Abstract
Purpose
To analyse the gender-specific differences in central serous chorioretinopathy (CSCR) based on a new multimodal imaging classification system.
Method
This was a retrospective, multicentric, longitudinal, observational study in patients with a diagnosis of unilateral or bilateral CSCR. Visual acuity outcomes and differences based on ‘Simple’ and ‘Complex’ CSCR were analysed. The occurrence of choroidal neovascularization (CNVM) and number of recurrences were also compared. Regression analysis was used to evaluate baseline predictors of final visual acuity.
Results
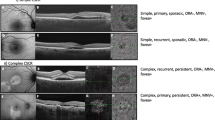
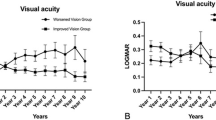
The study included 109 eyes of 58 patients (55 eyes of 28 female patients and 54 eyes of 30 male patients). Simple CSCR was seen in 8 (14.8%) eyes and 21 (38.2%) eyes in male and female groups respectively, while complex CSCR was seen in 46 (85.2%) eyes and 34 (61.8%) eyes in male and female groups respectively (p = 0.005). Recurrence was more commonly seen in males (34 eyes) than in females (23 eyes) (p = 0.03). Males (96.7%) were also significantly more likely to have a bilateral presentation (78.6% in females) (p = 0.03). Conversely, CNVM was more commonly seen in female eyes (8 eyes) than male eyes (4 eyes) (p = 0.23). On multivariable regression analysis, factors affecting reduced need for treatment were history of steroid use, good visual acuity at baseline, and simple CSCR. Factors affecting good final visual acuity were history of steroid use, good visual acuity at baseline, and younger age.
Conclusion
Males tended to have complex CSCR and recurrence compared to the female sub-group, while females exhibited CNVM more commonly than males.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, JC, upon reasonable request.
References
Von Graefe A. Ueber centrale recidivierende retinitis. Graefes Arch Clin Exp Ophthalmol. 1866;12:211–5.
Gilbert CM, Owens SL, Smith PD, Fine SL. Long-term follow-up of central serous chorioretinopathy. Br J Ophthalmol. 1984;68:815–20.
Sartini F, Figus M, Nardi M, Casini G, Posarelli C. Non-resolving, recurrent and chronic central serous chorioretinopathy: available treatment options. Eye. 2019;33:1035–43.
Hanumunthadu D, Tan AC, Singh SR, Sahu NK, Chhablani J. Management of chronic central serous chorioretinopathy. Indian J Ophthalmol. 2018;66:1704.
Sahoo NK, Lupidi M, Goud A, Gangakhedkar S, Cardillo Piccolino F, Chhablani J. One-year outcome of cystoid macular degeneration in central serous chorioretinopathy. Eur J Ophthalmol. 2022;32:2347–54.
Haimovici R, Koh S, Gagnon DR, Lehrfeld T, Wellik S, Group CSCCCS. Risk factors for central serous chorioretinopathy: a case–control study. Ophthalmology. 2004;111:244–9.
Chatziralli I, Kabanarou SA, Parikakis E, Chatzirallis A, Xirou T, Mitropoulos P. Risk factors for central serous chorioretinopathy: multivariate approach in a case-control study. Curr Eye Res. 2017;42:1069–73.
Matet A, Daruich A, Zola M, Behar-Cohen F. Risk factors for recurrences of central serous chorioretinopathy. Retina. 2018;38:1403–14.
Nicholson B, Noble J, Forooghian F, Meyerle C. Central serous chorioretinopathy: update on pathophysiology and treatment. Surv Ophthalmol. 2013;58:103–26.
Yoneyama S, Fukui A, Sakurada Y, Terao N, Shijo T, Kusada N, et al. Distinct characteristics of central serous chorioretinopathy according to gender. Sci Rep. 2022;12:1–8.
Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP. The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980–2002. Ophthalmology. 2008;115:169–73.
Ersoz MG, Arf S, Hocaoglu M, Muslubas IS, Karacorlu M. Patient characteristics and risk factors for central serous chorioretinopathy: an analysis of 811 patients. Br J Ophthalmol. 2019;103:725–9.
Todd KC, Hainsworth DP, Lee LR, Madsen RW. Longitudinal analysis of central serous chorioretinopathy and sex. Can J Ophthalmol. 2002;37:405–8.
Hanumunthadu D, Van Dijk EH, Gangakhedkar S, Goud A, Cheung CMG, Cherfan D, et al. Gender variation in central serous chorioretinopathy. Eye. 2018;32:1703–9.
van Dijk EH, Schellevis RL, van Bergen MG, Breukink MB, Altay L, Scholz P, et al. Association of a haplotype in the NR3C2 gene, encoding the mineralocorticoid receptor, with chronic central serous chorioretinopathy. JAMA Ophthalmol. 2017;135:446–51.
Baker ME, Katsu Y. Progesterone: an enigmatic ligand for the mineralocorticoid receptor. Biochem Pharm. 2020;177:113976.
Spaide RF, Cheung CMG, Matsumoto H, Kishi S, Boon CJ, van Dijk EH, et al. Venous overload choroidopathy: a hypothetical framework for central serous chorioretinopathy and allied disorders. Prog Retin Eye Res. 2021;86:100973.
Bacci T, Oh DJ, Singer M, Sadda S, Freund KB. Ultra-widefield indocyanine green angiography reveals patterns of choroidal venous insufficiency influencing pachychoroid disease. Invest Ophthalmol Vis Sci. 2022;63:17.
Imanaga N, Terao N, Nakamine S, Tamashiro T, Wakugawa S, Sawaguchi K, et al. Scleral thickness in central serous chorioretinopathy. Ophthalmol Retin. 2021;5:285–91.
Buckhurst HD, Gilmartin B, Cubbidge RP, Logan NS. Measurement of scleral thickness in humans using anterior segment optical coherent tomography. PloS One. 2015;10:e0132902.
Bousquet E, Torres-Villaros H, Provost J, Elalouf M, Gigon A, Mantel I, et al. Clinical characteristics and multimodal imaging findings of central serous chorioretinopathy in women versus men. J Clin Med. 2022;11:1706.
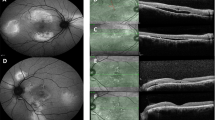
Chhablani J, Cohen FB, Aymard P, Beydoun T, Bousquet E, Daruich-Matet A, et al. Multimodal imaging-based central serous chorioretinopathy classification. Ophthalmol Retin. 2020;4:1043–6.
Sahoo NK, Parameshwarappa DC, Jhingan M, Tatti F, Iovino C, Peiretti E. Interobserver agreement of novel classification of central serous chorioretinopathy. Cureus. 2022;14:e25925.
Chhablani J, Behar-Cohen F. Validation of central serous chorioretinopathy multimodal imaging-based classification system. Graefes Arch Clin Exp Ophthalmol. 2022;260:1161–9.
Singh SR, Matet A, Van Dijk EH, Daruich A, Fauser S, Yzer S, et al. Discrepancy in current central serous chorioretinopathy classification. Br J Ophthalmol. 2019;103:737–42.
Arora S, Maltsev DS, Sahoo NK, Parameshwarappa DC, Iovino C, Arora T, et al. Visual acuity correlates with multimodal imaging-based categories of central serous chorioretinopathy. Eye. 2022;36:517–23.
Author information
Authors and Affiliations
Contributions
JC: design. JO, AS, AK, SH, RG: Conduct of the study. NKS, MA, RG, AK, SH: Collection. MA, ML, DZ, JC: Management. NKS, JO, AS, RB: Analysis. NKS, JO: Interpretation of the data. NKS, JO, RB: Preparation. RB, ML, DZ, JC: Review. ML, DZ, JC: approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sahoo, N.K., Ong, J., Selvam, A. et al. Gender differences in central serous chorioretinopathy based on the new multimodal imaging classification. Eye 38, 964–967 (2024). https://doi.org/10.1038/s41433-023-02812-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-023-02812-5