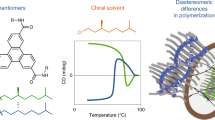
Abstract
Supramolecular chiral emergence from achiral molecules is studied with amphiphilic polypeptides upon forming molecular assemblies of nanosheets and vesicles in water. A newly designed amphiphilic polypeptide contains a hydrophilic poly(Sar) block, two hydrophobic helical blocks, and a π-conjugate block (SL-π-D). The two helical blocks are (L-Leu-Aib)4 and (D-Leu-Aib)4 of right- and left-handed helices, respectively, to compensate for the helical chirality resulting in the achiral molecule of SL-π-D. SL-π-D self-assembled in trifluoroethanol (TFE)/water solutions into uniform nanosheets and vesicles depending on the TFE content in the solutions. The induced Cotton effect due to exciton coupling appears in the achiral π-conjugate block upon self-assembling into nanosheets and vesicles. This supramolecular chiral emergence is enhanced in vesicles compared to nanosheets. The origin of the chirality intrinsically arises from the structure of (helix block)-(π-conjugate block)-(helix block) and further from vesicular morphology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Shen YH, Wang YF, Hamley IW, Qi W, Su RX, He ZM. Chiral self-assembly of peptides: toward the design of supramolecular polymers with enhanced chemical and biological functions. Prog Polym Sci. 2021;123:101469.
Park KS, Xue ZY, Patel BB, An H, Kwok JJ, Kafle P, et al. Chiral emergence in multistep hierarchical assembly of achiral conjugated polymers. Nat Commun. 2022;13:2738.
Niori T, Sekine T, Watanabe J, Furukawa T, Takezoe H. Distinct ferroelectric smectic liquid crystals consisting of banana shaped achiral molecules. J Mater Chem. 1996;6:1231–3.
Lehmann A, Alaasar M, Poppe M, Poppe S, Prehm M, Nagaraj M, et al. Stereochemical rules govern the soft self-assembly of achiral compounds: understanding the heliconical liquid-crystalline phases of bent-core mesogens. Chem Eur J. 2020;26:4714–33.
Mendes AC, Baran ET, Reis RL, Azevedo HS. Self-assembly in nature: using the principles of nature to create complex nanobiomaterials. WIREs Nanomed Nanobiotechnol. 2013;5:582–612.
Uji H, Ogawa J, Itabashi K, Imai T, Kimura S. Compartmentalized host spaces accommodating guest aromatic molecules in a chiral way in a helix-peptide-aromatic framework. Chem Commun. 2018;54:12483–6.
Zhou HC, Kitagawa S. Metal-organic frameworks (MOFs). Chem Soc Rev. 2014;43:5415–8.
Mantion A, Massuger L, Rabu P, Palivan C, McCusker LB, Taubert A. Metal-peptide frameworks (MPFs): “Bioinspired” metal organic frameworks. J Am Chem Soc. 2008;130:2517–26.
Dong JQ, Liu Y, Cui Y. Artificial metal-peptide assemblies: bioinspired assembly of peptides and metals through space and across length scales. J Am Chem Soc. 2021;143:17316–36.
Heinz-Kunert SL, Pandya A, Dang VT, Tran PN, Ghosh S, McElheny D, et al. Assembly of pi-stacking helical peptides into a porous and multivariable proteomimetic framework. J Am Chem Soc. 2022;144:7001–9.
Kimura S. Primary to quaternary structures of molecular assemblies. Polym J. 2019;51:739–51.
Kanzaki T, Horikawa Y, Makino A, Sugiyama J, Kimura S. Nanotube and three-way nanotube formation with nonionic amphiphilic block peptides. Macromol Biosci. 2008;8:1026–33.
Ueda M, Uesaka A, Kimura S. Selective disruption of each part of Janus molecular assemblies by lateral diffusion of stimuli-responsive amphiphilic peptides. Chem Commun. 2015;51:1601–4.
Watabe N, Kim CJ, Kimura S. Fusion and fission of molecular assemblies of amphiphilic polypeptides generating small vesicles from nanotubes. Biopolymers. 2017;108:e22903.
Son K, Takeoka S, Ito Y, Ueda M. End-sealing of peptide nanotubes by cationic amphiphilic polypeptides and their salt-responsive accordion-like opening and closing behavior. Biomacromolecules. 2022;23:2785–92.
Ueda M, Makino A, Imai T, Sugiyama J, Kimura S. Transformation of peptide nanotubes into a vesicle via fusion driven by stereo-complex formation. Chem Commun. 2011;47:3204–6.
Matsui H, Ueda M, Hara I, Kimura S. Precise control of nanoparticle surface by host-guest chemistry for delivery to tumor. RSC Adv. 2015;5:35346–51.
Itagaki T, Ito Y, Ueda M. Peptide flat-rod formation by precise arrangement among enantiomeric hydrophobic helices. J Colloid Interface Sci. 2022;617:129–35.
Kakuta M, Hirata M, Kimura Y. Stereoblock polylactides as high-performance bio-based polymers. Polym Rev. 2009;49:107–40.
Kim CJ, Ueda M, Imai T, Sugiyama J, Kimura S. Tuning the viscoelasticity of peptide vesicles by adjusting hydrophobic helical blocks comprising amphiphilic polypeptides. Langmuir. 2017;33:5423–9.
Itagaki T, Ueda Y, Itabashi K, Uji H, Kimura S. Joining nanotubes comprising nucleobase-carrying amphiphilic polypeptides. Chimia. 2018;72:842–7.
Storrs RW, Truckses D, Wemmer DE. Helix propagation in trifluoroethanol solutions. Biopolymers. 1992;32:1695–702.
Luo PZ, Baldwin RL. Mechanism of helix induction by trifluoroethanol: a framework for extrapolating the helix-forming properties of peptides from trifluoroethanol/water mixtures back to water. Biochemistry. 1997;36:8413–21.
Ishikawa T, Morita T, Kimura S. Unique helical triangle molecular geometry induced by dipole-dipole interactions. Bull Chem Soc Jpn. 2007;80:1483–91.
Liu MH, Zhang L, Wang TY. Supramolecular chirality in self-assembled systems. Chem Rev. 2015;115:7304–97.
Inai Y, Tagawa K, Takasu A, Hirabayashi T, Oshikawa T, Yamashita M. Induction of one-handed helical screw sense in achiral peptide through the domino effect based on interacting its N-terminal amino group with chiral carboxylic acid. J Am Chem Soc. 2000;122:11731–2.
Tschierske C. Development of structural complexity by liquid-crystal self-assembly. Angew Chem Int Ed. 2013;52:8828–78.
Ueda M, Seo S, Nair BG, Muller S, Takahashi E, Arai T, et al. End-sealed high aspect ratio hollow nanotubes encapsulating an anticancer drug: torpedo-shaped peptidic nanocapsules. ACS Nano. 2019;13:305–12.
Acknowledgements
This research was supported partially by JSPS KAKENHI Grant Numbers JP19K15375 and JP 22K14553, and The Kyoto University Foundation. TEM analysis was supported by the Analysis and Development System for Advanced Materials (ADAM) of RISH, Kyoto University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshimoto, Y., Suzuki, Y., Imai, T. et al. Supramolecular chiral emergence in water even after compensating for helix chirality in vesicular helix-peptide-aromatic frameworks. Polym J 55, 877–883 (2023). https://doi.org/10.1038/s41428-023-00781-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-023-00781-w