Abstract
Background
There are emerging evidences that support more aggressive feeding advancement among preterm infants. Our NICU had conservative feeding advancement guidelines that delayed enteral feeding and prolonged central line use. We aimed to reduce time to full feeds among infants born ≤ 32 0/7 weeks from 12.8 days to 8 days.
Methods
A multidisciplinary team implemented evidence-based feeding guidelines using quality improvement methods. Days to full enteral feeds, central line days, necrotizing enterocolitis (NEC) rates, and extrauterine growth restriction (EUGR) rates were analyzed.
Results
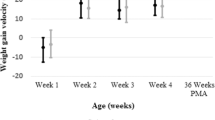
Average days to full enteral feeds decreased from 12.8 to 7.7 days and from 17.5 to 9.1 days for infants born ≤ 32 0/7 weeks and ≤ 28 0/7 weeks respectively, without significant change in NEC rate. Central line days decreased by 35%. Insignificant improvement in EUGR rate was found.
Conclusions
Faster feeding advancement guidelines led to earlier full enteral feeds and reduced central line utilization without increasing complications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL, Wrage LA, Poole WK. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics. 2006;117:1253–61.
Stephens BE, Walden RV, Gargus RA, Tucker R, McKinley L, Mance M, et al. First-week protein and energy intakes are associated with 18-month developmental outcomes in extremely low birth weight infants. Pediatrics. 2009;123:1337–43.
Franz AR, Pohlandt F, Bode H, Mihatsch WA, Sander S, Kron M, et al. Intrauterine, early neonatal, and postdischarge growth and neurodevelopmental outcome at 5.4 years in extremely preterm infants after intensive neonatal nutritional support. Pediatrics. 2009;123:e101–9.
Martin CR, Brown YF, Ehrenkranz RA, O’Shea TM, Allred EN, Belfort MB, et al. Nutritional practices and growth velocity in the first month of life in extremely premature infants. Pediatrics. 2009;124:649–57.
Roggero P, Giannì ML, Orsi A, Amato O, Piemontese P, Liotto N, et al. Implementation of nutritional strategies decreases postnatal growth restriction in preterm infants. PLoS One. 2012;7:e51166.
Chan SH, Johnson MJ, Leaf AA, Vollmer B. Nutrition and neurodevelopmental outcomes in preterm infants: a systematic review. Acta Paediatr. 2016;105:587–99.
Morgan J, Young L, McGuire W. Slow advancement of enteral feed volumes to prevent necrotising enterocolitis in very low birth weight infants. Cochrane Database Syst Rev. 2013;3:CD001241.
Hanson C, Sundermeier J, Dugick L, Lyden E, Anderson‐Berry AL. Implementation, process, and outcomes of nutrition best practices for infants < 1500 g. Nutr Clin Pr. 2011;26:614–24.
McKenzie BL, Edmonds L, Thomson R, Haszard JJ, Houghton LA. Nutrition practices and predictors of postnatal growth in preterm infants during hospitalization: a longitudinal study. J Pedia Gastroenterol Nutr. 2018;66:312–7.
Stevens TP, Shields E, Campbell D, Combs A, Horgan M, La Gamma EF, et al. Variation in enteral feeding practices and growth outcomes among very premature infants: a report from the New York State Perinatal Quality Collaborative. Am J Perinatol. 2016;2:009–19.
Patole S, De Klerk N. Impact of standardised feeding regimens on incidence of neonatal necrotising enterocolitis: a systematic review and meta-analysis of observational studies. Arch Dis Child Fetal Neonatal Ed. 2005;90:F147–51.
Shah SD, Dereddy N, Jones TL, Dhanireddy R, Talati AJ. Early versus delayed human milk fortification in very low birth weight infants–a randomized controlled trial. J Pedia. 2016;174:126–31. e121
Mimouni FB, Nathan N, Ziegler EE, Lubetzky R, Mandel D. The use of multinutrient human milk fortifiers in preterm infants: a systematic review of unanswered questions. Clin Perinatol. 2017;44:173–8.
Senterre T. Practice of enteral nutrition in very low birth weight and extremely low birth weight infants. Nutr Care Preterm Infants. 2014;110:201–14
Henderson G, Craig S, Brocklehurst P, McGuire W. Enteral feeding regimens and necrotising enterocolitis in preterm infants: a multicentre case–control study. Arch Dis Child Fetal Neonatal Ed. 2009;94:F120–3.
Berseth CL, Bisquera JA, Paje VU. Prolonging small feeding volumes early in life decreases the incidence of necrotizing enterocolitis in very low birth weight infants. Pediatrics. 2003;111:529–34.
Book LS, Herbst JJ, Jung AL. Comparison of fast-and slow-feeding rate schedules to the development of necrotizing enterocolitis. J Pediatr. 1976;89:463–6.
Kennedy KA, Tyson JE. Rapid versus slow rate of advancement of feedings for promoting growth and preventing necrotizing enterocolitis in parenterally fed low‐birth‐weight infants. Cochrane Database Syst Rev. 1998;CD001241.
Kennedy KA, Tyson JE. Early versus delayed initiation of progressive enteral feedings for parenterally fed low birth weight or preterm infants. Cochrane Database Syst Rev. 2000;CD001970.
Oddie SJ, Young L, McGuire W. Slow advancement of enteral feed volumes to prevent necrotising enterocolitis in very low birth weight infants. Cochrane Database Syst Rev. 2017;8:CD001241.
Rayyis SF, Ambalavanan N, Wright L, Carlo WA. Randomized trial of “slow” versus “fast” feed advancements on the incidence of necrotizing enterocolitis in very low birth weight infants. J Pedia. 1999;134:293–7.
Christensen R, Henry E, Wiedmeier S, Burnett J, Lambert D. Identifying patients, on the first day of life, at high-risk of developing parenteral nutrition-associated liver disease. J Perinatol. 2007;27:284.
Veenstra M, Danielson L, Brownie E, Saba M, Natarajan G, Klein M. Enteral nutrition and total parenteral nutrition components in the course of total parenteral nutrition-associated cholestasis in neonatal necrotizing enterocolitis. Surgery. 2014;156:578–83.
Robinson DT, Ehrenkranz RA. Parenteral nutrition-associated cholestasis in small for gestational age infants. J Pedia. 2008;152:59–62.
Beale E, Nelson R, Bucciarelli R, Donnelly W, Eitzman D. Intrahepatic cholestasis associated with parenteral nutrition in premature infants. Pediatrics. 1979;64:342–7.
Teitelbaum DH. Parenteral nutrition-associated cholestasis. Curr Opin Pedia. 1997;9:270–5.
Willis TC, Carter BA, Rogers SP, Hawthorne KM, Hicks PD, Abrams SA. High rates of mortality and morbidity occur in infants with parenteral nutrition-associated cholestasis. J Parenter Enter Nutr. 2010;34:32–37.
Nagata E, Brito AS, Matsuo T. Nosocomial infections in a neonatal intensive care unit: incidence and risk factors. Am J Infect Control. 2002;30:26–31.
Manzoni P, Farina D, Leonessa M, d’Oulx EA, Galletto P, Mostert M, et al. Risk factors for progression to invasive fungal infection in preterm neonates with fungal colonization. Pediatrics. 2006;118:2359–64.
Milstone AM, Reich NG, Advani S, Yuan G, Bryant K, Coffin SE, et al. Catheter dwell time and CLABSIs in neonates with PICCs: a multicenter cohort study. Pediatrics. 2013;132:e1609–15.
Bizzarro MJ, Sabo B, Noonan M, Bonfiglio M-P, Northrup V, Diefenbach K, et al. A quality improvement initiative to reduce central line-associated bloodstream infections in a neonatal intensive care unit. Infect Control Hosp Epidemiol. 2010;31:241–8.
Schulman J, Stricof R, Stevens TP, Horgan M, Gase K, Holzman IR, et al. Statewide NICU central-line-associated bloodstream infection rates decline after bundles and checklists. Pediatrics. 2011;127:436–44.
Fenton TR, Kim JH. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pedia. 2013;13:59.
Gordon A, Greenhalgh M, McGuire W. Early planned removal of umbilical venous catheters to prevent infection in newborn infants. Cochrane Database Syst Rev. 2017;10:CD012142.
Centers for Disease Control and Prevention (CfDCaP). NHSN patient safety component key terms.
Vermont Oxford Network. Manual of operations, part 2, Release 20.0. 2016.
Ogrinc G, Davies L, Goodman D, Batalden P, Davidoff F, Stevens D. SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): revised publication guidelines from a detailed consensus process. J Contin Educ Nurs. 2015;46:501–7.
Kuschel CA, Harding JE. Protein supplementation of human milk for promoting growth in preterm infants. Cochrane Database Syst. Rev. 2000;2:CD000433.
Fenton TR, Premji SS, Al‐Wassia H, Sauve RS. Higher versus lower protein intake in formula‐fed low birth weight infants. Cochrane Database Syst Rev. 2014;4:CD003959.
Olsen IE, Richardson DK, Schmid CH, Ausman LM, Dwyer JT. Intersite differences in weight growth velocity of extremely premature infants. Pediatrics. 2002;110:1125–32.
Acknowledgements
We thank the nutrition committee at the UMMC NICU for constantly striving toward improving the nutritional outcome of all the babies in the unit and being actively involved in this initiative. We also thank the entire staff in the NICU for participating in the discussion and implementation of the modified feeding guidelines.
Author information
Authors and Affiliations
Contributions
Dr. Chu conceptualized and designed this quality improvement project, assisted with data collection and analysis, and reviewed, critically revised and completed the final manuscript. Dr. Procaskey conceptualized and designed this quality improvement project, drafted the initial manuscript, assisted with data collection and analysis, and reviewed and critically revised the manuscript. Ms. Tripp RD conceptualized and designed this quality improvement project and reviewed and critically revised the manuscript. Ms. Naples RN and Ms. White assisted with data collection and analysis and reviewed and critically revised the manuscript. Dr. Rhein conceptualized and designed this quality improvement project, reviewed and critically revised the manuscript, and assisted with data collection and analysis. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Clinical Trial Registration: None
Supplementary information
Rights and permissions
About this article
Cite this article
Chu, S., Procaskey, A., Tripp, S. et al. Quality improvement initiative to decrease time to full feeds and central line utilization among infants born less than or equal to 32 0/7 weeks through compliance with standardized feeding guidelines. J Perinatol 39, 1140–1148 (2019). https://doi.org/10.1038/s41372-019-0398-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-019-0398-6