Abstract
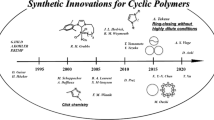
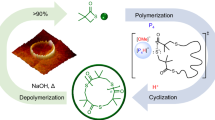
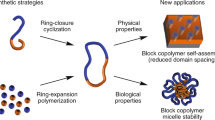
This focus review describes the status quo of synthetic routes to cyclic compounds and cyclic polymers via spontaneous cyclization processes using (1) rotaxane chemistry and (2) dynamic covalent chemistry. Systems with rotaxane-based structures undergo spontaneous and selective cyclization of two self-complementary molecules and a macromolecular rotaxane switch, where the relative position of each component can be controlled, which results in a topology change from linear to cyclic. Systems based on dynamic covalent chemistry use exchange reactions of bis(2,2,6,6-tetramethylpiperidin-1-yl)disulfide (BiTEMPS) units that behave as stable linkages at r.t. and exchange disulfide bonds upon heating. Controlling the dynamic nature of BiTEMPS-based low-molecular-weight compounds and polymers thus provides cyclic topologies via spontaneous and selective cyclization processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Villiers A. Sur la fermentation de la fécule par l’action du ferment butyrique. Compt. Rend. 1891;112:536–40.
Szejtli J. Introduction and general overview of cyclodextrin chemistry. Chem Rev. 1998;98:1743–53.
Pedersen CJ. Cyclic polyethers and their complexes with metal salts. J Am Chem Soc. 1967;89:2495–6.
Freeman WA, Mock WL, Shih NY. Cucurbituril. J Am Chem Soc. 1981;103:7367–8.
Ogoshi T, Yamagishi TA, Nakamoto Y. Pillar-shaped macrocyclic hosts pillar[n]arenes: new key players for supramolecular chemistry. Chem Rev. 2016;116:7937–8002.
Antesberger J, Cave GWV, Ferrarelli MC, Heaven MW, Raston CL & Atwood JL. Solvent-free, direct synthesis of supramolecular nano-capsules. Chem Commun. 2005:892–4.
Schneider HJ, Yatsimirsky AK. Selectivity in supramolecular host-guest complexes. Chem Soc Rev. 2008;37:263–77.
Yu GC, Jie KC, Huang FH. Supramolecular amphiphiles based on host-guest molecular recognition motifs. Chem Rev. 2015;115:7240–303.
Crowley JD, Goldup SM, Lee AL, Leigh DA, McBurney RT. Active metal template synthesis of rotaxanes, catenanes and molecular shuttles. Chem Soc Rev. 2009;38:1530–41.
Miyagawa N, Watanabe M, Matsuyama T, Koyama Y, Moriuchi T, Hirao T, et al. Successive catalytic reactions specific to Pd-based rotaxane complexes as a result of wheel translation along the axle. Chem Commun. 2010;46:1920–2.
Thordarson P, Bijsterveld EJA, Rowan AE, Nolte RJM. Epoxidation of polybutadiene by a topologically linked catalyst. Nature. 2003;424:915–8.
Harada A, Hashidzume A, Yamaguchi H, Takashima Y. Polymeric rotaxanes. Chem Rev. 2009;109:5974–6023.
Haino T. Designer supramolecular polymers with specific molecular recognitions. Polym J. 2019;51:303–18.
Goto Y, Ohta A, Sako Y, Yamagishi Y, Murakami H, Suga H. Reprogramming the translation initiation for the synthesis of physiologically stable cyclic peptides. ACS Chem Biol. 2008;3:120–9.
Zorzi A, Deyle K, Heinis C. Cyclic peptide therapeutics: past, present and future. Curr Opin Chem Biol. 2017;38:24–9.
Laurent BA, Grayson SM. Synthetic approaches for the preparation of cyclic polymers. Chem Soc Rev. 2009;38:2202–13.
Dong BT, Dong YQ, Du FS, Li ZC. Controlling polymer topology by atom transfer radical self-condensing vinyl polymerization of p-(2-bromoisobutyloylmethyl)styrene. Macromolecules. 2010;43:8790–8.
Castro-Osma JA, Alonso-Moreno C, Garcia-Martinez JC, Fernandez-Baeza J, Sanchez-Barba LF, Lara-Sanchez A et al. Ring-opening (ROP) versus ring-expansion (REP) polymerization of epsilon-caprolactone to give linear or cyclic polycaprolactones. Macromolecules. 2013;46:6388–94.
Bielawski CW, Benitez D, Grubbs RH. Synthesis of cyclic polybutadiene via ring-opening metathesis polymerization: The importance of removing trace linear contaminants. J Am Chem Soc. 2003;125:8424–5.
Kricheldorf HR. Cyclic polymers: synthetic strategies and physical properties. J Polym Sci Part A: Polym Chem. 2010;48:251–84.
Culkin DA, Jeong WH, Csihony S, Gomez ED, Balsara NR, Hedrick JL, et al. Zwitterionic polymerization of lactide to cyclic poly(lactide) by using N-heterocyclic carbene organocatalysts. Angew Chem Int Ed. 2007;46:2627–30.
Willans CE, Sinenkov MA, Fukin GK, Sheridan K, Lynam JM, Trifonov AA, et al. Lanthanide chloride complexes of amine-bis(phenolate) ligands and their reactivity in the ring-opening polymerization of epsilon-caprolactone. Dalton Trans. 2008:3592–8.
Kammiyada H, Konishi A, Ouchi M, Sawamoto M. Ring-expansion living cationic polymerization via reversible activation of a hemiacetal ester bond. ACS Macro Lett. 2013;2:531–4.
Roland CD, Li H, Abboud KA, Wagener KB, Veige AS. Cyclic polymers from alkynes. Nat Chem. 2016;8:791–6.
Li Z, Qu L, Zhu W, Liu JA, Chen JQ, Sun P, et al. Self-accelerating click reaction for preparing cyclic polymers from unconjugated vinyl monomers. Polymer. 2018;137:54–62.
Zhang K, Lackey MA, Wu Y, Tew GN. Universal cyclic polymer templates. J Am Chem Soc. 2011;133:6906–9.
Honda S, Yamamoto T, Tezuka Y. Tuneable enhancement of the salt and thermal stability of polymeric micelles by cyclized amphiphiles. Nat Commun. 2013;4:1574–9.
Oike H, Mouri T, Tezuka Y. A cyclic macromonomer designed for a novel polymer network architecture having both covalent and physical linkages. Macromolecules. 2001;34:6229–34.
Chang YA, Waymouth RM. Recent progress on the synthesis of cyclic polymers via ring-expansion strategies. J Polym Sci Part A: Polym Chem. 2017;55:2892–902.
Yamaguchi G, Higaki Y, Otsuka H, Takahara A. Reversible radical ring-crossover polymerization of an alkoxyamine-containing dynamic covalent macrocycle. Macromolecules. 2005;38:6316–20.
Schulz M, Tanner S, Barqawi H, Binder WH. Macrocyclization of polymers via ring-closing metathesis and azide/alkyne-“click”-reactions: an approach to cyclic polyisobutylenes. J Polym Sci Part A: Polym Chem. 2010;48:671–80.
Thompson MC, Busch DH. Reactions of coordinated ligands .6. Metal ion control in synthesis of planar nickel(2) complexes of alpha-diketo-bis-mercaptoimines. J Am Chem Soc. 1964;86:213–7.
Mandolini L, Masci B. Kinetic evidence for template effect of added cations on rate of formation of benzo-18-crown-6 in water solution. J Am Chem Soc. 1977;99:7709–10.
Jiang Y, Zhang Z, Wang D, Hadjichristidis N. An efficient and general strategy toward the synthesis of polyethylene-based cyclic polymers. Macromolecules. 2018;51:3193–202.
Sun P, Chen JQ, Liu JA, Zhang K. Self-accelerating click reaction for cyclic polymer. Macromolecules. 2017;50:1463–72.
Josse T, De Winter J, Gerbaux P, Coulembier O. Cyclic polymers by ring-closure strategies. Angew Chem Int Ed. 2016;55:13944–58.
Laurent BA, Grayson SM. An efficient route to well-defined macrocyclic polymers via “Click” cyclization. J Am Chem Soc. 2006;128:4238–9.
Tezuka Y, Oike H. Topological polymer chemistry: systematic classification of nonlinear polymer topologies. J Am Chem Soc. 2001;123:11570–6.
Yamamoto T, Hosokawa M, Nakamura M, Sato S, Isono T, Tajima K, et al. Synthesis, isolation, and properties of all head-to-tail cyclic poly(3-hexylthiophene): fully delocalized exciton over the defect-free ring polymer. Macromolecules. 2018;51:9284–93.
Takahashi A, Yuzaki R, Ishida Y & Kameyama A. Controlled ring-expansion polymerization of thiiranes based on cyclic aromatic thiourethane initiator. J. Polym. Sci. Part A: Polym. Chem. 2019;57:2442–9.
Narumi A, Yamada M, Unno Y, Kumaki J, Binder WH, Enomoto K, et al. Evaluation of ring expansion-controlled radical polymerization system by AFM observation. ACS Macro Lett. 2019;8:634–8.
Kricheldorf HR, Weidner SM, Scheliga F. Ring-expansion polymerization (REP) of L-lactide with cyclic tin(II) bisphenoxides. Eur Polym J. 2019;116:256–64.
Narumi A, Hasegawa S, Yanagisawa R, Tomiyama M, Yamada M, Binder WH, et al. Ring expansion-controlled radical polymerization: Synthesis of cyclic polymers and ring component quantification based on SEC-MALS analysis. React Funct Polym. 2016;104:1–8.
Zhang K, Lackey MA, Cui J, Tew GN. Gels based on cyclic polymers. J Am Chem Soc. 2011;133:4140–8.
Boydston AJ, Xia Y, Kornfield JA, Gorodetskaya IA, Grubbs RH. Cyclic ruthenium-alkylidene catalysts for ring-expansion metathesis polymerization. J Am Chem Soc. 2008;130:12775–82.
Haque FM, Grayson SM. The synthesis, properties and potential applications of cyclic polymers. Nat Chem. 2020;12:433–44.
Zhang SS, Tezuka Y, Zhang ZBA, Li N, Zhang W, Zhu XL. Recent advances in the construction of cyclic grafted polymers and their potential applications. Polym Chem. 2018;9:677–86.
Yamamoto T, Tezuka Y. Topological polymer chemistry: a cyclic approach toward novel polymer properties and functions. Polym Chem. 2011;2:1930–41.
Xue M, Yang Y, Chi XD, Yan XZ, Huang FH. Development of pseudorotaxanes and rotaxanes: from synthesis to stimuli-responsive motions to applications. Chem Rev. 2015;115:7398–501.
Raymo FM, Stoddart JF. Interlocked macromolecules. Chem Rev. 1999;99:1643–63.
Erbas-Cakmak S, Leigh DA, McTernan CT, Nussbaumer AL. Artificial molecular machines. Chem Rev. 2015;115:10081–206.
Arunachalam M, Gibson HW. Recent developments in polypseudorotaxanes and polyrotaxanes. Prog Polym Sci. 2014;39:1043–73.
Forgan RS, Sauvage JP, Stoddart JF. Chemical topology: complex molecular knots, links, and entanglements. Chem Rev. 2011;111:5434–64.
Takata T. Polyrotaxane and polyrotaxane network: supramolecular architectures based on the concept of dynamic covalent bond chemistry. Polym J. 2006;38:1–20.
Dietrichbuchecker CO, Sauvage JP. Interlocking of molecular threads—from the statistical approach to the templated synthesis of catenands. Chem Rev. 1987;87:795–810.
Jin YH, Yu C, Denman RJ, Zhang W. Recent advances in dynamic covalent chemistry. Chem Soc Rev. 2013;42:6634–54.
Rowan SJ, Cantrill SJ, Cousins GRL, Sanders JKM, Stoddart JF. Dynamic covalent chemistry. Angew Chem Int Ed. 2002;41:898–952.
Otsuka H. Reorganization of polymer structures based on dynamic covalent chemistry: polymer reactions by dynamic covalent exchanges of alkoxyamine units. Polym J. 2013;45:879–91.
Sauvage JP, Dietrichbuchecker CO. Molecular catenanes, rotaxanes and knots; New York: Wiley; 1999.
Badjic JD, Balzani V, Credi A, Silvi S, Stoddart JF. A molecular elevator. Science. 2004;303:1845–9.
Bruns CJ, Stoddart JF. The nature of the mechanical bond: from molecules to machines. Hoboken, NJ: Wiley; 2016. https://doi.org/10.1002/9781119044123.
Takata T. Switchable polymer materials controlled by rotaxane macromolecular switches. ACS Cent Sci. 2020;6:129–43.
Aoki D, Takata T. Mechanically linked supramolecular polymer architectures derived from macromolecular [2]rotaxanes: synthesis and topology transformation. Polymer. 2017;128:276–96.
Takata T, Aoki D. Topology-transformable polymers: linear-branched polymer structural transformation via the mechanical linking of polymer chains. Polym J. 2018;50:127–47.
Aoki D, Uchida S, Nakazono K, Koyama Y, Takata T. Macromolecular [2]rotaxanes: effective synthesis and characterization. ACS Macro Lett. 2013;2:461–5.
De BoG, De Winter J, Gerbaux P, Fustin CA. Rotaxane-based mechanically linked block copolymers. Angew Chem Int Ed. 2011;50:9093–6.
Chen Z, Aoki D, Uchida S, Marubayashi H, Nojima S, Takata T. Effect of component mobility on the properties of macromolecular [2] rotaxanes. Angew Chem Int Ed. 2016;55:2778–81.
Makiguchi K, Satoh T, Kakuchi T. Diphenyl phosphate as an efficient cationic organocatalyst for controlled/living ring-opening polymerization of delta-valerolactone and epsilon-caprolactone. Macromolecules. 2011;44:1999–2005.
Nakazono K, Ogawa T, Takata T. Synthesis of a cyclic poly(methyl acrylate) via topological transformation of a [1]rotaxane. Mater Chem Front. 2019;3:2716–20.
Valentina S, Ogawa T, Nakazono K, Aoki D, Takata T. Efficient synthesis of cyclic block copolymers by rotaxane protocol by linear/cyclic topology transformation. Chem Eur J. 2016;22:8759–62.
Sato H, Aoki D, Takata T. Synthesis and star/linear topology transformation of a mechanically linked ABC terpolymer. ACS Macro Lett. 2016;5:699–703.
Aoki D, Uchida S, Takata T Angew. Chem. Int. Ed. 2015;54:6770.
Aoki D, Uchida S, Takata T. Synthesis and characterization of a mechanically linked transformable polymer. Polym J. 2014;46:546–52.
Aoki D, Aibara G, Uchida S, Takata T. A rational entry to cyclic polymers via selective cyclization by self-assembly and topology transformation of linear polymers. J Am Chem Soc. 2017;139:6791–4.
Schneiderman DK, Hillmyer MA. Aliphatic polyester block polymer design. Macromolecules. 2016;49:2419–28.
Hodge P. Entropically driven ring-opening polymerization of strainless organic macrocycles. Chem Rev. 2014;114:2278–312.
Amador AG, Watts A, Neitzel AE, Hillmyer MA. Entropically driven macrolide polymerizations for the synthesis of aliphatic polyester copolymers using titanium isopropoxide. Macromolecules. 2019;52:2371–83.
llarduya AM & Guerra SM Ring opening polymerization of macrocyclic oligoesters derived from renewable sources. Polym. Chem. 2020;11:4850–60.
Takahashi A, Goseki R, Otsuka H. Thermally adjustable dynamic disulfide linkages mediated by highly air-stable 2,2,6,6-tetramethyl-piperidine-1-sulfanyl (TEMPS) radicals. Angew Chem Int Ed. 2017;56:2016–21.
Hoyle CE, Lowe AB, Bowman CN. Thiol-click chemistry: a multifaceted toolbox for small molecule and polymer synthesis. Chem Soc Rev. 2010;39:1355–87.
Iha RK, Wooley KL, Nystrom AM, Burke DJ, Kade MJ, Hawker CJ. Applications of orthogonal “click” chemistries in the synthesis of functional soft materials. Chem Rev. 2009;109:5620–86.
Tsurumi N, Takashima R, Aoki D, Kuwata S, Otsuka H. A strategy toward cyclic topologies based on the dynamic behavior of a bis(hindered amino)disulfide linker. Angew Chem Int Ed. 2020;59:4269–73.
Takashima R, Aoki D, Otsuka H. Rational entry to cyclic polymers via thermally induced radical ring-expansion polymerization of macrocycles with one bis(hindered amino)disulfide linkage. Macromolecules. 2020;53:4670–7.
Yokochi H, Takashima R, Aoki D, Otsuka H. Using the dynamic behavior of macrocyclic monomers with a bis(hindered amino)disulfide linker for the preparation of end-functionalized polymers. Polym Chem. 2020;11:3557–63.
Acknowledgements
The author would like to express his gratitude to all his past and present colleagues for their valuable contributions to this focus review. The author is deeply indebted to Prof. Toshikazu Takata (Hiroshima University) and Prof. Hideyuki Otsuka (Tokyo Institute of Technology) for their continuous encouragement and constructive discussions. This research was supported by KAKENHI grants 16K17910, 18K14272, and 20H02783 from the Japan Society for the Promotion of Science (JSPS), the Mizuho Foundation for the Promotion of Science, the Eno Scientific Foundation, the Iketani Science and Technology Foundation, and JST PRESTO grant JPMJPR18L1 (Japan).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aoki, D. A rational entry to cyclic polymers via spontaneous and selective cyclization reactions. Polym J 53, 257–269 (2021). https://doi.org/10.1038/s41428-020-00422-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-020-00422-6