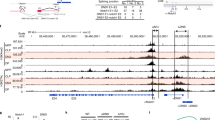
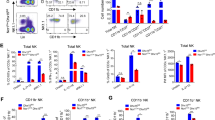
Abstract
Ikaros transcription factors are essential for adaptive lymphocyte function, yet their role in innate lymphopoiesis is unknown. Using conditional genetic inactivation, we show that Ikzf1/Ikaros is essential for normal natural killer (NK) cell lymphopoiesis and IKZF1 directly represses Cish, a negative regulator of interleukin-15 receptor resulting in impaired interleukin-15 receptor signaling. Both Bcl2l11 and BIM levels, and intrinsic apoptosis were increased in Ikzf1-null NK cells, which in part accounts for NK lymphopenia as both were restored to normal levels when Ikzf1 and Bcl2l11 were co-deleted. Ikzf1-null NK cells presented extensive transcriptional alterations with reduced AP-1 transcriptional complex expression and increased expression of Ikzf2/Helios and Ikzf3/Aiolos. IKZF1 and IKZF3 directly bound AP-1 family members and deletion of both Ikzf1 and Ikzf3 in NK cells resulted in further reductions in Jun/Fos expression and complete loss of peripheral NK cells. Collectively, we show that Ikaros family members are important regulators of apoptosis, cytokine responsiveness and AP-1 transcriptional activity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Spits, H. et al. Innate lymphoid cells—a proposal for uniform nomenclature. Nat. Rev. Immunol. 13, 145–149 (2013).
Cerwenka, A. & Lanier, L. L. Natural killer cells, viruses and cancer. Nat. Rev. Immunol. 1, 41–49 (2001).
Huntington, N. D., Cursons, J. & Rautela, J. The cancer–natural killer cell immunity cycle. Nat. Rev. Cancer 20, 437–454 (2020).
Boos, M. D., Yokota, Y., Eberl, G. & Kee, B. L. Mature natural killer cell and lymphoid tissue–inducing cell development requires Id2-mediated suppression of E protein activity. J. Exp. Med. 204, 1119–1130 (2007).
Delconte, R. B. et al. The helix-loop-helix protein ID2 governs NK cell fate by tuning their sensitivity to interleukin-15. Immunity 44, 103–115 (2016).
Gascoyne, D. M. et al. The basic leucine zipper transcription factor E4BP4 is essential for natural killer cell development. Nat. Immunol. 10, 1118–1124 (2009).
Kamizono, S. et al. Nfil3/E4bp4 is required for the development and maturation of NK cells in vivo. J. Exp. Med. 206, 2977–2986 (2009).
Goh, W. et al. Hhex directly represses BIM-dependent apoptosis to promote NK cell development and maintenance. Cell Rep. 33, 108285 (2020).
Viant, C. et al. Cell cycle progression dictates the requirement for BCL2 in natural killer cell survival. J. Exp. Med. 214, 491–510 (2017).
Huntington, N. D. et al. Interleukin 15-mediated survival of natural killer cells is determined by interactions among Bim, Noxa and Mcl-1. Nat. Immunol. 8, 856–863 (2007).
Sathe, P. et al. Innate immunodeficiency following genetic ablation of Mcl1 in natural killer cells. Nat. Commun. 5, 4539 (2014).
Huntington, N. D. The unconventional expression of IL-15 and its role in NK cell homeostasis. Immunol. Cell Biol. 92, 210–213 (2014).
Robbins, S. H. et al. Cutting edge: inhibitory functions of the killer cell lectin-like receptor G1 molecule during the activation of mouse NK cells. J. Immunol. 168, 2585–2589 (2002).
Delconte, R. B. et al. CIS is a potent checkpoint in NK cell-mediated tumor immunity. Nat. Immunol. 17, 816–824 (2016).
Hayashi, T. et al. Molecular mechanisms whereby immunomodulatory drugs activate natural killer cells: clinical application. Br. J. Haematol. 128, 192–203 (2005).
Krönke, J. et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science 343, 301–305 (2014).
Lu, G. et al. The myeloma drug lenalidomide promotes the cereblon-dependent destruction of Ikaros proteins. Science 343, 305–309 (2014).
Georgopoulos, K., Winandy, S. & Avitahl, N. The role of the Ikaros gene in lymphocyte development and homeostasis. Annu. Rev. Immunol. 15, 155–176 (1997).
Schjerven, H. et al. Selective regulation of lymphopoiesis and leukemogenesis by individual zinc fingers of Ikaros. Nat. Immunol. 14, 1073–1083 (2013).
Georgopoulos, K., Moore, D. D. & Derfler, B. Ikaros, an early lymphoid-specific transcription factor and a putative mediator for T cell commitment. Science 258, 808–812 (1992).
Georgopoulos, K. et al. The Ikaros gene is required for the development of all lymphoid lineages. Cell 79, 143–156 (1994).
Yoshida, T., Ng, S. Y. -M., Zuniga-Pflucker, J. C. & Georgopoulos, K. Early hematopoietic lineage restrictions directed by Ikaros. Nat. Immunol. 7, 382–391 (2006).
Papathanasiou, P. et al. Self-renewal of the long-term reconstituting subset of hematopoietic stem cells is regulated by Ikaros. Stem Cells 27, 3082–3092 (2009).
Schwickert, T. A. et al. Stage-specific control of early B cell development by the transcription factor Ikaros. Nat. Immunol. 15, 283–293 (2014).
Ma, S. et al. Ikaros and Aiolos inhibit pre-B-cell proliferation by directly suppressing c-Myc expression. Mol. Cell. Biol. 30, 4149–4158 (2010).
Avitahl, N. et al. Ikaros sets thresholds for T cell activation and regulates chromosome propagation. Immunity 10, 333–343 (1999).
Kim, H. -J. et al. Stable inhibitory activity of regulatory T cells requires the transcription factor Helios. Science 350, 334–339 (2015).
Holmes, M. L. et al. Peripheral natural killer cell maturation depends on the transcription factor Aiolos. EMBO J. 33, 2721–2734 (2014).
Sun, L., Liu, A. & Georgopoulos, K. Zinc finger-mediated protein interactions modulate Ikaros activity, a molecular control of lymphocyte development. EMBO J. 15, 5358–5369 (1996).
Rautela, J. & Huntington, N. D. IL-15 signaling in NK cell cancer immunotherapy. Curr. Opin. Immunol. 44, 1–6 (2017).
Adams, N. M. et al. Transcription factor IRF8 orchestrates the adaptive natural killer cell response. Immunity 48, 1172–1182 (2018).
Beaulieu, A. M., Zawislak, C. L., Nakayama, T. & Sun, J. C. The transcription factor Zbtb32 controls the proliferative burst of virus-specific natural killer cells responding to infection. Nat. Immunol. 15, 546–553 (2014).
Iacobucci, I. et al. IKAROS deletions dictate a unique gene expression signature in patients with adult B-cell acute lymphoblastic leukemia. PLoS ONE 7, e40934 (2012).
Javed, A. et al. Ikaros family proteins redundantly regulate temporal patterning in the developing mouse retina. Development 150, dev200436 (2023).
Kim, W. S. et al. Suppressor of cytokine signaling 2 negatively regulates NK cell differentiation by inhibiting JAK2 activity. Sci. Rep. 7, 46153 (2017).
Delconte, R. B. et al. NK cell priming from endogenous homeostatic signals is modulated by CIS. Front. Immunol. 11, 75 (2020).
Bernard, P. -L. et al. Targeting CISH enhances natural cytotoxicity receptor signaling and reduces NK cell exhaustion to improve solid tumor immunity. J. Immunother. Cancer 10, e004244 (2022).
Putz, E. M. et al. Targeting cytokine signaling checkpoint CIS activates NK cells to protect from tumor initiation and metastasis. Oncoimmunology 6, e1267892 (2017).
Karin, M., Liu, Z. G. & Zandi, E. AP-1 function and regulation. Curr. Opin. Cell Biol. 9, 240–246 (1997).
Schnoegl, D., Hiesinger, A., Huntington, N. D. & Gotthardt, D. AP-1 transcription factors in cytotoxic lymphocyte development and antitumor immunity. Curr. Opin. Immunol. 85, 102397 (2023).
Delpoux, A. et al. FOXO1 constrains activation and regulates senescence in CD8 T cells. Cell Rep. 34, 108674 (2021).
Passegué, E., Jochum, W., Behrens, A., Ricci, R. & Wagner, E. F. JunB can substitute for Jun in mouse development and cell proliferation. Nat. Genet. 30, 158–166 (2002).
Hildner, K. et al. Batf3 deficiency reveals a critical role for CD8α+ dendritic cells in cytotoxic T cell immunity. Science 322, 1097–1100 (2008).
Riera-Sans, L. & Behrens, A. Regulation of αβ/γδ T cell development by the activator protein 1 transcription factor c-Jun. J. Immunol. 178, 5690–5700 (2007).
Li, P. et al. BATF-JUN is critical for IRF4-mediated transcription in T cells. Nature 490, 543–546 (2012).
Ellin, F., Landström, J., Jerkeman, M. & Relander, T. Real-world data on prognostic factors and treatment in peripheral T-cell lymphomas: a study from the Swedish Lymphoma Registry. Blood 124, 1570–1577 (2014).
Fedele, P. L. et al. Loss of IRF4 results in multiple myeloma cell apoptosis through the transcriptional upregulation of the BH3-only proteins Bmf and BIM. Blood 134, 3103 (2019).
Fedele, P. L. et al. The transcription factor IRF4 represses proapoptotic BMF and BIM to licence multiple myeloma survival. Leukemia 35, 2114–2118 (2021).
Fehniger, T. A. et al. Fatal leukemia in interleukin 15 transgenic mice follows early expansions in natural killer and memory phenotype CD8+ T cells. J. Exp. Med. 193, 219–231 (2001).
Narni-Mancinelli, E. et al. Fate mapping analysis of lymphoid cells expressing the NKp46 cell surface receptor. Proc. Natl Acad. Sci. USA 108, 18324–18329 (2011).
Rautela, J., Surgenor, E. & Huntington, N. D. Drug target validation in primary human natural killer cells using CRISPR RNP. J. Leukoc. Biol. 108, 1397–1408 (2020).
Schuster, I. S. et al. TRAIL+ NK cells control CD4+ T cell responses during chronic viral infection to limit autoimmunity. Immunity 41, 646–656 (2014).
Liao, Y., Smyth, G. K. & Shi, W. The Subread aligner: fast, accurate and scalable read mapping by seed-and-vote. Nucleic Acids Res. 41, e108 (2013).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Zhang, Y. et al. Model-based analysis of ChIP–seq (MACS). Genome Biol. 9, R137 (2008).
Lun, A. T. L. & Smyth, G. K. csaw: a Bioconductor package for differential binding analysis of ChIP-seq data using sliding windows. Nucleic Acids Res. 44, e45 (2016).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Liang, S. et al. A small molecule inhibitor of PTP1B and PTPN2 enhances T cell anti-tumor immunity. Nat. Commun. 14, 4524 (2023).
Acknowledgements
We thank the N.D.H. laboratory members A. Doan and T. Camilleri for technical assistance and animal husbandry. We extend our gratitude to Monash University platform staff and administration for their services. We are extremely grateful to M. Busslinger (IMP, Austria) for providing the Ikzf1loxP mice, E. Vivier (CIML, France) for providing the Ncr1iCre mice, M. Caligiuri (City of Hope, USA) for providing the Il15Tg and F. Ishikawa (RIKEN, Japan) for providing the human IL-15/IL-7 transgenic NSG mice and D. Lee (Nationwide Children’s Hospital, USA) for the K562-CSTX002 cell line. W.G. was supported by a Melbourne International Research Scholarship. M.F. was supported by an Investigator Fellowship from the Prostate Cancer Foundation (USA). This work is supported by project grants from the NHMRC of Australia (GNT1124784, GNT1066770, GNT1057852, GNT1124907, GNT1057812, GNT1049407, GNT1027472 and GNT1184615 to N.D.H.) and an NHMRC Investigator Fellowship (GNT1195296 to N.D.H.). N.D.H. is a recipient of a Melanoma Research Grant from the Harry J. Lloyd Charitable Trust, Melanoma Research Alliance Young Investigator Award, an Ian Potter Foundation equipment grant, the National Foundation for Medical Research and Innovation (NFMRI) John Dixon Hughes Medal, and a CLIP grant from the Cancer Research Institute. S.L.N. is an NHMRC SPRF Fellow (GNT1155342). M.A.D.-E. is an NHMRC PRF Fellow (GNT1119298). J.R. was supported by a Victorian Cancer Agency grant (ECSG18020).
Author information
Authors and Affiliations
Contributions
W.G., S.S., H.S., Z.S., A.P., R.H., I.K., R.B.D., C.E.A., I.S.S., P.B., D.T., I.P., M.C., X.M., F.S.-F.-G and J.R. designed and performed experiments. M.F., S.H.-Z. and S.S. provided bioinformatics analysis and input into interpretation of results. N.D.H., S.L.N., M.J.D., J.C., J.R., A.K. and M.A.D.-E. supervised experimental design and provided input into interpretation of results. S.L.N., A.K. and N.D.H. wrote the manuscript with editorial input from all authors.
Corresponding author
Ethics declarations
Competing interests
N.D.H. and J.R. are founders and shareholders in oNKo-Innate. N.D.H. is an inventor on patents relating to this work. N.D.H. serves on an advisory board for Bristol Myers Squibb. The other authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: N. Bernard, in collaboration with the rest of the Nature Immunology team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Goh, W., Sudholz, H., Foroutan, M. et al. IKAROS and AIOLOS directly regulate AP-1 transcriptional complexes and are essential for NK cell development. Nat Immunol 25, 240–255 (2024). https://doi.org/10.1038/s41590-023-01718-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-023-01718-4