Abstract
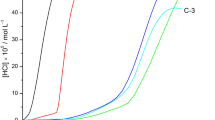
The early stages of thermal degradation below 1% dehydrochlorination poly(vinylidene chloride) (PVDC), can be divided into an induction period, period with conversion below 0.1% dehydrochlorination, and that with conversion ranging from 0.1 to 1%. The time of induction period increases with oxygen concentration due to retardation effect. The second and third stages of degradation follow the apparent zero-order reaction and rates of dehydrochlorination of both stages increase linearly with oxygen concentration. The melting points of samples with different degrees of degradation show a decreasing trend with degrading time regardless of annealing effect. By combination with our differential scanning calorimetry (DSC) thermograms and wide-angle X-ray diffraction (WAXD) patterns, in the presence of pure oxygen which first reacts with the PVDC dangling chains outside the crystalline, and cuts through the degraded crystalline along the backbones resulting in many pieces of small crystallite. A polarity change of PVDC characterized by solid-state nuclear magnetic resonance (NMR) due to degradation can be observed.
Similar content being viewed by others
Article PDF
References
C. E. Schidknecht, “Vinyl and Related Polymers,” John Wiley and Sons, New York, N.Y., 1952, Chapter 8.
R. A. Wesseling, “Encyclopedia of Polymer Science and Technology,” John Wiley and Sons, New York, N.Y., 1971, Chapter 14, p 540.
G. M. Burnett and R. A. Haldon, Eur. Polym. J., 4, 83 (1968).
B. Dolezel, M. Pegoraro, and E. Beati, Eur. Polym. J., 1, 1411 (1970).
R. A. Wesseling, “Polyvinylidene Chloride,” John Wiley and Sons, New York, N.Y., 1977, Chapter 9, p 133 and 140.
J. N. Hay, J. Polym. Sci., A-1, 8, 1201 (1970).
R. D. Bohme and R. A. Wessling, J. Appl. Polym. Sci., 16, 1761 (1972).
G. Montaudo, J. Polym. Sci., 24, 301 (1986).
K. B. Abbas, J. Appl. Polym. Sci., 17, 3567 (1973).
S. E Evsyukov, S. Paasch, and B. Thomas, Ber. Bunsen-Ges, 101, 837 (1997).
S. Biniak and J. Siedlewski, Polym. J. Appl. Chem., 36, 353 (1993).
B. A. Howell, A. M. Kelly-Rowley, and P. B. Smith, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 30, 289 (1989).
A. E. Gurgiolo, M. E. Winquist, T. M Knobel, and D. C. Teeters, U.S. Patent 4643953.
M. P. Farr and I. R. Harrison, J. Polym. Sci., Part C, Polym. Lett., 24, 257 (1986).
B. A. Howell, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 27, 439 (1986).
Y. S. Chiu, J. Jagur-Grodzinski, and D. J. Vofsi, J. Polym. Sci., Polym. Chem. Ed., 23, 1193 (1985).
J. D. Danforth, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 4, 163 (1984).
J. D. Danforth, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 21, 140 (1980).
A. Ballistrert, S. Foti, G. Mantaudo, and E. Scamporrino, Polymer, 22, 131 (1981).
P. Pendleton, B. Vincent, and M. L. Hair, J. Colloid Interface Sci., 80, 512 (1981).
M. Maciejewski, Polym. J. Macromol. Sci.-Chem., 13, 1175 (1979).
D. H. Davies, J. Chem. Soc., Faraday Trans. 1, 72, 2390 (1976).
W. Ripperger, W. Oettinger, R. Kaiser, K. Pfitzner, and R. A. Palm,. U.S. Patent 3960768.
G. J. Howard, S. Szynaka, and A. L Gatzke, J. Appl. Polym. Sci., 19, 2633 (1975).
D. E. Agostini and A. L. Gatzke, J. Polym. Sci., Polym. Chem. Ed., 11, 649 (1973).
S. S.Barton, P. G. Beswick, and B. H. Harrison, J. Chem. Soc., Faraday Trans. 1, 68, 1647 (1972).
D. H. Davies, D. H. Everett, and J. Taylor, Trans. Faraday Soc., 67, 382 (1971).
D. H. Everett and D. J. Taylor, Trans. Faraday Soc., 67, 402 (1971).
G. M . Burnett, R. A. Haldon, and J. N. Hay, Eur. Polym. J., 3, 449 (1967).
Y.-L. In, J.-H. Yang, and M.-H. Wang, Polymer Testing, 15, 525 (1996).
W. C. Geddes, Rubber Chem. and Tech., 40, 177 (1967).
G. M. Burnett and R. A. Haldon, Eur. Polym. J., 3, 449 (1967).
K. Matsuo and W. H. Stockmayer, Macromolecules, 8, 660 (1975).
T.H. Hsieh and K. S. Ho, J. Polym. Sci., Part A, Polym. Chem., in press.
H. Wiener, J. Polym. Sci., 7, 1 (1951).
K. B. Abbas and E. M. Sorvik, J. Appl. Polym. Sci., 17, 3557 (1973).
T.H. Hsieh and K.S. Ho, J. Polym. Sci., Part A, Polym. Chem., in press.
B. Wunderlich, “Macromolecular Physics,” Vol. I, Academic Press, New York, N.Y., 1973, p 349.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hsieh, TH. Effects of Oxygen on Thermal Dehydrochlorination of Poly(vinylidene chloride). Polym J 31, 948–954 (1999). https://doi.org/10.1295/polymj.31.948
Issue Date:
DOI: https://doi.org/10.1295/polymj.31.948
Keywords
This article is cited by
-
Fabrication of self-cross-linking silicified polyvinylidene chloride emulsions with core–shell structure and its film properties
Polymer Bulletin (2024)
-
Transparency, moisture barrier property, and performance of the alternative solar cell encapsulants based on PU/PVDC blend reinforced with different types of cellulose nanocrystals
Materials for Renewable and Sustainable Energy (2018)