Abstract
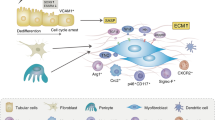
LncRNAs are reported to participate in the progression of various diseases including diabetic nephropathy. Currently, we reported that SNHG16 was obviously upregulated in db/db mice and high glucose-treated mice mesangial cells. Then, functional experiments showed that SNHG16 silencing significantly inhibited proliferation of mice mesangial cells, which induced the apoptosis and triggered cell cycle arrest. Meanwhile, proliferation-related biomarkers PCNA and Cyclin D1 (CCND1) were greatly repressed. Furthermore, western blot analysis was conducted to test fibrogenesis-associated genes Fibronectin and α-SMA. Meanwhile, the increased protein expression levels of Fibronectin and α-SMA under high glucose conditions were reversed by loss of SNHG16. miR-141-3p has been reported to be involved in various diseases. Then, RNA immunoprecipitation assay revealed the relation between SNHG16 and miR-141-3p. Downregulation of SNHG16 was able to induce expression of miR-141-3p, which was obviously reduced in db/db diabetic nephropathy mice. In addition, CCND1 is a crucial cell cycle master in human diseases. CCND1 was speculated as the target of miR-141-3p and miR-141-3p inhibited CCND1 expression significantly. Meanwhile, we observed that loss of CCND1 greatly repressed mice mesangial cell proliferation and induced cell apoptosis. Taken these together, we revealed for the first time that SNHG16 induced proliferation and fibrogenesis via modulating miR-141-3p and CCND1 in diabetic nephropathy. SNHG16/miR-141-3p/CCND1 axis can suggest a pathological mechanism of progression of diabetic nephropathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All relevant data are within the paper.
References
Tsai YC, Lee CS, Chiu YW, Lee JJ, Lee SC, Hsu YL, et al. Angiopoietin-2, renal deterioration, major adverse cardiovascular events and all-cause mortality in patients with diabetic nephropathy. Kidney Blood Press Res. 2018;43:545–54.
Oates PJ. Aldose reductase inhibitors and diabetic kidney disease. Curr Opin Investig Drugs. 2010;11:402–17.
Zhu L, Han J, Yuan R, Xue L, Pang W. Berberine ameliorates diabetic nephropathy by inhibiting TLR4/NF-kappaB pathway. Biol Res. 2018;51:9.
Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136:629–41.
Neumann P, Jae N, Knau A, Glaser SF, Fouani Y, Rossbach O, et al. The lncRNA GATA6-AS epigenetically regulates endothelial gene expression via interaction with LOXL2. Nat Commun. 2018;9:237.
Yan X, Zhang D, Wu W, Wu S, Qian J, Hao Y, et al. Mesenchymal stem cells promote hepatocarcinogenesis via lncRNA-MUF interaction with ANXA2 and miR-34a. Cancer Res. 2017;77:6704–16.
Zhang L, Yang Z, Trottier J, Barbier O, Wang L. Long noncoding RNA MEG3 induces cholestatic liver injury by interaction with PTBP1 to facilitate shp mRNA decay. Hepatology. 2017;65:604–15.
Roy S, Awasthi A. Emerging roles of noncoding RNAs in T cell differentiation and functions in autoimmune diseases. Int Rev Immunol. 2019;38:232–45.
Lopez-Urrutia E, Bustamante Montes LP, Ladron de Guevara Cervantes D, Perez-Plasencia C, Campos-Parra AD. Crosstalk between long non-coding RNAs, micro-RNAs and mRNAs: deciphering molecular mechanisms of master regulators in cancer. Front Oncol. 2019;9:669.
Zhao J, Li L, Han ZY, Wang ZX, Qin LX. Long noncoding RNAs, emerging and versatile regulators of tumor-induced angiogenesis. Am J Cancer Res. 2019;9:1367–81.
Ignarski M, Islam R, Muller RU. Long non-coding RNAs in kidney disease. Int J Mol Sci. 2019;20:3276.
Li Y, Xu K, Xu K, Chen S, Cao Y, Zhan H. Roles of identified long noncoding RNA in diabetic nephropathy. J Diabetes Res. 2019;2019:5383010.
Zhang J, Jiang T, Liang X, Shu S, Xiang X, Zhang W, et al. lncRNA MALAT1 mediated high glucose-induced HK-2 cell epithelial-to-mesenchymal transition and injury. J Physiol Biochem. 2019;75:443–52.
Liu DW, Zhang JH, Liu FX, Wang XT, Pan SK, Jiang DK, et al. Silencing of long noncoding RNA PVT1 inhibits podocyte damage and apoptosis in diabetic nephropathy by upregulating FOXA1. Exp Mol Med. 2019;51:88.
Zhang Y, Chang B, Zhang J, Wu X. LncRNA SOX2OT alleviates the high glucose-induced podocytes injury through autophagy induction by the miR-9/SIRT1 axis. Exp Mol Pathol. 2019;110:104283.
Liu H, Sun HL. LncRNA TCF7 triggered endoplasmic reticulum stress through a sponge action with miR-200c in patients with diabetic nephropathy. Eur Rev Med pharmacol Sci. 2019;23:5912–22.
Xie X, Xu X, Sun C, Yu Z. Long intergenic noncoding RNA SNHG16 interacts with miR-195 to promote proliferation, invasion and tumorigenesis in hepatocellular carcinoma. Exp Cell Res. 2019;383:111501.
Xu C, Hu C, Wang Y, Liu S. Long noncoding RNA SNHG16 promotes human retinoblastoma progression via sponging miR-140-5p. Biomed Pharmacother. 2019;117:109153.
Zhou XY, Liu H, Ding ZB, Xi HP, Wang GW. lncRNA SNHG16 promotes glioma tumorigenicity through miR-373/EGFR axis by activating PI3K/AKT pathway. Genomics. 2020;112:1021–9.
Leti F, Morrison E, DiStefano JK. Long noncoding RNAs in the pathogenesis of diabetic kidney disease: implications for novel therapeutic strategies. Personal Med. 2017;14:271–8.
Alvarez ML, Distefano JK. The role of non-coding RNAs in diabetic nephropathy: potential applications as biomarkers for disease development and progression. Diabetes Res Clin Pract. 2013;99:1–11.
Christensen LL, True K, Hamilton MP, Nielsen MM, Damas ND, Damgaard CK, et al. SNHG16 is regulated by the Wnt pathway in colorectal cancer and affects genes involved in lipid metabolism. Mol Oncol. 2016;10:1266–82.
Cai C, Huo Q, Wang X, Chen B, Yang Q. SNHG16 contributes to breast cancer cell migration by competitively binding miR-98 with E2F5. Biochem Biophys Res Commun. 2017;485:272–8.
Yu Y, Chen F, Yang Y, Jin Y, Shi J, Han S, et al. lncRNA SNHG16 is associated with proliferation and poor prognosis of pediatric neuroblastoma. Int J Oncol. 2019;55:93–102.
Guo G, Kang Q, Zhu X, Chen Q, Wang X, Chen Y, et al. A long noncoding RNA critically regulates Bcr-Abl-mediated cellular transformation by acting as a competitive endogenous RNA. Oncogene. 2015;34:1768–79.
Liang Z, Li X, Liu S, Li C, Wang X, Xing J. MiR-141-3p inhibits cell proliferation, migration and invasion by targeting TRAF5 in colorectal cancer. Biochem Biophys Res Commun. 2019;514:699–705.
Li JH, Zhang Z, Du MZ, Guan YC, Yao JN, Yu HY, et al. microRNA-141-3p fosters the growth, invasion, and tumorigenesis of cervical cancer cells by targeting FOXA2. Arch Biochem Biophys. 2018;657:23–30.
Zhang MN, Tang QY, Li RM, Song MG. MicroRNA-141-3p/200a-3p target and may be involved in post-transcriptional repression of RNA decapping enzyme Dcp2 during renal development. Biosci Biotechnol Biochem. 2018;82:1724–32.
Wang W, Medeiros LJ. Utility of cyclin D1 in the diagnostic workup of hematopoietic neoplasms: what can cyclin D1 do for us? Ad Anatomic Pathol. 2019;26:281–91.
Ramos-Garcia P, Gil-Montoya JA, Scully C, Ayen A, Gonzalez-Ruiz L, Navarro-Trivino FJ, et al. An update on the implications of cyclin D1 in oral carcinogenesis. Oral Dis. 2017;23:897–912.
Neumeister P, Pixley FJ, Xiong Y, Xie H, Wu K, Ashton A, et al. Cyclin D1 governs adhesion and motility of macrophages. Mol Biol Cell. 2003;14:2005–15.
Zhong Z, Yeow WS, Zou C, Wassell R, Wang C, Pestell RG, et al. Cyclin D1/cyclin-dependent kinase 4 interacts with filamin A and affects the migration and invasion potential of breast cancer cells. Cancer Res. 2010;70:2105–14.
Zhou Q, Zhang W, Wang Z, Liu S. Long non-coding RNA PTTG3P functions as an oncogene by sponging miR-383 and up-regulating CCND1 and PARP2 in hepatocellular carcinoma. BMC Cancer. 2019;19:731.
Wang M, Yu W, Gao J, Ma W, Frentsch M, Thiel A, et al. MicroRNA-487a-3p functions as a new tumor suppressor in prostate cancer by targeting CCND1. J Cell Physiol. 2020;235:1588–600.
Funding
This work was supported by the Key Program of Science and Technology Committee of Changning District, Shanghai, China, [Grant No. WKW2016 Z03].
Author information
Authors and Affiliations
Contributions
SH, XJ, PL, and QR designed the study and the experiments. XJ, PL, and QR performed the experiments. XG and KS collected the data. LX, BX, and QW did the analysis. XJ drafted the manuscript. SH revised the manuscript. All of the authors finally approved the proof for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, X., Ru, Q., Li, P. et al. LncRNA SNHG16 induces proliferation and fibrogenesis via modulating miR-141-3p and CCND1 in diabetic nephropathy. Gene Ther 27, 557–566 (2020). https://doi.org/10.1038/s41434-020-0160-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-020-0160-x
This article is cited by
-
Silencing LncRNA SNHG16 suppresses the diabetic inflammatory response by targeting the miR-212-3p/NF-κB signaling pathway
Diabetology & Metabolic Syndrome (2023)
-
LncRNA SNHG16 regulates RAS and NF-κB pathway-mediated NLRP3 inflammasome activation to aggravate diabetes nephropathy through stabilizing TLR4
Acta Diabetologica (2023)