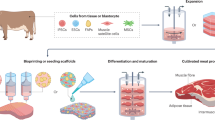
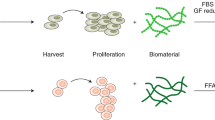
Abstract
Cultivated meat scale-up and industrial production will require multiple stable cell lines from different species to recreate the organoleptic and nutritional properties of meat from livestock. In this Review, we explore the potential of stem cells to create the major cellular components of cultivated meat. By using developments in the fields of tissue engineering and biomedicine, we explore the advantages and disadvantages of strategies involving primary adult and pluripotent stem cells for generating cell sources that can be grown at scale. These myogenic, adipogenic or extracellular matrix-producing adult stem cells as well as embryonic or inducible pluripotent stem cells are discussed for their proliferative and differentiation capacity, necessary for cultivated meat. We examine the challenges for industrial scale-up, including differentiation and culture protocols, as well as genetic modification options for stem cell immortalization and controlled differentiation. Finally, we discuss stem cell-related safety and regulatory challenges for bringing cultivated meat to the marketplace.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Li, X., Fu, X., Yang, G. & Du, M. Enhancing intramuscular fat development via targeting fibro-adipogenic progenitor cells in meat animals. Animal 14, 312–321 (2020).
Weston, A. R., Rogers, R. W. & Althen, T. G. The role of collagen in meat tenderness. Prof. Anim. Sci. 18, 107–111 (2002).
Le Grand, F. & Rudnicki, M. A. Skeletal muscle satellite cells and adult myogenesis. Curr. Opin. Cell Biol. 19, 628–633 (2007).
Liu, G. & Chen, X. Isolating and characterizing adipose-derived stem cells. Methods Mol. Biol. 1842, 193–201 (2018).
Reggio, A. et al. Adipogenesis of skeletal muscle fibro/adipogenic progenitors is affected by the WNT5a/GSK3/β-catenin axis. Cell Death Differ. 27, 2921–2941 (2020).
Bogliotti, Y. S. et al. Efficient derivation of stable primed pluripotent embryonic stem cells from bovine blastocysts. Proc. Natl Acad. Sci. USA 115, 2090–2095 (2018).
Hayflick, L. & Moorhead, P. S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 25, 585–621 (1961).
Lee, S. H., Joo, S. T. & Ryu, Y. C. Skeletal muscle fiber type and myofibrillar proteins in relation to meat quality. Meat Sci. 86, 166–170 (2010).
Listrat, A. et al. How muscle structure and composition influence meat and flesh quality. Sci. World J. 2016, 3182746 (2016).
Fraeye, I., Kratka, M., Vandenburgh, H. & Thorrez, L. Sensorial and nutritional aspects of cultured meat in comparison to traditional meat: much to be inferred. Front. Nutr. 7, 35 (2020).
Will, K., Schering, L., Albrecht, E., Kalbe, C. & Maak, S. Differentiation of bovine satellite cell-derived myoblasts under different culture conditions. In Vitro Cell. Dev. Biol. Anim. 51, 885–889 (2015).
Li, B. J. et al. Isolation, culture and identification of porcine skeletal muscle satellite cells. Asian Australas. J. Anim. Sci. 28, 1171–1177 (2015).
Yablonka-Reuveni, Z., Quinn, L. B. S. & Nameroff, M. Isolation and clonal analysis of satellite cells from chicken pectoralis muscle. Dev. Biol. 119, 252–259 (1987).
Suárez-Calvet, X. et al. Isolation of human fibroadipogenic progenitors and satellite cells from frozen muscle biopsies. FASEB J. 35, e21819 (2021).
Melzener, L. et al. Comparative analysis of cattle breeds as satellite cell donors for cultured beef. Preprint at bioXriv https://doi.org/10.1101/2022.01.14.476358 (2022).
Ding, S. et al. Characterization and isolation of highly purified porcine satellite cells. Cell Death Discov. 3, 17003 (2017).
Campion, D. R., Richardson, R. L., Reagan, J. O. & Kraeling, R. R. Changes in the satellite cell population during postnatal growth of pig skeletal muscle. J. Anim. Sci. 52, 1014–1018 (1981).
Mulvaney, D. R., Marple, D. N. & Merkel, R. A. Proliferation of skeletal muscle satellite cells after castration and administration of testosterone propionate. Proc. Soc. Exp. Biol. Med. 188, 40–45 (1988).
Fish, K. D., Rubio, N. R., Stout, A. J., Yuen, J. S. K. & Kaplan, D. L. Prospects and challenges for cell-cultured fat as a novel food ingredient. Trends Food Sci. Technol. 98, 53–67 (2020).
Ben-Arye, T. & Levenberg, S. Tissue engineering for clean meat production. Front. Sustain. Food Syst. 3, 46 (2019).
Wood, J. D. et al. Effects of fatty acids on meat quality: a review. Meat Sci. 66, 21–32 (2004).
van Vliet, S. et al. A metabolomics comparison of plant-based meat and grass-fed meat indicates large nutritional differences despite comparable Nutrition Facts panels. Sci. Rep. 11, 13828 (2021).
Dohmen, R. G. J. et al. Muscle-derived fibro-adipogenic progenitor cells for production of cultured bovine adipose tissue. npj Sci. Food 6, 6 (2022).
Chen, Q. et al. Fate decision of mesenchymal stem cells: adipocytes or osteoblasts? Cell Death Differ. 23, 1128–1139 (2016).
Bosnakovski, D. et al. Isolation and multilineage differentiation of bovine bone marrow mesenchymal stem cells. Cell Tissue Res. 319, 243–253 (2005).
Cawthorn, W. P., Scheller, E. L. & MacDougald, O. A. Adipose tissue stem cells meet preadipocyte commitment: going back to the future. J. Lipid Res. 53, 227–246 (2012).
Gupta, R. K. et al. Transcriptional control of preadipocyte determination by Zfp423. Nature 464, 619–623 (2010).
Huang, Y., Das, A. K., Yang, Q. Y., Zhu, M. J. & Du, M. Zfp423 promotes adipogenic differentiation of bovine stromal vascular cells. PLoS ONE 7, e47496 (2012).
Rosen, E. D. et al. PPARγ is required for the differentiation of adipose tissue in vivo and in vitro. Mol. Cell 4, 611–617 (1999).
Lefterova, M. I. & Lazar, M. A. New developments in adipogenesis. Trends Endocrinol. Metab. 20, 107–114 (2009).
Yamada, T., Kawakami, S.-I. & Nakanishi, N. Expression of adipogenic transcription factors in adipose tissue of fattening Wagyu and Holstein steers. Meat Sci. 81, 86–92 (2009).
Sztalryd, C. & Brasaemle, D. L. The perilipin family of lipid droplet proteins: gatekeepers of intracellular lipolysis. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1862, 1221–1232 (2017).
Kraus, N. A. et al. Quantitative assessment of adipocyte differentiation in cell culture. Adipocyte 5, 351–358 (2016).
Zhang, J. et al. Adipose-derived stem cells: current applications and future directions in the regeneration of multiple tissues. Stem Cells Int. 2020, 8810813 (2020).
Chen, Y. J. et al. Isolation and differentiation of adipose-derived stem cells from porcine subcutaneous adipose tissues. J. Vis. Exp. 2016, e53886 (2016).
Wei, S. et al. Bovine mature adipocytes readily return to a proliferative state. Tissue Cell 44, 385–390 (2012).
Matsumoto, T. et al. Mature adipocyte-derived dedifferentiated fat cells exhibit multilineage potential. J. Cell. Physiol. 215, 210–222 (2008).
Oki, Y., Hagiwara, R., Matsumaru, T. & Kano, K. Effect of volatile fatty acids on adipocyte differentiation in bovine dedifferentiated fat (DFAT) cells in vitro. Genes Cells 27, 5–13 (2022).
Spadaro, V., Allen, D. H., Keeton, J. T., Moreira, R. & Boleman, R. M. Biomechanical properties of meat and their correlation to tenderness. J. Texture Stud. 33, 59–87 (2002).
Mackey, A. L., Magnan, M., Chazaud, B. & Kjaer, M. Human skeletal muscle fibroblasts stimulate in vitro myogenesis and in vivo muscle regeneration. J. Physiol. 595, 5115–5127 (2017).
Contreras, O., Rossi, F. M. & Brandan, E. Adherent muscle connective tissue fibroblasts are phenotypically and biochemically equivalent to stromal fibro/adipogenic progenitors. Matrix Biol. Plus 2, 100006 (2019).
Soundararajan, M. & Kannan, S. Fibroblasts and mesenchymal stem cells: two sides of the same coin? J. Cell. Physiol. 233, 9099–9109 (2018).
Green, B. B. & Kerr, D. E. Epigenetic contribution to individual variation in response to lipopolysaccharide in bovine dermal fibroblasts. Vet. Immunol. Immunopathol. 157, 49–58 (2014).
Melzener, L., Verzijden, K. E., Buijs, A. J., Post, M. J. & Flack, J. E. Cultured beef: from small biopsy to substantial quantity. J. Sci. Food Agric. 101, 7–14 (2021).
Ding, S. et al. Maintaining bovine satellite cells stemness through p38 pathway. Sci. Rep. 8, 10808 (2018).
Westerman, K. A. Myospheres are composed of two cell types: one that is myogenic and a second that is mesenchymal. PLoS ONE 10, e0116956 (2015).
Kolkmann, A. M., Van Essen, A., Post, M. J. & Moutsatsou, P. Development of a chemically defined medium for in vitro expansion of primary bovine satellite cells. Front. Bioeng. Biotechnol. 10, 895289 (2022).
Steakholder Foods. Steakholder Foods https://steakholderfoods.com/ (2022).
Rubio, N., Datar, I., Stachura, D., Kaplan, D. & Krueger, K. Cell-based fish: a novel approach to seafood production and an opportunity for cellular agriculture. Front. Sustain. Food Syst. 3, 43 (2019).
Benson, C. A. & Madden, L. R. Cell-cultured food products and related cells, compositions, methods and systems. US patent WO2022221261A2 (2022).
Our approach—full of flavor, free of harm. Mission Barns https://missionbarns.com/process/ (2022).
A new source of seafood. Wildtype https://www.wildtypefoods.com/about-us (2023).
About our innovation. Shiok Meats https://shiokmeats.com/about-our-innovation/ (2023)
New Zealand’s first cultivated meat company emerges from stealth mode. Protein Report https://www.proteinreport.org/newswire/new-zealands-first-cultivated-meat-company-emerges-stealth-mode (2022).
Evans, M. J. & Kaufman, M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981).
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Kinoshita, M. et al. Pluripotent stem cells related to embryonic disc exhibit common self-renewal requirements in diverse livestock species. Development 148, dev199901 (2021).
Soto, D. A. et al. Simplification of culture conditions and feeder-free expansion of bovine embryonic stem cells. Sci. Rep. 11, 11045 (2021).
Choi, K.-H. et al. Pluripotent pig embryonic stem cell lines originating from in vitro-fertilized and parthenogenetic embryos. Stem Cell Res. 49, 102093 (2020).
Vilarino, M. et al. Derivation of sheep embryonic stem cells under optimized conditions. Reproduction 160, 761–772 (2020).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Wu, S. M. & Hochedlinger, K. Harnessing the potential of induced pluripotent stem cells for regenerative medicine. Nat. Cell Biol. 13, 497–505 (2011).
Pillai, V. V. et al. Efficient induction and sustenance of pluripotent stem cells from bovine somatic cells. Biol. Open 10, bio058756 (2021).
Ezashi, T. et al. Derivation of induced pluripotent stem cells from pig somatic cells. Proc. Natl Acad. Sci. USA 106, 10993–10998 (2009).
Tesar, P. J. et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448, 196–199 (2007).
Wu, J. et al. An alternative pluripotent state confers interspecies chimaeric competency. Nature 521, 316–321 (2015).
Gao, X. et al. Establishment of porcine and human expanded potential stem cells. Nat. Cell Biol. 21, 687–699 (2019).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Huangfu, D. et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nat. Biotechnol. 26, 795–797 (2008).
Brambrink, T. et al. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell 2, 151–159 (2008).
Stadtfeld, M., Nagaya, M., Utikal, J., Weir, G. & Hochedlinger, K. Induced pluripotent stem cells generated without viral integration. Science 322, 945–949 (2008).
Okita, K., Nakagawa, M., Hyenjong, H., Ichisaka, T. & Yamanaka, S. Generation of mouse induced pluripotent stem cells without viral vectors. Science 322, 949–953 (2008).
Woltjen, K. et al. piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature 458, 766–770 (2009).
Kim, D. et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4, 472–476 (2009).
Sokka, J. et al. CRISPR activation enables high-fidelity reprogramming into human pluripotent stem cells. Stem Cell Rep. 17, 413–426 (2022).
Bressan, F. F. et al. Generation of induced pluripotent stem cells from large domestic animals. Stem Cell Res. Ther. 11, 247 (2020).
Kawaguchi, T. et al. Generation of naïve bovine induced pluripotent stem cells using piggyBac transposition of doxycycline-inducible transcription factors. PLoS ONE 10, e0135403 (2015).
Yang, X. et al. Nuclear reprogramming of cloned embryos and its implications for therapeutic cloning. Nat. Genet. 39, 295–302 (2007).
Li, D. et al. Generation of transgene-free porcine intermediate type induced pluripotent stem cells. Cell Cycle 17, 2547–2563 (2018).
Liu, K. et al. Generation of porcine-induced pluripotent stem cells by using OCT4 and KLF4 porcine factors. Cell. Reprogram. 14, 505–513 (2012).
Esteban, M. A. et al. Porcine induced pluripotent stem cells may bridge the gap between mouse and human iPS. IUBMB Life https://doi.org/10.1002/iub.307 (2010).
Zhang, W. et al. Pluripotent and metabolic features of two types of porcine iPSCs derived from defined mouse and human ES cell culture conditions. PLoS ONE 10, e0124562 (2015).
Marei, H. E., Althani, A., Lashen, S., Cenciarelli, C. & Hasan, A. Genetically unmatched human iPSC and ESC exhibit equivalent gene expression and neuronal differentiation potential. Sci. Rep. 7, 17504 (2017).
Hussein, S. M. et al. Copy number variation and selection during reprogramming to pluripotency. Nature 471, 58–62 (2011).
Mayshar, Y. et al. Identification and classification of chromosomal aberrations in human induced pluripotent stem cells. Cell Stem Cell 7, 521–531 (2010).
Kim, K. et al. Epigenetic memory in induced pluripotent stem cells. Nature 467, 285–290 (2010).
Chal, J. et al. Generation of human muscle fibers and satellite-like cells from human pluripotent stem cells in vitro. Nat. Protoc. 11, 1833–1850 (2016).
Shelton, M. et al. Derivation and expansion of PAX7-positive muscle progenitors from human and mouse embryonic stem cells. Stem Cell Rep. 3, 516–529 (2014).
Genovese, N. J., Domeier, T. L., Telugu, B. P. V. L. & Roberts, R. M. Enhanced development of skeletal myotubes from porcine induced pluripotent stem cells. Sci. Rep. 7, 41833 (2017).
Wittler, L. et al. Expression of Msgn1 in the presomitic mesoderm is controlled by synergism of WNT signalling and Tbx6. EMBO Rep. 8, 784–789 (2007).
Takada, S. et al. Wnt-3a regulates somite and tailbud formation in the mouse embryo. Genes Dev. 8, 174–189 (1994).
Miyagoe-Suzuki, Y. & Takeda, S. Skeletal muscle generated from induced pluripotent stem cells—induction and application. World J. Stem Cells 9, 89–97 (2017).
Brand-Saberi, B., Müller, T. S., Wilting, J., Christ, B. & Birchmeier, C. Scatter factor/hepatocyte growth factor (SF/HGF) induces emigration of myogenic cells at interlimb levelin vivo. Dev. Biol. 179, 303–308 (1996).
Laron, Z. Somatomedin-1 (recombinant insulin-like growth factor-1): clinical pharmacology and potential treatment of endocrine and metabolic disorders. BioDrugs 11, 55–70 (1999).
Tahimic, C. G. T., Wang, Y. & Bikle, D. D. Anabolic effects of IGF-1 signaling on the skeleton. Front. Endocrinol. 4, 6 (2013).
van der Velden, J. L. J. et al. Inhibition of glycogen synthase kinase-3β activity is sufficient to stimulate myogenic differentiation. Am. J. Physiol. Cell Physiol. 290, C453–C462 (2006).
Barberi, T. et al. Derivation of engraftable skeletal myoblasts from human embryonic stem cells. Nat. Med. 13, 642–648 (2007).
Chang, H. et al. Generation of transplantable, functional satellite-like cells from mouse embryonic stem cells. FASEB J. 23, 1907–1919 (2009).
Baker, M. Reproducibility: respect your cells! Nature 537, 433–435 (2016).
Messmer, T. et al. A serum-free media formulation for cultured meat production supports bovine satellite cell differentiation in the absence of serum starvation. Nat. Food 3, 74–85 (2022).
Ahfeldt, T. et al. Programming human pluripotent stem cells into white and brown adipocytes. Nat. Cell Biol. 14, 209–219 (2012).
Taura, D. et al. Adipogenic differentiation of human induced pluripotent stem cells: comparison with that of human embryonic stem cells. FEBS Lett. 583, 1029–1033 (2009).
Rubin, C. S., Hirsch, A., Fung, C. & Rosen, O. M. Development of hormone receptors and hormonal responsiveness in vitro. Insulin receptors and insulin sensitivity in the preadipocyte and adipocyte forms of 3T3-L1 cells. J. Biol. Chem. 253, 7570–7578 (1978).
Elks, M. L. & Manganiello, V. C. A role for soluble cAMP phosphodiesterases in differentiation of 3T3-L1 adipocytes. J. Cell. Physiol. 124, 191–198 (1985).
Liao, W. et al. Suppression of PPAR-γ attenuates insulin-stimulated glucose uptake by affecting both GLUT1 and GLUT4 in 3T3-L1 adipocytes. Am. J. Physiol. Endocrinol. Metab. 293, E219–E227 (2007).
Houseknecht, K. L., Cole, B. M. & Steele, P. J. Peroxisome proliferator-activated receptor gamma (PPARγ) and its ligands: a review. Domest. Anim. Endocrinol. 22, 1–23 (2002).
Tang, Q.-Q., Otto, T. C. & Lane, M. D. Commitment of C3H10T1/2 pluripotent stem cells to the adipocyte lineage. Proc. Natl Acad. Sci. USA. 101, 9607–9611 (2004).
Mehta, F., Theunissen, R. & Post, M. J. Adipogenesis from bovine precursors. Methods Mol. Biol. 1889, 111–125 (2019).
Busato, S. & Bionaz, M. When two plus two is more than four: evidence for a synergistic effect of fatty acids on peroxisome proliferator-activated receptor activity in a bovine hepatic model. Genes 12, 1283 (2021).
Hesami, M. & Jones, A. M. P. Application of artificial intelligence models and optimization algorithms in plant cell and tissue culture. Appl. Microbiol. Biotechnol. 104, 9449–9485 (2020).
Rupert, J. E., Jengelley, D. H. A. & Zimmers, T. A. In vitro, in vivo, and in silico methods for assessment of muscle size and muscle growth regulation. Shock 53, 605–615 (2020).
Trott, D. A., Cuthbert, A. P., Overell, R. W., Russo, I. & Newbold, R. F. Mechanisms involved in the immortalization of mammalian cells by ionizing radiation and chemical carcinogens. Carcinogenesis 16, 193–204 (1995).
Gardell, A. M., Qin, Q., Rice, R. H., Li, J. & Kültz, D. Derivation and osmotolerance characterization of three immortalized tilapia (Oreochromis mossambicus) cell lines. PLoS ONE 9, e95919 (2014).
Keller, C. & Guttridge, D. C. Mechanisms of impaired differentiation in rhabdomyosarcoma. FEBS J. 280, 4323–4334 (2013).
Sulak, M. et al. TP53 copy number expansion is associated with the evolution of increased body size and an enhanced DNA damage response in elephants. eLife 5, e11994 (2016).
Pasitka, L. et al. Spontaneous immortalization of chicken fibroblasts generates stable, high-yield cell lines for serum-free production of cultured meat. Nat. Food 4, 35–50 (2022).
Saad, M. K. et al. Continuous fish muscle cell line with capacity for myogenic and adipogenic-like phenotypes. Sci. Rep. 13, 5098 (2023).
Soice, E. & Johnston, J. Immortalizing cells for human consumption. Int. J. Mol. Sci. 22, 11660 (2021).
Harada, H. et al. Telomerase induces immortalization of human esophageal keratinocytes without p16INK4a inactivation. Mol. Cancer Res. 1, 729–738 (2003).
Mamchaoui, K. et al. Immortalized pathological human myoblasts: towards a universal tool for the study of neuromuscular disorders. Skelet. Muscle 1, 34 (2011).
Thorley, M. et al. Skeletal muscle characteristics are preserved in hTERT/cdk4 human myogenic cell lines. Skelet. Muscle 6, 43 (2016).
Wang, W. et al. Immortalization of chicken preadipocytes by retroviral transduction of chicken TERT and TR. PLoS ONE 12, e0177348 (2017).
Stout, A. J. et al. Immortalized bovine satellite cells for cultured meat applications. ACS Synth. Biol. 12, 1567–1573 (2023).
Sharples, A. P., Al-Shanti, N., Lewis, M. P. & Stewart, C. E. Reduction of myoblast differentiation following multiple population doublings in mouse C2C12 cells: a model to investigate ageing? J. Cell. Biochem. 112, 3773–3785 (2011).
Quevedo, R. et al. Assessment of genetic drift in large pharmacogenomic studies. Cell Syst. 11, 393–401.e2 (2020).
Davis, R. L., Weintraub, H. & Lassar, A. B. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell 51, 987–1000 (1987).
Lattanzi, L. et al. High efficiency myogenic conversion of human fibroblasts by adenoviral vector-mediated MyoD gene transfer. An alternative strategy for ex vivo gene therapy of primary myopathies. J. Clin. Invest. 101, 2119–2128 (1998).
Ito, N., Kii, I., Shimizu, N., Tanaka, H. & Shin’Ichi, T. Direct reprogramming of fibroblasts into skeletal muscle progenitor cells by transcription factors enriched in undifferentiated subpopulation of satellite cells. Sci. Rep. 7, 8097 (2017).
Yin, J. et al. In vitro myogenic and adipogenic differentiation model of genetically engineered bovine embryonic fibroblast cell lines. Biotechnol. Lett. 32, 195–202 (2010).
Tontonoz, P., Hu, E. & Spiegelman, B. M. Stimulation of adipogenesis in fibroblasts by PPARγ2, a lipid-activated transcription factor. Cell 79, 1147–1156 (1994).
Teboul, L. et al. Thiazolidinediones and fatty acids convert myogenic cells into adipose-like cells. J. Biol. Chem. 270, 28183–28187 (1995).
Tanaka, A. et al. Efficient and reproducible myogenic differentiation from human iPS cells: prospects for modeling miyoshi myopathy in vitro. PLoS ONE 8, e61540 (2013).
Rao, L., Qian, Y., Khodabukus, A., Ribar, T. & Bursac, N. Engineering human pluripotent stem cells into a functional skeletal muscle tissue. Nat. Commun. 9, 126 (2018).
Shahini, A. et al. Ameliorating the hallmarks of cellular senescence in skeletal muscle myogenic progenitors in vitro and in vivo. Sci. Adv. 7, eabe5671 (2021).
Shahini, A. et al. NANOG restores the impaired myogenic differentiation potential of skeletal myoblasts after multiple population doublings. Stem Cell Res. 26, 55–66 (2018).
Kallunki, T., Barisic, M., Jäättelä, M. & Liu, B. How to choose the right inducible gene expression system for mammalian studies? Cells 8, 796 (2019).
Stout, A. J. et al. Engineered autocrine signaling eliminates muscle cell FGF2 requirements for cultured meat production. Preprint at bioRxiv https://doi.org/10.1101/2023.04.17.537163 (2023).
Stout, A. J., Mirliani, A. B., Soule-Albridge, E. L., Cohen, J. M. & Kaplan, D. L. Engineering carotenoid production in mammalian cells for nutritionally enhanced cell-cultured foods. Metab. Eng. 62, 126–137 (2020).
Simsa, R. et al. Extracellular heme proteins influence bovine myosatellite cell proliferation and the color of cell-based meat. Foods 8, 521 (2019).
Genovese, N. J., Desmet, D. N. & Schulze, E. Methods for extending the replicative capacity of somatic cells during an ex vivo cultivation process. US patent WO2017124100A1 (2017).
Genovese, N. J., Roberts, R. M. & Telugu, B. P. V. L. Method for scalable skeletal muscle lineage specification and cultivation. US patent US20160227830A1 (2021).
Genovese, N. J., Schulze, E. & Desmet, D. N. Compositions and methods for increasing the efficiency of cell cultures used for food production. US patent WO2019014652A1 (2019).
Dhadwar, S. S., Kayser, K. J. & Genovese, N. J. Generation of cell-based products for consumption that comprise proteins from exotic, endangered, and extinct species. US patent US20220333081A1 (2022).
Factsheet on Alternative Proteins (Singapore Food Agency, 2022).
FDA completes first pre-market consultation for human food made using animal cell culture technology. FDA https://www.fda.gov/food/cfsan-constituent-updates/fda-completes-first-pre-market-consultation-human-food-made-using-animal-cell-culture-technology (2022).
Schulze, E. Premarket Notice for Integral Tissue Cultured Poultry Meat (FDA, 2021).
Holt, S. E., Wright, W. E. & Shay, J. W. Regulation of telomerase activity in immortal cell lines. Mol. Cell. Biol. 16, 2932–2939 (1996).
Van Eenennaam, A. L., De Figueiredo Silva, F., Trott, J. F. & Zilberman, D. Genetic engineering of livestock: the opportunity cost of regulatory delay. Annu. Rev. Anim. Biosci. 9, 453–478 (2021).
USDA and FDA announce a formal agreement to regulate cell-cultured food products from cell lines of livestock and poultry. USDA https://www.usda.gov/media/press-releases/2019/03/07/usda-and-fda-announce-formal-agreement-regulate-cell-cultured-food (2019).
Human food made with cultured animal cells. FSIS USDA http://www.fsis.usda.gov/inspection/compliance-guidance/labeling/labeling-policies/human-food-made-cultured-animal-cells (2023).
Food Safety Aspects of Cell-Based Food (FAO and WHO, 2023); https://doi.org/10.4060/cc4855en
O’Neill, E. N., Cosenza, Z. A., Baar, K. & Block, D. E. Considerations for the development of cost-effective cell culture media for cultivated meat production. Compr. Rev. Food Sci. Food Saf. 20, 686–709 (2021).
Bodiou, V., Moutsatsou, P. & Post, M. J. Microcarriers for upscaling cultured meat production. Front. Nutr. 7, 10 (2020).
Stout, A. J. et al. Simple and effective serum-free medium for sustained expansion of bovine satellite cells for cell cultured meat. Commun. Biol. 5, 466 (2022).
Skrivergaard, S., Rasmussen, M. K., Therkildsen, M. & Young, J. F. Bovine satellite cells isolated after 2 and 5 days of tissue storage maintain the proliferative and myogenic capacity needed for cultured meat production. Int. J. Mol. Sci. 22, 8376 (2021).
Zhu, H. et al. Production of cultured meat from pig muscle stem cells. Biomaterials 287, 121650 (2022).
Sacco, A., Doyonnas, R., Kraft, P., Vitorovic, S. & Blau, H. M. Self-renewal and expansion of single transplanted muscle stem cells. Nature 456, 502–506 (2008).
Garcia, S. M., Tamaki, S., Xu, X. & Pomerantz, J. H. Human satellite cell isolation and xenotransplantation. Methods Mol. Biol. 1668, 105–123 (2017).
Baquero-Perez, B., Kuchipudi, S. V., Nelli, R. K. & Chang, K.-C. A simplified but robust method for the isolation of avian and mammalian muscle satellite cells. BMC Cell Biol. 13, 16 (2012).
Burns, T. A., Kadegowda, A. K. G., Duckett, S. K., Pratt, S. L. & Jenkins, T. C. Palmitoleic (16:1 cis-9) and cis-vaccenic (18:1 cis-11) acid alter lipogenesis in bovine adipocyte cultures. Lipids 47, 1143–1153 (2012).
Hirai, S. et al. Myostatin inhibits differentiation of bovine preadipocyte. Domest. Anim. Endocrinol. 32, 1–14 (2007).
Yuen, J. S. K. Jr et al. Aggregating in vitro-grown adipocytes to produce macroscale cell-cultured fat tissue with tunable lipid compositions for food applications. eLife 12, e82120 (2023).
Shi, X.-E. et al. MicroRNA-199a-5p affects porcine preadipocyte proliferation and differentiation. Int. J. Mol. Sci. 15, 8526–8538 (2014).
Wojciechowicz, T. et al. Original research: orexins A and B stimulate proliferation and differentiation of porcine preadipocytes. Exp. Biol. Med. 241, 1786–1795 (2016).
Vahmani, P. et al. Individual trans 18:1 isomers are metabolised differently and have distinct effects on lipogenesis in 3T3-L1 adipocytes. Lipids 50, 195–204 (2015).
Rodeheffer, M. S., Birsoy, K. & Friedman, J. M. Identification of white adipocyte progenitor cells in vivo. Cell 135, 240–249 (2008).
Gojanovich, A. D. et al. Human adipose-derived mesenchymal stem/stromal cells handling protocols. Lipid droplets and proteins double-staining. Front. Cell Dev. Biol. 6, 33 (2018).
Hemmrich, K. et al. Implantation of preadipocyte-loaded hyaluronic acid-based scaffolds into nude mice to evaluate potential for soft tissue engineering. Biomaterials 26, 7025–7037 (2005).
Acknowledgements
We thank the UC Davis Cultivated Meat Consortium led by D. Block, along with K. Baar, for discussions supporting this work. The National Science Foundation has supported the Cultivated Meat Consortium (2021132) and L.R.S. has received support from the Good Food Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jara, T.C., Park, K., Vahmani, P. et al. Stem cell-based strategies and challenges for production of cultivated meat. Nat Food 4, 841–853 (2023). https://doi.org/10.1038/s43016-023-00857-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43016-023-00857-z