Abstract
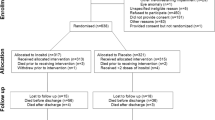
In this follow-up at 2.5 years of children from the STRIDER NZAus Trial (N = 112), in which women with singleton pregnancies affected by severe early fetal growth restriction were randomized to sildenafil citrate 75 mg daily or placebo until 32 weeks, there was no difference between groups in survival without neurosensory impairment, defined as any of cerebral palsy, deafness, blindness, cognitive delay (Bayley III cognition or language score >1 SD below mean) or motor delay: 30/56[54%] vs. 34/56[61%]; aOR = 0.74, 95%CI: 0.31, 1.77. However, children exposed to sildenafil appeared to be more likely to have cognitive delay (13/45[29%] vs. 4/40[10%]; aOR = 3.71, 95% CI: 1.01, 13.63) but less likely to have emotional-behavioural difficulties (2/43[5%] vs. 8/38[21%]; aOR = 0.19, 95%CI: 0.03, 1.00). Conclusion: maternal sildenafil treatment for severe early-onset FGR was not associated with altered survival free of neurosensory impairment at 2.5 years’ corrected age.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
The GRIT Study Group. A randomised trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. Br J Obstet Gynaecol. 2003;110:27–32.
Shah PS, Ye XY, Synnes A, Rouvinez-Bouali N, Yee W, Lee SK, et al. Prediction of survival without morbidity for infants born at under 33 weeks gestational age: a user-friendly graphical tool. Arch Dis Child Fetal Neonatal Ed. 2012;97:F110–5.
Schmidt B, Asztalos EV, Roberts RS, Robertson CM, Sauve RS, Whitfield MF. Impact of bronchopulmonary dysplasia, brain injury, and severe retinopathy on the outcome of extremely low-birth-weight infants at 18 months: results from the trial of indomethacin prophylaxis in preterms. JAMA. 2003;289:1124–9.
Baschat AA, Cosmi E, Bilardo CM, Wolf H, Berg C, Rigano S, et al. Predictors of neonatal outcome in early-onset placental dysfunction. Obstet Gynecol. 2007;109:253–61.
Merialdi M, Widmer M, Gulmezoglu AM, Abdel-Aleem H, Bega G, Benachi A, et al. WHO multicentre study for the development of growth standards from fetal life to childhood: the fetal component. BMC Pregnancy Childbirth. 2014;14:157.
Wareing M, Myers JE, O’Hara M, Baker PN. Sildenafil citrate (Viagra) enhances vasodilatation in fetal growth restriction. J Clin Endocrinol Metab. 2005;90:2550–5.
Satterfield MC, Bazer FW, Spencer TE, Wu G. Sildenafil citrate treatment enhances amino acid availability in the conceptus and fetal growth in an ovine model of intrauterine growth restriction. J Nutr. 2010;140:251–8.
Sanchez-Aparicio P, Mota-Rojas D, Nava-Ocampo AA, Trujillo-Ortega ME, Alfaro-Rodriguez A, Arch E, et al. Effects of sildenafil on the fetal growth of guinea pigs and their ability to survive induced intrapartum asphyxia. Am J Obstet Gynaecol. 2008;198:127.e1–6.
Stanley JL, Andersson IJ, Poudel R, Rueda-Clausen CF, Sibley CP, Davidge ST, et al. Sildenafil citrate rescues fetal growth in the catechol-O-methyl transferase knockout mouse model. Hypertension. 2012;59:1021–8.
von Dadelszen P, Dwinnell S, Magee LA, Carleton BC, Gruslin A, Lee B, et al. Sildenafil citrate therapy for severe early-onset intrauterine growth restriction. BJOG. 2011;118:624–8.
Panda S, Das A, Md Nowroz H. Sildenafil citrate in fetal growth restriction. J Reprod Infertil. 2014;15:168–9.
Lin TH, Su YN, Shih JC, Hsu HC, Lee CN. Resolution of high uterine artery pulsatility index and notching following sildenafil citrate treatment in a growth-restricted pregnancy. Ultrasound Obstet Gynecol. 2012;40:609–10.
Dastjerdi MV, Hosseini S, Bayani L. Sildenafil citrate and uteroplacental perfusion in fetal growth restriction. J Res Med Sci. 2012;17:632–6.
Groom KM, McCowan LM, Mackay LK, Lee AC, Gardener G, Unterscheider J, et al. STRIDER NZAus: a multicentre randomised controlled trial of sildenafil therapy in early-onset fetal growth restriction. BJOG. 2019;126:997–1006.
Sharp A, Cornforth C, Jackson R, Harrold J, Turner MA, Kenny LC, et al. Maternal sildenafil for severe fetal growth restriction (STRIDER): a multicentre, randomised, placebo-controlled, double-blind trial. Lancet Child Adolesc Health. 2017. https://doi.org/10.1016/S2352-4642(17)30173-6.
Pels A, Derks J, Elvan-Taspinar A, van Drongelen J, de Boer M, Duvekot H, et al. Maternal sildenafil vs placebo in pregnant women with severe early-onset fetal growth restriction: a randomized clinical trial. JAMA Netw Open. 2020;3:e205323.
von Dadelszen P, Audibert F, Bujold E, Bone JN, Sandhu A, Li J, et al. Halting the Canadian STRIDER randomised controlled trial of sildenafil for severe, early-onset fetal growth restriction: ethical, methodological, and pragmatic considerations. BMC Res Notes. 2022;15:244.
Groom KM, Ganzevoort W, Alfirevic Z, Lim K, Papageorghiou AT. Clinicians should stop prescribing sildenafil for fetal growth restriction (FGR): comment from the STRIDER Consortium. Ultrasound Obstet Gynecol. 2018;52:295–6.
Terstappen F, Richter AE, Lely AT, Hoebeek FE, Elvan-Taspinar A, Bos AF, et al. Prenatal use of sildenafil in fetal growth restriction and its effect on neonatal tissue oxygenation-a retrospective analysis of hemodynamic data from participants of the Dutch STRIDER Trial. Front Pediatr. 2020;8:595693.
Rakhanova Y, Almawi WY, Aimagambetova G, Riethmacher D. The effects of sildenafil citrate on intrauterine growth restriction: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2023;23:409.
Pels A, Ganzevoort W, Kenny LC, Baker PN, von Dadelszen P, Gluud C, et al. Interventions affecting the nitric oxide pathway versus placebo or no therapy for fetal growth restriction in pregnancy. Cochrane Database Syst Rev. 2023;7:CD014498.
Hitzerd E, Broekhuizen M, Mirabito Colafella KM, Glisic M, de Vries R, Koch BCP, et al. Placental effects and transfer of sildenafil in healthy and preeclamptic conditions. EBioMedicine. 2019;45:447–55.
Maher MA, Sayyed TM, Elkhouly N. Sildenafil citrate therapy for oligohydramnios: a randomized controlled trial. Obstet Gynecol. 2017;129:615–20.
Turner J, Dunn L, Tarnow-Mordi W, Flatley C, Flenady V, Kumar S. Safety and efficacy of sildenafil citrate to reduce operative birth for intrapartum fetal compromise at term: a phase 2 randomized controlled trial. Am J Obstet Gynecol. 2020;222:401–14.
De Bie FR, Basurto D, Kumar S, Deprest J, Russo FM. Sildenafil during the 2nd and 3rd trimester of pregnancy: trials and tribulations. Int J Environ Res Public Health. 2022;19:11207.
Cans C, Dolk H, Platt MJ, Colver A, Prasauskiene A, Krageloh-Mann I, et al. Recommendations from the SCPE collaborative group for defining and classifying cerebral palsy. Dev Med Child Neurol (Suppl). 2007;109:35–8.
Gioia GA, Espy KA, Isquith PK. Behavior rating inventory of executive function, preschool version (BRIEF-P). Odessa, FL: Psychological Assessment Resources; 2003.
Achenbach TM, Rescorla LA. Manual for the ASEBA preschool forms & profiles. Burlington, VT: University of Vermont, Research Center for Children, Youth, & Families; 2000.
Raat H, Landgraf JM, Oostenbrink R, Moll HA, Essink-Bot ML. Reliability and validity of the Infant and Toddler Quality of Life Questionnaire (ITQOL) in a general population and respiratory disease sample. Qual Life Res. 2007;16:445–60.
Landgraf JM, Vogel I, Oostenbrink R, van Baar ME, Raat H. Parent-reported health outcomes in infants/toddlers: measurement properties and clinical validity of the ITQOL-SF47. Qual Life Res. 2013;22:635–46.
WHO Multicentre Growth Reference Study Group. WHO Child Growth Standards based on length/height, weight and age. Acta Paediatr Suppl. 2006;450:76–85.
National Heart Lung and Blood Institute. Report of the second task force on blood pressure control in children-1987. Pediatrics. 1987;79:1–25.
Spittle AJ, Treyvaud K, Doyle LW, Roberts G, Lee KJ, Inder TE, et al. Early emergence of behavior and social-emotional problems in very preterm infants. J Am Acad Child Adolesc Psychiatry. 2009;48:909–18.
Torrance HL, Bloemen MC, Mulder EJ, Nikkels PG, Derks JB, de Vries LS, et al. Predictors of outcome at 2 years of age after early intrauterine growth restriction. Ultrasound Obstet Gynecol. 2010;36:171–7.
Baschat AA. Neurodevelopment following fetal growth restriction and its relationship with antepartum parameters of placental dysfunction. Ultrasound Obstet Gynecol. 2011;37:501–14.
Pierce CM, Zhang MH, Jonsson B, Iorga D, Cheruvu N, Balagtas CC, et al. Efficacy and safety of IV sildenafil in the treatment of newborn infants with, or at risk of, persistent pulmonary hypertension of the newborn (PPHN): a multicenter, randomized, placebo-controlled trial. J Pediatr. 2021;237:154.–61.e3.
Jackson W, Gonzalez D, Smith PB, Ambalavanan N, Atz AM, Sokol GM, et al. Safety of sildenafil in extremely premature infants: a phase I trial. J Perinatol. 2022;42:31–6.
Harris SL, McKinlay C, Groom K, Beker F, Kochar A, Gill A. Neonatal cardiovascular function after antenatal sildenafil for severe, early-onset intrauterine growth restriction: a substudy of the STRIDER NZAus randomized placebo-controlled trial. J Pediatr X. 2019:1;100009.
Yazdani A, Khoja Z, Johnstone A, Dale L, Rampakakis E, Wintermark P. Sildenafil improves brain injury recovery following term neonatal hypoxia-ischemia in male rat pups. Dev Neurosci. 2016;38:251–63.
Engels J, Elting N, Braun L, Bendix I, Herz J, Felderhoff-Müser U, et al. Sildenafil enhances quantity of immature neurons and promotes functional recovery in the developing ischemic mouse brain. Dev Neurosci. 2017;39:287–97.
Yazdani A, Howidi B, Shi MZ, Tugarinov N, Khoja Z, Wintermark P. Sildenafil improves hippocampal brain injuries and restores neuronal development after neonatal hypoxia-ischemia in male rat pups. Sci Rep. 2021;11:22046.
Lees CC, Marlow N, van Wassenaer-Leemhuis A, Arabin B, Bilardo CM, Brezinka C, et al. 2 year neurodevelopmental and intermediate perinatal outcomes in infants with very preterm fetal growth restriction (TRUFFLE): a randomised trial. Lancet. 2015;385:2162–72.
Groom KM, David AL. The role of aspirin, heparin, and other interventions in the prevention and treatment of fetal growth restriction. Am J Obstet Gynecol. 2018;218:S829–40.
de Jong F, Monuteaux MC, van Elburg RM, Gillman MW, Belfort MB. Systematic review and meta-analysis of preterm birth and later systolic blood pressure. Hypertension. 2012;59:226–34.
Andraweera PH, Condon B, Collett G, Gentilcore S, Lassi ZS. Cardiovascular risk factors in those born preterm - systematic review and meta-analysis. J Dev Orig Health Dis. 2021;12:539–54.
Juonala M, Cheung MM, Sabin MA, Burgner D, Skilton MR, Kähönen M, et al. Effect of birth weight on life-course blood pressure levels among children born premature: the Cardiovascular Risk in Young Finns Study. J Hypertens. 2015;33:1542–8.
Chatmethakul T, Roghair RD. Risk of hypertension following perinatal adversity: IUGR and prematurity. J Endocrinol. 2019;242:T21–32.
Aatola H, Koivistoinen T, Tuominen H, Juonala M, Lehtimäki T, Viikari JSA, et al. Influence of child and adult elevated blood pressure on adult arterial stiffness: the Cardiovascular Risk in Young Finns Study. Hypertension. 2017;70:531–6.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:e20171904.
Lee PA, Chernausek SD, Hokken-Koelega AC, Czernichow P. International Small for Gestational Age Advisory Board consensus development conference statement: management of short children born small for gestational age. Pediatrics. 2003;111:1253–61.
Hokken-Koelega AC, De Waal WJ, Sas TC, Van Pareren Y, Arends NJ. Small for gestational age (SGA): endocrine and metabolic consequences and effects of growth hormone treatment. J Pediatr Endocrinol Metab. 2004;17:463–9.
Burakevych N, McKinlay CJ, Alsweiler JM, Wouldes TA, Harding JE. Bayley-III motor scale and neurological examination at 2 years do not predict motor skills at 4.5 years. Dev Med Child Neurol. 2017;59:216–23.
Acknowledgements
We thank the women and children who participated in this study. The following were non-author contributors to this study: Frank Bloomfield, Liggins Institute, University of Auckland; John Thompson, Department of Pediatrics: Child and Youth Health, University of Auckland; Trecia Wouldes, Department of Psychological Medicine, University of Auckland; Janice Taylor, Auckland City Hospital; Helen Liley, Leith Poulsen, Jane Fox and Suzanne Bates, Mater Mothers’ Hospital, Brisbane; Marion McDonald, Royal Women’s Hospital, Melbourne; Lisa McKeown, Royal Brisbane and Women’s Hospital, Brisbane; Mei’En Lim, Safiyyah Abdul Aziz, Lori Gardiner, Emma Anderton and Carolyn Smargiassi, King Edward Memorial Hospital, Perth; Pat Ashwood, Ros Lontis, Women’s and Children’s Hospital, Adelaide; Di Leishman, Christchurch Women’s Hospital; Annemarie Lawrence, Townsville Hospital and Health Service, Townsville; Anne Lainchbury, The Royal Hospital for Women, Sydney; Gabrielle Pell, Elaine Kelly, Elisha Josev, Julianne Duff, Noni Davis and Anne-Marie Turner, Mercy Hospital for Women, Melbourne. We also acknowledge the investigators of the STRIDER NZAus Trial: Katie Groom and Joanna de Sousa, National Women’s Health, Auckland City Hospital; Glenn Gardener, Anne Tremellen, Bekki Cavallaro, Frances Maguire and Akina Kato, Mater Mothers’ Hospital, Brisbane; Julia Unterscheider and Stefan Kane, The Royal Women’s Hospital, Melbourne; Renuka Sekar, Lisa McKeown, Gillian Lack and Katie Foxcroft, Royal Brisbane and Women’s Hospital, Brisbane; Jan Dickinson and Paula Carter, King Edward Memorial Hospital, Perth; Peter Muller, Pat Ashwood and Andrea Deussen, Women’s and Children’s Hospital, Adelaide; Rosemary Reid, Christchurch Women’s Hospital; David Watson, Audra Davis and Cherie Boniface, Townsville Hospital and Health Service, Townsville; Antonia Shand, Alec Welsh and Anne Lainchbury, The Royal Hospital for Women, Sydney; Jaynana Marlow, Madeline Dymond-Cate and Trish Burke, Wellington Hospital; Susan Walker and Gabrielle Pell, Mercy Hospital for Women, Melbourne; Jon Hyett and Jane Tooher, Royal Prince Alfred Hospital, Sydney; Jonathan Morris and Kathryn Austin, Royal North Shore Hospital, Sydney.
Funding
Cure Kids, Auckland, New Zealand (3570); Royal Australian and New Zealand College of Obstetricians and Gynaecologists Te Kāhui Oranga ō Nuku Mercia Barnes Trust (3703775); Auckland Medical Research Foundation (1117001); Neurological Foundation of New Zealand (1708-PG); University of Auckland (FRDF 3714288); Leadership Fellowship, National Health and Medical Research Council of Australia (2016390). The funders had no role in study design, analysis and interpretation of data, or in the decision to publish.
Author information
Authors and Affiliations
Contributions
Conception and design: CJDM, KMG, and LM; Supervision and oversight: CJDM, KMG, LM, and MO-TB; Funding: CJDM, KMG, and LM; Data collection and processing: CJDM, CA, JLYC, AG, SLH, EH, SI, PK, KL, LM, MO-TB, GO, MS, and KMG. Analysis and interpretation of data: CJDM, KMG, and LM. Drafting of manuscript: CJDM, KMG, LM. Critical review and approval of final version: All authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McKinlay, C.J.D., Anderson, C., Cheong, J.L.Y. et al. Childhood outcomes after maternal antenatal sildenafil treatment for severe early-onset fetal growth restriction: a randomized trial (STRIDER NZAus). J Perinatol 44, 396–403 (2024). https://doi.org/10.1038/s41372-023-01838-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-023-01838-7