Abstract
Background
Obesity is an important cause for the precocious or early puberty. However, the association between obesity-related loci and the risk of precocious puberty as well as the effect of gene-environment interaction are unclear, especially in the Chinese children population.
Methods
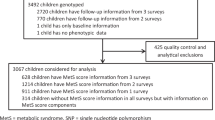
This was a case-control study using baseline data from two cohorts and hospital cases in China. 15 SNPs loci and several environmental factors were included in the analysis of 1201 participants. Chi-square test and logistic regression were used to analyze the association between SNPs and precocious puberty. Additionally, exploratory factor analysis was conducted on 13 environmental variables, and then to explore their interaction with genes on precocious puberty.
Results
The effect allele C of rs571312, and G of rs12970134 MC4R were associated with precocious puberty in girls with obesity. Regarding the gene-environment interaction, we found that when girls were in the high socioeconomic status, the rs571312 (OR: 3.996; 95% CI: 1.694–9.423) and rs12970134 (OR: 3.529; 95% CI: 1.452–8.573) risk genotypes had a greater effect on precocious puberty.
Conclusions
The obesity risk gene polymorphisms MC4R rs571312 and rs12970134 were associated with precocious puberty in Chinese girls with obesity, and girls with risk genotypes and high socioeconomic status should be given extra attention.
Impact
-
This is the first study that identified the association between rs571312 and rs12970134 of MC4R gene and precocious puberty in Chinese children.
-
We found that when girls were in the high socioeconomic status, the risk genotypes of rs571312 and rs12970134 had a greater effect on precocious puberty.
-
The results of this study have great public health implications. It is recommended that girls who are in high socioeconomic status and have a high genetic risk for early sexual maturity should closely monitor their pubertal development and consider early intervention strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The raw data reported in this paper has been deposited in the Genome Variation Map in National Genomics Data Center, China National Center for Bioinformation / Beijing Institute of Genomics, Chinese Academy of Sciences, under accession number GVM000377 that can be publicly accessible at http://bigd.big.ac.cn/gvm/getProjectDetail?project= GVM000377.
References
Abreu, A. P. & Kaiser, U. B. Pubertal development and regulation. Lancet Diabetes Endocrinol. 4, 254–264 (2016).
Brix, N. et al. Timing of puberty in boys and girls: a population-based study. Paediatr. Perinat. Epidemiol. 33, 70–78 (2019).
Lee, M. H., Kim, S. H., Oh, M., Lee, K. W. & Park, M. J. Age at menarche in Korean adolescents: trends and influencing factors. Reprod. Health 13, 121 (2016).
Meng, X., Li, S., Duan, W., Sun, Y. & Jia, C. Secular trend of age at menarche in Chinese adolescents born from 1973 to 2004. Pediatrics 140, e20170085 (2017).
Liu, Y. et al. Prevalence of precocious puberty among Chinese children: a school population-based study. Endocrine 72, 573–581 (2021).
Zhang, Y. et al. The prevalence of precocious puberty among children in Qufu City, Shandong Province, China, a population-based study. Front. Endocrinol. 13, 910119 (2022).
Berkey, C. S., Frazier, A. L., Gardner, J. D. & Colditz, G. A. Adolescence and breast carcinoma risk. Cancer 85, 2400–2409 (1999).
Day, F. R., Elks, C. E., Murray, A., Ong, K. K. & Perry, J. R. B. Puberty timing associated with diabetes, cardiovascular disease and also diverse health outcomes in men and women: the UK Biobank study. Sci. Rep. 5, 11208 (2015).
Graber, J. A. Pubertal timing and the development of psychopathology in adolescence and beyond. Horm. Behav. 64, 262–269 (2013).
Elks, C. E. et al. Thirty new loci for age at menarche identified by a meta-analysis of genome-wide association studies. Nat. Genet. 42, 1077–1085 (2010).
Day, F. R. et al. Shared genetic aetiology of puberty timing between sexes and with health-related outcomes. Nat. Commun. 6, 8842 (2015).
Ong, K. K. et al. Genetic variation in LIN28B is associated with the timing of puberty. Nat. Genet. 41, 729–733 (2009).
Karapanou, O. & Papadimitriou, A. Determinants of menarche. Reprod. Biol. Endocrin. 8, 115 (2010).
Fujimura, Y., Sekine, M. & Yamada, M. The relationship between quality of life and pubertal timing in adolescence: the Toyama birth cohort study, Japan. J. Adolesc. Health 65, 790–798 (2019).
Sun, Y., Fang, J., Wan, Y., Su, P. & Tao, F. Role of polygenic risk in susceptibility to accelerated pubertal onset following chronic stress exposure. Eur. J. Endocrinol. 181, 129–137 (2019).
Chen, C. et al. Investigating the relationship between precocious puberty and obesity: a cross-sectional study in Shanghai, China. BMJ Open 7, e014004 (2017).
Chen, Y.-C. et al. Assessing causality between childhood adiposity and early puberty: a bidirectional Mendelian randomization and longitudinal study. Metabolism 100, 153961 (2019).
Gavela-Pérez, T., Garcés, C., Navarro-Sánchez, P., López Villanueva, L. & Soriano-Guillén, L. Earlier menarcheal age in Spanish girls is related with an increase in body mass index between pre-pubertal school age and adolescence. Pediatr. Obes. 10, 410–415 (2015).
Busch, A. S., Højgaard, B., Hagen, C. P. & Teilmann, G. Obesity is associated with earlier pubertal onset in boys. J. Clin. Endocrinol. Metab. 105, dgz222 (2020).
Lee, J. M. et al. Timing of puberty in overweight versus obese boys. Pediatrics 137, e20150164 (2016).
Li, D. et al. Obesity-related genetic polymorphisms are associated with the risk of early puberty in Han Chinese girls. Clin. Endocrinol. 96, 319–327 (2022).
Jiang, Y. et al. Interaction effects of FTO rs9939609 polymorphism and lifestyle factors on obesity indices in early adolescence. Obes. Res. Clin. Pract. 13, 352–357 (2019).
Wei, B. L. et al. The MC4R SNPs, their haplotypes and gene-environment interactions on the risk of obesity. Mol. Med. 26, 77 (2020).
Hu, Y. et al. Exposure to antibiotics and precocious puberty in children: a school-based cross-sectional study in China. Environ. Res. 212, 113365 (2022).
Yu, T. et al. Effects of childhood obesity and related genetic factors on precocious puberty: protocol for a multi-center prospective cohort study. BMC Pediatr. 22, 310 (2022).
Cole, T. J. & Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 7, 284–294 (2012).
Marshall, W. A. & Tanner, J. M. Variations in pattern of pubertal changes in girls. Arch. Dis. Child. 44, 291–303 (1969).
Marshall, W. A. & Tanner, J. M. Variations in the pattern of pubertal changes in boys. Arch. Dis. Child. 45, 13–23 (1970).
Cheuiche, A. V., da Silveira, L. G., de Paula, L. C. P., Lucena, I. R. S. & Silveiro, S. P. Diagnosis and management of precocious sexual maturation: an updated review. Eur. J. Pediatr. 180, 3073–3087 (2021).
Chen, C. et al. Association between dietary patterns and precocious puberty in children: a population-based study. Int. J. Endocrinol. 2018, 4528704 (2018).
Akiyama, M. et al. Genome-wide association study identifies 112 new loci for body mass index in the Japanese population. Nat. Genet. 49, 1458–1467 (2017).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 (2015).
Perry, J. R. et al. Parent-of-origin-specific allelic associations among 106 genomic loci for age at menarche. Nature 514, 92–97 (2014).
Purcell, S. et al. Plink: PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Gong, W. et al. Effects of gene-environment interaction on obesity among Chinese adults born in the early 1960s. Genes 12, 270 (2021).
Farooqi, I. S. et al. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N. Engl. J. Med. 348, 1085–1095 (2003).
Lotta, L. A. et al. Human gain-of-function MC4R variants show signaling bias and protect against obesity. Cell 177, 597–607.e9 (2019).
Speliotes, E. K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat. Genet. 42, 937–948 (2010).
Wen, W. et al. Meta-analysis identifies common variants associated with body mass index in East Asians. Nat. Genet. 44, 307–311 (2012).
Cousminer, D. L. et al. Genome-wide association study of sexual maturation in males and females highlights a role for body mass and menarche loci in male puberty. Hum. Mol. Genet. 23, 4452–4464 (2014).
Karim, A., Qaisar, R. & Hussain, M. A. Growth and socio-economic status, influence on the age at menarche in school going girls. J. Adolesc. 86, 40–53 (2021).
Hiatt, R. A. et al. Childhood socioeconomic status and menarche: a prospective study. J. Adolesc. Health 69, 33–40 (2021).
Oelkers, L. et al. Socioeconomic status is related to pubertal development in a German cohort. Horm. Res. Paediatr. 93, 548–557 (2020).
Cheng, M. et al. The influence of socioeconomic status on menarcheal age among Chinese school-age girls in Tianjin, China. Eur. J. Pediatr. 180, 825–832 (2021).
Cerutti, J., Lussier, A. A., Zhu, Y., Liu, J. & Dunn, E. C. Associations between indicators of socioeconomic position and DNA methylation: a scoping review. Clin. Epigenetics 13, 221 (2021).
Gao, L. et al. MC4R single nucleotide polymorphisms were associated with metabolically healthy and unhealthy obesity in Chinese northern Han populations. Int. J. Endocrinol. 2019, 4328909 (2019).
Rasmussen, L. et al. The impact of lifestyle, diet and physical activity on epigenetic changes in the offspring-a systematic review. Nutrients 13, 2821 (2021).
Acknowledgements
We would like to express our sincere gratitude to all the staff, as well as all the study participants, their teachers and parents who participated in this study.
Funding
This study was supported by National Natural Science Foundation of China [82173534, 81872637].
Author information
Authors and Affiliations
Contributions
S.L., C.L. and H.K. performed study design and supervision. P.X., T.Y., J.L., J.T. and C.C. collected the data. P.X., J.L., J.T. and Y.C. carried out the data analyses and interpretation. P.X., J.L. and Y.C. drafted the manuscript. S.L. revised the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of this study.
Ethics approval and consent to participate
Informed consent was obtained by the parents or guardians of the participating children.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xue, P., Lin, J., Tang, J. et al. Association of obesity and menarche SNPs and interaction with environmental factors on precocious puberty. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03168-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03168-6