Abstract
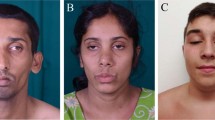
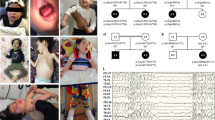
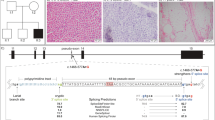
We report the cases of two Spanish pediatric patients with hypotonia, muscle weakness and feeding difficulties at birth. Whole-exome sequencing (WES) uncovered two new homozygous VAMP1 (Vesicle Associated Membrane Protein 1) splicing variants, NM_014231.5:c.129+5 G > A in the boy patient (P1) and c.341-24_341-16delinsAGAAAA in the girl patient (P2). This gene encodes the vesicle-associated membrane protein 1 (VAMP1) that is a component of a protein complex involved in the fusion of synaptic vesicles with the presynaptic membrane. VAMP1 has a highly variable C-terminus generated by alternative splicing that gives rise to three main isoforms (A, B and D), being VAMP1A the only isoform expressed in the nervous system. In order to assess the pathogenicity of these variants, expression experiments of RNA for VAMP1 were carried out. The c.129+5 G > A and c.341-24_341-16delinsAGAAAA variants induced aberrant splicing events resulting in the deletion of exon 2 (r.5_131del; p.Ser2TrpfsTer7) in the three isoforms in the first case, and the retention of the last 14 nucleotides of the 3′ of intron 4 (r.340_341ins341-14_341-1; p.Ile114AsnfsTer77) in the VAMP1A isoform in the second case. Pathogenic VAMP1 variants have been associated with autosomal dominant spastic ataxia 1 (SPAX1) and with autosomal recessive presynaptic congenital myasthenic syndrome (CMS). Our patients share the clinical manifestations of CMS patients with two important differences: they do not show the typical electrophysiological pattern that suggests pathology of pre-synaptic neuromuscular junction, and their muscular biopsies present hypertrophic fibers type 1. In conclusion, our data expand both genetic and phenotypic spectrum associated with VAMP1 variants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data of the study (variants and phenotypes) have been submitted to Global Variome shared LOVD (LOVD3) database (https://databases.lovd.nl/shared/genes/VAMP1; individual ID 00437926 (P1) and 00437931 (P2)). Further original sequencing and experimental data are available upon reasonable request.
References
Trimble WS, Cowan DM, Scheller RH. VAMP-1: a synaptic vesicle-associated integral membrane protein. Proc Natl Acad Sci USA. 1988;85:4538–42.
Raptis A, Torrejon-Escribano B, Gomez de Aranda I, Blasi J. Distribution of synaptobrevin/VAMP 1 and 2 in rat brain. J Chem Neuroanat. 2005;30:201–11.
Elferink LA, Trimble WS, Scheller RH. Two vesicle-associated membrane protein genes are differentially expressed in the rat central nervous system. J Biol Chem. 1989;264:11061–4.
Hasan N, Corbin D, Hu C. Fusogenic pairings of vesicle-associated membrane proteins (VAMPs) and plasma membrane t-SNAREs-VAMP5 as the exception. PLoS One. 2010;5:e14238.
Yan C, Jiang J, Yang Y, Geng X, Dong W. The function of VAMP2 in mediating membrane fusion: an overview. Front Mol Neurosci. 2022;15:948160.
Liu Y, Sugiura Y, Lin W. The role of synaptobrevin1/VAMP1 in Ca2+-triggered neurotransmitter release at the mouse neuromuscular junction. J Physiol. 2011;589:1603–18.
Berglund L, Hoffmann HJ, Dahl R, Petersen TE. VAMP-1 has a highly variable C-terminus generated by alternative splicing. Biochem Biophys Res Commun. 1999;264:777–80.
Bourassa CV, Meijer IA, Merner ND, Grewal KK, Stefanelli MG, Hodgkinson K, et al. VAMP1 mutation causes dominant hereditary spastic ataxia in Newfoundland families. Am J Hum Genet. 2012;91:548–52.
Shen XM, Scola RH, Lorenzoni PJ, Kay CS, Werneck LC, Brengman J, et al. Novel synaptobrevin-1 mutation causes fatal congenital myasthenic syndrome. Ann Clin Transl Neurol. 2017;4:130–38.
Salpietro V, Lin W, Delle Vedove A, Storbeck M, Liu Y, Efthymiou S, et al. Homozygous mutations in VAMP1 cause a presynaptic congenital myasthenic syndrome. Ann Neurol. 2017;81:597–603.
Monies D, Abouelhoda M, AlSayed M, Alhassnan Z, Alotaibi M, Kayyali H, et al. The landscape of genetic diseases in Saudi Arabia based on the first 1000 diagnostic panels and exomes. Hum Genet. 2017;136:921–39.
Al-Muhaizea MA, AlQuait L, AlRasheed A, AlHarbi S, Albader AA, AlMass R, et al. Pyrostigmine therapy in a patient with VAMP1-related congenital myasthenic syndrome. Neuromuscul Disord. 2020;30:611–15.
Polavarapu K, Vengalil S, Preethish-Kumar V, Arunachal G, Nashi S, Mohan D, et al. Recessive VAMP1 mutations associated with severe congenital myasthenic syndromes - A recognizable clinical phenotype. Eur J Paediatr Neurol. 2021;31:54–60.
Luque J, Mendes I, Gomez B, Morte B, Lopez de Heredia M, Herreras E, et al. CIBERER: Spanish national network for research on rare diseases: A highly productive collaborative initiative. Clin Genet. 2022;101:481–93.
Arteche-Lopez A, Gomez Rodriguez MJ, Sanchez Calvin MT, Quesada-Espinosa JF, Lezana Rosales JM, Palma Milla C, et al. Towards a change in the diagnostic algorithm of autism spectrum disorders: evidence supporting whole exome sequencing as a first-tier test. Genes (Basel). 2021;12:560.
Sutton RB, Fasshauer D, Jahn R, Brunger AT. Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 A resolution. Nature. 1998;395:347–53.
Zimmermann J, Trimbuch T, Rosenmund C. Synaptobrevin 1 mediates vesicle priming and evoked release in a subpopulation of hippocampal neurons. J Neurophysiol. 2014;112:1559–65.
Pang ZP, Sudhof TC. Cell biology of Ca2+-triggered exocytosis. Curr Opin Cell Biol. 2010;22:496–505.
Pinero TA, Soukarieh O, Rolain M, Alvarez K, Lopez-Kostner F, Torrezan GT, et al. MLH1 intronic variants mapping to + 5 position of splice donor sites lead to deleterious effects on RNA splicing. Fam Cancer. 2020;19:323–36.
Freund M, Asang C, Kammler S, Konermann C, Krummheuer J, Hipp M, et al. A novel approach to describe a U1 snRNA binding site. Nucleic Acids Res. 2003;31:6963–75.
Yeo G, Burge CB. Maximum entropy modeling of short sequence motifs with applications to RNA splicing signals. J Comput Biol. 2004;11:377–94.
Crehalet H, Latour P, Bonnet V, Attarian S, Labauge P, Bonello N, et al. U1 snRNA mis-binding: a new cause of CMT1B. Neurogenetics. 2010;11:13–9.
Supek F, Lehner B, Lindeboom RGH. To NMD or Not To NMD: nonsense-mediated mRNA decay in cancer and other genetic diseases. Trends Genet. 2021;37:657–68.
Ohno K, Takeda JI, Masuda A. Rules and tools to predict the splicing effects of exonic and intronic mutations. Wiley Interdiscip Rev RNA. 2018;9:e1451.
Acknowledgements
We thank the patients and their families for their contribution.
Funding
This work was supported by the Spanish Instituto de Salud Carlos III (ISCIII) and European Regional Development Fund (ERDF) (PI17/00487 and PI20/00150 to FM-A, PI21/00050 to LAP-J). FJC-V and LdPF were supported by fellowship from the Instituto de Investigación Hospital 12 de Octubre (i + 12) and MER-G was supported by fellowship from ISCIII and ERDF (PI17/00487) and FJC-V by fellowship from ISCIII and ERDF (PI20/00150).
Author information
Authors and Affiliations
Contributions
FJC-V, MER-G, and LdPF performed the experimental work; AH-L performed histopathological studies; AA-L, BM, MS and LAP-J performed the analysis of sequencing data; AC and PQ-F examined the patients and characterized the clinical features of the disease; FM-A and FJC-V interpreted data; FM-A supervised the study and wrote the original draft of the manuscript. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
LAP-J is founding partner and scientific advisor of qGenomics Laboratory. All other authors declare no competing commercial interest.
Ethical approval
The Ethic Committee of the Instituto de Investigación Hospital 12 de Octubre (i + 12) approved the study. The study was carried out in accordance with the Declaration of Helsinki (2013). Written informed consent was obtained from the patient’s parents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cotrina-Vinagre, F.J., Rodríguez-García, M.E., del Pozo-Filíu, L. et al. Expanding the genetic and phenotypic spectrum of congenital myasthenic syndrome: new homozygous VAMP1 splicing variants in 2 novel individuals. J Hum Genet 69, 187–196 (2024). https://doi.org/10.1038/s10038-024-01228-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-024-01228-7