Abstract
Objective
This prospective study compared PIVKA-II and PT-INR levels in infants who received two vitamin K (VK) prophylactic regimens.
Methods
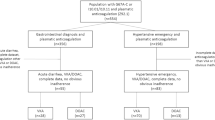
A single institution administered 119 healthy newborns 2 mg of VK syrup. Infants were assigned to a 3-time regimen (n = 56) with VK at birth, five days (5D), and 1-month-old (1 M), or a 13-time regimen (n = 63) with VK at birth, 5D, and then weekly for 11 weeks.
Results
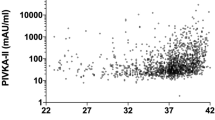
The 13-time regimen significantly lowered PIVKA-II and reduced PT-INR at 1 M in both breastfed (PIVKA-II: 18–16 mAU/mL, p = 0.02; PT-INR: 1.37–1.13, p < 0.01) and formula-fed infants (PIVKA-II: 18–15 mAU/mL, p = 0.01; PT-INR: 1.54–1.24, p < 0.01), compared to baseline measurements taken at 5D. The 3-time regimen did not significantly alter PIVKA-II levels and only improved PT-INR (2.00–1.50, p < 0.01) in formula-fed infants.
Conclusion
The 13-time VK regimen significantly enhanced coagulation profiles more effectively than the 3-time regimen.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are not publicly available due to institutional ethical policy.
References
Townsend CW. The haemorrhagic disease of the newborn. Arch Pediatr. 1894;11:559–65.
Zipursky A. Prevention of vitamin K deficiency bleeding in newborns. Br J Haematol. 1999;104:430–7.
Shearer MJ. Vitamin K deficiency bleeding (VKDB) in early infancy. Blood Rev. 2009;23:49–59.
Hand I, Noble L, Abrams SA. Vitamin K and the newborn Infant. Pediatrics. 2022;149:e2021056036.
Norman MG, Temple AR, Murphy JV. Infantile quadriceps-femoris contracture resulting from intramuscular injections. N Engl J Med. 1970;282:964–6.
Tripp JH, McNinch AW. Haemorrhagic disease and vitamin K. Arch Dis Child. 1987;62:436–7.
Araki S, Shirahata A. Vitamin K deficiency bleeding in Infancy. Nutrients. 2020;12:780.
Jullien S. Vitamin K prophylaxis in newborns. BMC Pediatr. 2021;21:350.
Sankar MJ, Chandrasekaran A, Kumar P, Thukral A, Agarwal R, Paul VK. Vitamin K prophylaxis for prevention of vitamin K deficiency bleeding: a systematic review. J Perinatol. 2016;36:S29–35.
Tada Y, Misina J. Clinical study of oral administration of VK to newborn infants. Annual Report of the Comprehensive Study of Health Problems of Newborn: 13-14 (1988).
Shirahata A, Itoh S, Takahashi Y, Nishiguchi T, Matsuda Y. Revised guidelines for vitamin K deficiency bleeding in newborns and infants (Modified Version). J Jp Ped Soc. 2011;115:705–12.
Cornelissen EA, Kollée LA, De Abreu RA, Motohara K, Monnens LA. Prevention of vitamin K deficiency in infancy by weekly administration of vitamin K. Acta Paediatr. 1993;82:656–9.
van Hasselt PM, de Koning TJ, Kvist N, de Vries E, Lundin CR, Berger R, et al. Prevention of vitamin K deficiency bleeding in breastfed infants: lessons from the Dutch and Danish biliary atresia registries. Pediatrics. 2008;121:e857–63.
Motohara K, Endo F, Matsuda I. Effect of vitamin K administration on acarboxy prothrombin (PIVKA-II) levels in newborns. Lancet. 1985;2:242–4.
Van Winckel M, De Bruyne R, Van De Velde S, Van Biervliet S. Vitamin K, an update for the paediatrician. Eur J Pediatr. 2009;168:127–34.
Motohara K, Endo F, Matsuda I. Screening for late neonatal vitamin K deficiency by acarboxyprothrombin in dried blood spots. Arch Dis Child. 1987;62:370–5.
Matsuda I, Nishiyama S, Motohara K, Endo F, Ogata T, Futagoishi Y. Late neonatal vitamin K deficiency associated with subclinical liver dysfunction in human milk-fed infants. J Pediatr. 1989;114:602–5.
Motohara K, Matsukura M, Matsuda I, Iribe K, Ikeda T, Kondo Y, et al. Severe vitamin K deficiency in breast-fed infants. J Pediatr. 1984;105:943–5.
Ashina M, Fujioka K, Nishida K, Iijima K. Neonatal vitamin K deficiency in the son of a mother with short bowel syndrome. Pediatr Int. 2018;60:991–2.
Yamada K, Meguro T, Miyaji R. Measuring the PIVKA-II levels using the PIVKAL test in multiple institutions (in Japanese). Shinryo Shinyaku (Med Cons N.-Remed). 1985;22:1408–14.
Sutor AH. New aspects of vitamin K prophylaxis. Semin Thromb Hemost. 2003;29:373–6.
Cornelissen M, von Kries R, Loughnan P, Schubiger G. Prevention of vitamin K deficiency bleeding: efficacy of different multiple oral dose schedules of vitamin K. Eur J Pediatr. 1997;156:126–30.
Greer FR, Marshall SP, Severson RR, Smith DA, Shearer MJ, Pace DG, et al. A new mixed micellar preparation for oral vitamin K prophylaxis: randomised controlled comparison with an intramuscular formulation in breast fed infants. Arch Dis Child. 1998;79:300–5.
Nishizono I, Iida S, Suzuki N, Kawada H, Murakami H, Ashihara Y, et al. Rapid and sensitive chemiluminescent enzyme immunoassay for measuring tumor markers. Clin Chem. 1991;37:1639–44.
Hunnali CR, Devi U, Kitchanan S, Sethuraman G. Three different regimens for vitamin K birth prophylaxis in infants born preterm: a randomized clinical trial. J Pediatr. 2023;255:98–104.
Khalilov Z, Ünsal A, Altuntaş N. The D-dimer reference intervals in healty term newborns. Transfus Apher Sci. 2022;61:103493.
Acknowledgements
We sincerely thank Dr. Akira Shirahata (Kitakyushu Yahata Higashi Hospital) and Dr. Yukihiro Takahashi (Todaiji Ryoiku Hospital for Children) for their helpful advice on our study design and Miyuki Sakemoto MT and Kumiko Watanabe MT for the advice concerning technical procedures and guidance on sample measurements.
Funding
This work was supported by JSPS KAKENHI (JP22K07916 [Ochiai M]), AMED (JP20ek0109481 [Ohga S]), and the Japanese Society of Obstetrical, Gynecological and Neonatal Hematology.
Author information
Authors and Affiliations
Contributions
Takahashi D, Egami N, and Ochiai M conceptualized and designed the study, designed the data collection instruments, collected the data, carried out initial analyses, drafted the initial manuscript, and critically reviewed and revised the manuscript. Hotta T measured and analyzed the collected samples and critically reviewed and revised the manuscript. Suga S conducted the initial analyses and critically reviewed and revised the manuscript. Suga S, Ishimura M, Kawaguchi C, Uchiumi T, Nishikubo T, Nogami K, and Goto K conceptualized and designed the study and critically reviewed and revised the manuscript. Ohga S supervised the study design, drafted the initial manuscript, and critically reviewed and revised the manuscript. All authors had access to the data, approved the final manuscript as submitted, and agreed to be responsible for all aspects of this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was reviewed and approved by the Fukuda Hospital institutional review board (27-1-2). Written informed consent was obtained from each parent of candidate newborns.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takahashi, D., Egami, N., Ochiai, M. et al. Vitamin K prophylaxis in neonates: comparing two different oral regimens. J Perinatol (2024). https://doi.org/10.1038/s41372-024-01981-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41372-024-01981-9