Abstract
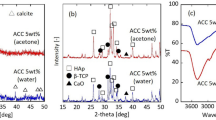
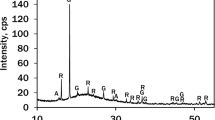
The effect of poly(acrylic acid) (PAA) on the mineralization of CaCO3 was studied by varying the molecular weights (PAA1.2k, Mw=1200; PAA25k, Mw=25000; PAA250k, Mw=250000) as well as by changing the addition time of the sodium salts of PAA (PAA-Na) to an aqueous solution of calcium carbonate. The precipitation of CaCO3 was carried out by a double jet method. Stable vaterite crystals were successfully obtained by delaying the addition of all the sodium salts of PAA from 1 to 60 min, and the resulting particles were formed by a spherulitic growth mechanism. The vaterite particles modified with PAA25k-Na and PAA250k-Na showed higher stability than the ones stabilized by PAA1.2k-Na in an aqueous solution. However, in the initial presence of PAA-Na, amorphous calcium carbonate (ACC), vaterite or calcite were induced under various conditions. It is interesting to find that the ACC product induced by PAA1.2k-Na was more stable than that induced by PAA25k-Na in an aqueous solution as well as in a dry state. It was also found that the CaCO3 particles formed through the nano-aggregation mechanism might be induced by the strong inhibiting effects of PAA-Na, while the spherulitic growth mechanism might be due to the insufficient inhibiting efficiency for the crystallization. These results suggest that the selective interaction of PAA-Na with CaCO3 at different stages as well as the inhibiting strength varied with the chain lengths of PAA-Na could play an important role for the controlling of the crystal nucleation and growth during the crystallization process.
Similar content being viewed by others
Article PDF
References
S. Mann, “Biomineralization: Principles and Concepts in Bioinorganic Material Chemistry,” Oxford University Press, New York, 2001.
E. Bauerlein, “Biomineralization: Progress in Biology, Molecular Biology and Application,” Wiley-VCH, Weinheim, Germany, 2004.
K. Gorna, R. Muñoz-Espí, F. Gröhn, and G. Wegner, Macromol. Biosci., 7, 163 (2007).
Z. Tang, N. A. Kotov, S. Magonov, and B. Ozturk, Nat. Mater., 2, 413 (2003).
L. Addadi and S. Weiner, Nature, 389, 912 (1997).
B. L. Smith, Nature, 399, 761 (1999).
S. Mann and G. A. Ozin, Nature, 382, 313 (1996).
J. Küther, R. Seshadri, W. Knoll, and W. Tremel, J. Mater. Chem., 8, 641 (1998).
H. Wei, Q. Shen, Y. Zhao, D.-J. Wang, and D.-F. Xu, J. Cryst. Growth, 250, 516 (2003).
X. Xu, J. T. Han, and K. Cho, Chem. Mater., 16, 1740 (2004).
J. T. Han, X. Xu, D. H. Kim, and K. Cho, Chem. Mater., 17, 136 (2005).
J. Peric, M. Vucak, R. Krstulovic, L. Brecevic, and D. Kralj, Thermochimica Acta, 277, 175 (1996).
H. Cölfen and S. Mann, Angew. Chem., Int. Ed., 42, 2350 (2003).
G. Xu, N. Yao, I. A. Akasay, and J. T. Groves, J. Am. Chem. Soc., 120, 11977 (1998).
D.-K. Keum, K. Naka, and Y. Chujo, Bull. Chem. Soc. Jpn., 76, 1687 (2003).
H. Cölfen and M. Antonietti, Langmuir, 14, 582 (1998).
L. Brecevic, V. Nöthig-Laslo, D. Kralj, and S. Popovic, J. Chem. Soc., Faraday Trans., 92, 1017 (1996).
Z. Zhang, D. Gao, H. Zhao, C. Xie, G. Guan, D. Wang, and S.-H. Yu, J. Phys. Chem. B, 110, 8613 (2006).
N. Goldenfeld, J. Cryst. Growth, 84, 601 (1987).
J.-P. Andreassen, J. Cryst. Growth, 274, 256 (2005).
A.-X. Xu, Y. Ma, and H. Cölfen, J. Mater. Chem., 17, 415 (2007).
H. Cölfen and S. Mann, Angew. Chem., Int. Ed., 44, 5576 (2005).
K. Naka and Y. Chujo, Chem. Mater., 13, 3245 (2001).
T. Kato, A. Sugawara, and N. Hosoda, Adv. Mater., 14, 869 (2002).
H. Cölfen, Curr. Opin. Colloid Interface Sci., 8, 23 (2003).
H. Cölfen and L. Qi, Chem. Eur. J., 7, 106 (2001).
N. Ueyama, T. Hosoi, Y. Yamada, M. Doi, T. Okamura, and A. Nakamura, Macromolecules, 31, 7119 (1998).
F. Manoli and E. Dalas, J. Cryst. Growth, 222, 293 (2001).
T. Kato, T. Suzuki, T. Amamiya, T. Irie, M. Komiyama, and H. Yui, Supramol. Sci., 5, 411 (1998).
L. A. Gower and D. A. Tirrell, J. Cryst. Growth, 191, 153 (1998).
S.-H. Yu and H. Cölfen, J. Mater. Chem., 14, 2124 (2004).
K. Naka, D.-K. Keum, Y. Tanaka, and Y. Chujo, Bull. Chem. Soc. Jpn., 77, 827 (2004).
K. Naka, S.-C. Huang, and Y. Chujo, Langmuir, 22, 7760 (2006).
M. Sedlák, M. Antonietti, and H. Cölfen, Macromol. Chem. Phys., 199, 247 (1998).
Joint Committee on Powder Diffraction Standards—International Center for Diffraction Data, file no. 13-192 (Vaterite).
Joint Committee on Powder Diffraction Standards—International Center for Diffraction Data, file no. 47-1743 (Calcite).
M. S. Rao, Bull. Chem. Soc. Jpn., 46, 1414 (1973).
D. Chakrabarty and S. Mahapatra, J. Mater. Chem., 9, 2953 (1999).
L. Wang, I. Sondi, and E. Matijevic, J. Colloid Interface Sci., 218, 545 (1999).
A. López-Macipe, J. Gómez-Morales, and R. Rodríguez-Clemente, J. Cryst. Growth, 166, 1015 (1996).
D. Kralj, L. Brecevic, and J. Kontrec, J. Cryst. Growth, 177, 248 (1997).
L. Addadi, S. Raz, and S. Weiner, Adv. Mater., 15, 959 (2003).
S.-C. Huang, K. Naka, and Y. Chujo, Langmuir, in press (2007).
K. Naka, Y. Tanaka, and Y. Chujo, Langmuir, 18, 3655 (2002).
F. Fantinel, J. Rieger, F. Molnar, and P. Hübler, Langmuir, 20, 2539 (2004).
A. Kotachi, T. Miura, and H. Imai, Chem. Mater., 16, 3191 (2004).
N. Koga, Y. Nakagoe, and H. Tanaka, Thermochim. Acta, 318, 239 (1998).
N. Spanos and P. G. Koutsoukos, J. Phys. Chem. B, 102, 6679 (1998).
M. Balz, H. A. Therese, J. Li, J. S. Gutmann, M. Kappl, L. Nasdala, W. Hofmeister, H.-J. Butt, and W. Tremel, Adv. Funct. Mater., 15, 683 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, SC., Naka, K. & Chujo, Y. Effect of Molecular Weights of Poly(acrylic acid) on Crystallization of Calcium Carbonate by the Delayed Addition Method. Polym J 40, 154–162 (2008). https://doi.org/10.1295/polymj.PJ2007162
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2007162
Keywords
This article is cited by
-
The effect of polyacrylic acid and reaction conditions on nanocluster formation of precipitated calcium carbonate on microcellulose
Cellulose (2017)
-
Crispatotrochus-mimicking coatings improve the flexural properties of organic fibres
Journal of Materials Science (2013)
-
A carbonate controlled-addition method for size-controlled calcium carbonate spheres by carboxylic acid-terminated poly(amidoamine) dendrimers
Polymer Journal (2010)