydroquinone side which indicates the greater presence of the hydroquinone nuclei in an equimolar ratio compound.
Similar content being viewed by others
Article PDF
References
M. W. Sabaa, T. M. Madkour, and A. A. Yassin, Polym. Degrad. Stab., 22, 205 (1988).
M. W. Sabaa, T. M. Madkour, and A. A. Yassin, Polym. Degrad. Stab., 26, 347 (1989).
T. M. Madkour and M. S. Hamdi, J. Appl. Polym. Sci., 61, 1239 (1996).
M. A. Sharaf and J. E. Mark, Macromol. Rep., A28, 67 (1991).
M. W. Sabaa, T. M. Madkour, and A. A. Yassin, Polym. Degrad. Stab., 22, 195 (1988).
G. G. Odian, “Principles of Polymerization,” John Wiley & Sons, Inc., New York, N.Y., 1981.
S. H. Tersigni and W. J. Welsh, Comput. Polym. Sci., 2, 1 (1992).
W. J. Welsh, L. DeBolt, and J. E. Mark, Macromolecules, 19, 2978 (1986).
M. Depner and B. L. Schürmann, Polymer, 33, 398 (1992).
B. R. Brooks, R. E. Bruccoleri, B. D. Olafson, D. J. States, S. Swaminathan, and M. Karplus, J. Comput. Chem., 4, 187 (1983).
B. Jung and B. L. Schürmann, Macromolecules, 22, 447 (1989).
T. M. Madkour, A. Kloczkowski, J. E. Mark, and R. J. Roe, “Material Research Society Symposium Proceedings,” Vol. 278, J. E. Mark, M. E. Glicksman, and S. P. Marsh, Ed., Materials Research Society, Pittsburgh, 1992, p. 33.
L. Verlet, Phys. Rev., 159, 98 (1967).
N. L. Allinger, J. Am. Chem. Soc., 99, 8127 (1977).
F. A. Momany and R. Rone, J. Comput. Chem., 13, 888 (1992).
J.-H. Lii and N. L. Allinger, J. Am. Chem. Soc., 111, 8576 (1989).
M. P. Allen and D. J. Tildesley, “Computer Simulation of Liquids,” Clarendon Press, Oxford, 1987.
H. J. C. Berendsen, J. P. M. Postma, W. F. van Gunsteren, A. DiNola, and J. R. Haak, J. Chem. Phys., 81, 3684 (1984).
S. Kobayashi, S. Iwata, M. Abe, and S.-I. Shoda, J. Am. Chem. Soc., 117, 2187 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Madkour, T. Chemistry of Polymerization Products of p-Benzoquinone. 13C NMR and Molecular Dynamics Study. Polym J 29, 670–677 (1997). https://doi.org/10.1295/polymj.29.670
Issue Date:
DOI: https://doi.org/10.1295/polymj.29.670