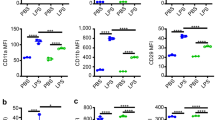
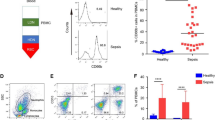
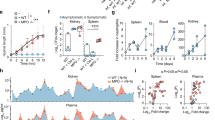
Abstract
Distinct neutrophil populations arise during certain pathological conditions. The generation of dysfunctional neutrophils during sepsis and their contribution to septicemia-related systemic immune suppression remain unclear. In this study, using an experimental sepsis model that features immunosuppression, we identified a novel population of pathogenic CD200Rhigh neutrophils that are generated during the initial stages of sepsis and contribute to systemic immune suppression by enhancing regulatory T (Treg) cells. Compared to their CD200Rlow counterparts, sepsis-generated CD200Rhigh neutrophils exhibit impaired autophagy and dysfunction, with reduced chemotactic migration, superoxide anion production, and TNF-α production. Increased soluble CD200 blocks autophagy and neutrophil maturation in the bone marrow during experimental sepsis, and recombinant CD200 treatment in vitro can induce neutrophil dysfunction similar to that observed in CD200Rhigh neutrophils. The administration of an α-CD200R antibody effectively reversed neutrophil dysfunction by enhancing autophagy and protecting against a secondary infection challenge, leading to increased survival. Transcriptome analysis revealed that CD200Rhigh neutrophils expressed high levels of Igf1, which elicits the generation of Treg cells, while the administration of an α-CD200R antibody inhibited Treg cell generation in a secondary infection model. Taken together, our findings revealed a novel CD200Rhigh neutrophil population that mediates the pathogenesis of sepsis-induced systemic immunosuppression by generating Treg cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hotchkiss RS, Moldawer LL, Opal SM, Reinhart K, Turnbull IR, Vincent JL. Sepsis and septic shock. Nat Rev Dis Prim. 2016;2:16045. https://doi.org/10.1038/nrdp.2016.45.
Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348:138–50. https://doi.org/10.1056/NEJMra021333.
Vincent JL, Sun Q, Dubois MJ. Clinical trials of immunomodulatory therapies in severe sepsis and septic shock. Clin Infect Dis. 2002;34:1084–93. https://doi.org/10.1086/339549.
Presneill JJ, Harris T, Stewart AG, Cade JF, Wilson JW. A randomized phase II trial of granulocyte-macrophage colony-stimulating factor therapy in severe sepsis with respiratory dysfunction. Am J Respir Crit Care Med. 2002;166:138–43. https://doi.org/10.1164/rccm.2009005.
Zeiher BG, Steingrub J, Laterre PF, Dmitrienko A, Fukiishi Y, Abraham E, et al. LY315920NA/S-5920, a selective inhibitor of group IIA secretory phospholipase A2, fails to improve clinical outcome for patients with severe sepsis. Crit Care Med. 2005;33:1741–8. https://doi.org/10.1097/01.ccm.0000171540.54520.69.
Guillon A, Preau S, Aboab J, Azabou E, Jung B, Silva S, et al. Preclinical septic shock research: why we need an animal ICU. Ann Intensive Care. 2019;9:66. https://doi.org/10.1186/s13613-019-0543-6.
Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34:1589–96. https://doi.org/10.1097/01.CCM.0000217961.75225.E9.
Liu VX, Fielding-Singh V, Greene JD, Baker JM, Iwashyna TJ, Bhattacharya J, et al. The timing of early antibiotics and hospital mortality in sepsis. Am J Respir Crit Care Med. 2017;196:856–63. https://doi.org/10.1164/rccm.201609-1848OC.
Hotchkiss RS, Monneret G, Payen D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol. 2013;13:862–74. https://doi.org/10.1038/nri3552.
Alves-Filho JC, Spiller F, Cunha FQ. Neutrophil paralysis in sepsis. Shock. 2010;34:15–21. https://doi.org/10.1097/SHK.0b013e3181e7e61b.
Patel JM, Sapey E, Parekh D, Scott A, Dosanjh D, Gao F, et al. Sepsis induces a dysregulated neutrophil phenotype that is associated with increased mortality. Mediators Inflamm. 2018;2018:4065362. https://doi.org/10.1155/2018/4065362.
Kwok AJ, Allcock A, Ferreira RC, Cano-Gamez E, Smee M, Burnham KL, et al. Neutrophils and emergency granulopoiesis drive immune suppression and an extreme response endotype during sepsis. Nat Immunol. 2023;24:767–79. https://doi.org/10.1038/s41590-023-01490-5.
Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 2006;6:173–82. https://doi.org/10.1038/nri1785.
Shi T, Ma Y, Yu L, Jiang J, Shen S, Hou Y, et al. Cancer immunotherapy: a focus on the regulation of immune checkpoints. Int J Mol Sci. 2018;19:1389. https://doi.org/10.3390/ijms19051389.
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020;30:660–69. https://doi.org/10.1038/s41422-020-0343-4.
McBride MA, Patil TK, Bohannon JK, Hernandez A, Sherwood ER, Patil NK. Immune checkpoints: novel therapeutic targets to attenuate sepsis-induced immunosuppression. Front Immunol. 2020;11:624272. https://doi.org/10.3389/fimmu.2020.624272.
Noh H, Hu J, Wang X, Xia X, Satelli A, Li S. Immune checkpoint regulator PD-L1 expression on tumor cells by contacting CD11b positive bone marrow derived stromal cells. Cell Commun Signal. 2015;13:14. https://doi.org/10.1186/s12964-015-0093-y.
Melichar B, Nash MA, Lenzi R, Platsoucas CD, Freedman RS. Expression of costimulatory molecules CD80 and CD86 and their receptors CD28, CTLA-4 on malignant ascites CD3+ tumour-infiltrating lymphocytes (TIL) from patients with ovarian and other types of peritoneal carcinomatosis. Clin Exp Immunol. 2000;119:19–27. https://doi.org/10.1046/j.1365-2249.2000.01105.x.
Zhang Y, Zhou Y, Lou J, Li J, Bo L, Zhu K, et al. PD-L1 blockade improves survival in experimental sepsis by inhibiting lymphocyte apoptosis and reversing monocyte dysfunction. Crit Care. 2010;14:R220. https://doi.org/10.1186/cc9354.
Riffelmacher T, Clarke A, Richter FC, Stranks A, Pandey S, Danielli S, et al. Autophagy-dependent generation of free fatty acids is critical for normal neutrophil differentiation. Immunity. 2017;47:466–80.e5. https://doi.org/10.1016/j.immuni.2017.08.005.
Evrard M, Kwok IWH, Chong SZ, Teng KWW, Becht E, Chen J, et al. Developmental analysis of bone marrow neutrophils reveals populations specialized in expansion, trafficking, and effector functions. Immunity. 2018;48:364–79.e8. https://doi.org/10.1016/j.immuni.2018.02.002.
Eash KJ, Greenbaum AM, Gopalan PK, Link DC. CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. J Clin Investig. 2010;120:2423–31. https://doi.org/10.1172/jci41649.
Sledzinska A, Menger L, Bergerhoff K, Peggs KS, Quezada SA. Negative immune checkpoints on T lymphocytes and their relevance to cancer immunotherapy. Mol Oncol. 2015;9:1936–65. https://doi.org/10.1016/j.molonc.2015.10.008.
Deniset JF, Surewaard BG, Lee WY, Kubes P. Splenic Ly6G(high) mature and Ly6G(int) immature neutrophils contribute to eradication of S. pneumoniae. J Exp Med. 2017;214:1333–50. https://doi.org/10.1084/jem.20161621.
Shrestha S, Lee JM, Hong CW. Autophagy in neutrophils. Korean J Physiol Pharmacol. 2020;24:1–10. https://doi.org/10.4196/kjpp.2020.24.1.1.
Martin KR, Wong HL, Witko-Sarsat V, Wicks IP. G-CSF - a double edge sword in neutrophil mediated immunity. Semin Immunol. 2021;54:101516 https://doi.org/10.1016/j.smim.2021.101516.
Leveque-El Mouttie L, Vu T, Lineburg KE, Kuns RD, Bagger FO, Teal BE, et al. Autophagy is required for stem cell mobilization by G-CSF. Blood. 2015;125:2933–6. https://doi.org/10.1182/blood-2014-03-562660.
Chen EY, Chu S, Gov L, Kim YK, Lodoen MB, Tenner AJ, et al. CD200 modulates macrophage cytokine secretion and phagocytosis in response to poly(lactic co-glycolic acid) microparticles and films. J Mater Chem B. 2017;5:1574–84. https://doi.org/10.1039/C6TB02269C.
Bhattacharya A, Wei Q, Shin JN, Abdel Fattah E, Bonilla DL, Xiang Q, et al. Autophagy is required for neutrophil-mediated inflammation. Cell Rep. 2015;12:1731–9. https://doi.org/10.1016/j.celrep.2015.08.019.
Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13:159–75. https://doi.org/10.1038/nri3399.
Phan QT, Sipka T, Gonzalez C, Levraud JP, Lutfalla G, Nguyen-Chi M. Neutrophils use superoxide to control bacterial infection at a distance. PLoS Pathog. 2018;14:e1007157. https://doi.org/10.1371/journal.ppat.1007157.
Twito T, Chen Z, Khatri I, Wong K, Spaner D, Gorczynski R. Ectodomain shedding of CD200 from the B-CLL cell surface is regulated by ADAM28 expression. Leuk Res. 2013;37:816–21. https://doi.org/10.1016/j.leukres.2013.04.014.
Morgan HJ, Rees E, Lanfredini S, Powell KA, Gore J, Gibbs A, et al. CD200 ectodomain shedding into the tumor microenvironment leads to NK cell dysfunction and apoptosis. J Clin Invest. 2022;132:e150750. https://doi.org/10.1172/JCI150750.
Chousterman BG, Arnaud M. Is there a role for hematopoietic growth factors during sepsis? Front Immunol. 2018;9:1015. https://doi.org/10.3389/fimmu.2018.01015.
Schwulst SJ, Muenzer JT, Peck-Palmer OM, Chang KC, Davis CG, McDonough JS, et al. Bim siRNA decreases lymphocyte apoptosis and improves survival in sepsis. Shock. 2008;30:127–34. https://doi.org/10.1097/shk.0b013e318162cf17.
Luan YY, Yao YM, Xiao XZ, Sheng ZY. Insights into the apoptotic death of immune cells in sepsis. J Interferon Cytokine Res. 2015;35:17–22. https://doi.org/10.1089/jir.2014.0069.
Liu D, Huang SY, Sun JH, Zhang HC, Cai QL, Gao C, et al. Sepsis-induced immunosuppression: mechanisms, diagnosis and current treatment options. Mil Med Res. 2022;9:56. https://doi.org/10.1186/s40779-022-00422-y.
Chang KC, Burnham CA, Compton SM, Rasche DP, Mazuski RJ, McDonough JS, et al. Blockade of the negative co-stimulatory molecules PD-1 and CTLA-4 improves survival in primary and secondary fungal sepsis. Crit Care. 2013;17:R85 https://doi.org/10.1186/cc12711.
Nascimento DC, Melo PH, Pineros AR, Ferreira RG, Colon DF, Donate PB, et al. IL-33 contributes to sepsis-induced long-term immunosuppression by expanding the regulatory T cell population. Nat Commun. 2017;8:14919. https://doi.org/10.1038/ncomms14919.
Johannesson B, Sattler S, Semenova E, Pastore S, Kennedy-Lydon TM, Sampson RD, et al. Insulin-like growth factor-1 induces regulatory T cell-mediated suppression of allergic contact dermatitis in mice. Dis Model Mech. 2014;7:977–85. https://doi.org/10.1242/dmm.015362.
Brahmamdam P, Inoue S, Unsinger J, Chang KC, McDunn JE, Hotchkiss RS. Delayed administration of anti-PD-1 antibody reverses immune dysfunction and improves survival during sepsis. J Leukoc Biol. 2010;88:233–40. https://doi.org/10.1189/jlb.0110037.
Huang X, Venet F, Wang YL, Lepape A, Yuan Z, Chen Y, et al. PD-1 expression by macrophages plays a pathologic role in altering microbial clearance and the innate inflammatory response to sepsis. Proc Natl Acad Sci USA. 2009;106:6303–8. https://doi.org/10.1073/pnas.0809422106.
Miller BC, Zhao Z, Stephenson LM, Cadwell K, Pua HH, Lee HK, et al. The autophagy gene ATG5 plays an essential role in B lymphocyte development. Autophagy. 2008;4:309–14. https://doi.org/10.4161/auto.5474.
Zhang Y, Morgan MJ, Chen K, Choksi S, Liu ZG. Induction of autophagy is essential for monocyte-macrophage differentiation. Blood. 2012;119:2895–905. https://doi.org/10.1182/blood-2011-08-372383.
Lee HK, Mattei LM, Steinberg BE, Alberts P, Lee YH, Chervonsky A, et al. In vivo requirement for Atg5 in antigen presentation by dendritic cells. Immunity. 2010;32:227–39. https://doi.org/10.1016/j.immuni.2009.12.006.
Rozman S, Yousefi S, Oberson K, Kaufmann T, Benarafa C, Simon HU. The generation of neutrophils in the bone marrow is controlled by autophagy. Cell Death Differ. 2015;22:445–56. https://doi.org/10.1038/cdd.2014.169.
Bo L, Wang F, Zhu J, Li J, Deng X. Granulocyte-colony stimulating factor (G-CSF) and granulocyte-macrophage colony stimulating factor (GM-CSF) for sepsis: a meta-analysis. Crit Care. 2011;15:R58. https://doi.org/10.1186/cc10031.
Akkaya M, Aknin ML, Akkaya B, Barclay AN. Dissection of agonistic and blocking effects of CD200 receptor antibodies. PLoS ONE. 2013;8:e63325. https://doi.org/10.1371/journal.pone.0063325.
Vieira P, Rajewsky K. The half-lives of serum immunoglobulins in adult mice. Eur J Immunol. 1988;18:313–6. https://doi.org/10.1002/eji.1830180221.
Talebian F, Liu JQ, Liu Z, Khattabi M, He Y, Ganju R, et al. Melanoma cell expression of CD200 inhibits tumor formation and lung metastasis via inhibition of myeloid cell functions. PLoS ONE. 2012;7:e31442. https://doi.org/10.1371/journal.pone.0031442.
Pilch Z, Tonecka K, Skorzynski M, Sas Z, Braniewska A, Kryczka T, et al. The pro-tumor effect of CD200 expression is not mimicked by agonistic CD200R antibodies. PLoS ONE. 2019;14:e0210796. https://doi.org/10.1371/journal.pone.0210796.
Lee SK, Kim SD, Kook M, Lee HY, Ghim J, Choi Y, et al. Phospholipase D2 drives mortality in sepsis by inhibiting neutrophil extracellular trap formation and down-regulating CXCR2. J Exp Med. 2015;212:1381–90. https://doi.org/10.1084/jem.20141813.
Park MY, Kim HS, Lee HY, Zabel BA, Bae YS. Novel CD11b(+)Gr-1(+)Sca-1(+) myeloid cells drive mortality in bacterial infection. Sci Adv. 2020;6:eaax8820. https://doi.org/10.1126/sciadv.aax8820.
Bae GH, Kim YS, Park JY, Lee M, Lee SK, Kim JC, et al. Unique characteristics of lung-resident neutrophils are maintained by PGE2/PKA/Tgm2-mediated signaling. Blood. 2022;140:889–99. https://doi.org/10.1182/blood.2021014283.
Acknowledgements
This work was supported by Basic Science Research Program (NRF-2020M3A9D3038435, NRF-2017R1A5A1014560) and the Korea Initiative for Fostering the University of Research and Innovation Program (NRF-2020M3H1A1077095) through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning and by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI22C2004).
Author information
Authors and Affiliations
Contributions
Y.K. designed and performed the research, analyzed the data, and wrote the paper. Y.J. designed and performed the research, analyzed data, and wrote the paper. G.B. designed the experiments and analyzed the data. J.K. performed the research and analyzed the data. M.L. designed the experiments and analyzed the data. B.A.Z. wrote the paper. Y.B. designed the research and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
YSK, YSJ, and YSB have filed a patent related to this work. The other authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, Y.S., Jeong, Y.S., Bae, G.H. et al. CD200Rhigh neutrophils with dysfunctional autophagy establish systemic immunosuppression by increasing regulatory T cells. Cell Mol Immunol 21, 349–361 (2024). https://doi.org/10.1038/s41423-024-01136-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-024-01136-y