Featured
-
-
Article
| Open Access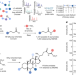 Reverse metabolomics for the discovery of chemical structures from humans
Reverse metabolomics for the discovery of chemical structures from humans
A new discovery strategy, ‘reverse metabolomics’, facilitates high-throughput matching of mass spectrometry spectra in public untargeted metabolomics datasets, and a proof-of-concept experiment identified an association between microbial bile amidates and inflammatory bowel disease.
- Emily C. Gentry
- , Stephanie L. Collins
- & Pieter C. Dorrestein
-
Article |
 Computational prediction of complex cationic rearrangement outcomes
Computational prediction of complex cationic rearrangement outcomes
Computers equipped with a comprehensive knowledge-base of mechanistic steps augmented by physical-organic chemistry rules, as well as quantum mechanical and kinetic calculations, can use a reaction-network approach to analyse the mechanisms of cationic rearrangements.
- Tomasz Klucznik
- , Leonidas-Dimitrios Syntrivanis
- & Bartosz A. Grzybowski
-
Review Article |
 Computational approaches streamlining drug discovery
Computational approaches streamlining drug discovery
Recent advances in computational approaches and challenges in their application to streamlining drug discovery are discussed.
- Anastasiia V. Sadybekov
- & Vsevolod Katritch
-
Article |
 Computer-designed repurposing of chemical wastes into drugs
Computer-designed repurposing of chemical wastes into drugs
A forward-synthesis platform, Allchemy, computationally determines how to ‘close the circle’, or use waste chemicals to make valuable pharmaceutical or agrochemical products, ranking possible routes by environmental, geospatial, and other factors.
- Agnieszka Wołos
- , Dominik Koszelewski
- & Bartosz A. Grzybowski
-
Article |
 Synthon-based ligand discovery in virtual libraries of over 11 billion compounds
Synthon-based ligand discovery in virtual libraries of over 11 billion compounds
V-SYNTHES, a scalable and computationally cost-effective synthon-based approach to compound screening, identified compounds with a high affinity for CB2 and CB1 in a hierarchical structure-based screen of more than 11 billion compounds.
- Arman A. Sadybekov
- , Anastasiia V. Sadybekov
- & Vsevolod Katritch
-
Article |
 Computational planning of the synthesis of complex natural products
Computational planning of the synthesis of complex natural products
A synthetic route-planning algorithm, augmented with causal relationships that allow it to strategize over multiple steps, can design complex natural-product syntheses that are indistinguishable from those designed by human experts.
- Barbara Mikulak-Klucznik
- , Patrycja Gołębiowska
- & Bartosz A. Grzybowski
-
Article |
 A map of the amine–carboxylic acid coupling system
A map of the amine–carboxylic acid coupling system
All possible chemical transformations between amine and carboxylic acid groups are mapped using an automated string-based combinatorics method, showing that the properties and functions of the products vary considerably over all plausible reactions.
- Babak Mahjour
- , Yuning Shen
- & Tim Cernak
-
Article |
 Data-driven design of metal–organic frameworks for wet flue gas CO2 capture
Data-driven design of metal–organic frameworks for wet flue gas CO2 capture
Data mining of a computational library of metal–organic frameworks identifies motifs that bind CO2 sufficiently strongly and whose uptake is not affected by water, with application for the capture of CO2 from flue gases.
- Peter G. Boyd
- , Arunraj Chidambaram
- & Berend Smit
-
Letter |
 Anthropogenic biases in chemical reaction data hinder exploratory inorganic synthesis
Anthropogenic biases in chemical reaction data hinder exploratory inorganic synthesis
Human scientists make unrepresentative chemical reagent and reaction condition choices, and machine-learning algorithms trained on human-selected experiments are less capable of successfully predicting reaction outcomes than those trained on randomly generated experiments.
- Xiwen Jia
- , Allyson Lynch
- & Joshua Schrier
-
Article |
 Holistic prediction of enantioselectivity in asymmetric catalysis
Holistic prediction of enantioselectivity in asymmetric catalysis
A workflow for deriving statistical models of one set of reactions that can be used to predict related reactions is presented, facilitating catalyst and enantioselective reaction development.
- Jolene P. Reid
- & Matthew S. Sigman
-
Letter |
 Unsupervised word embeddings capture latent knowledge from materials science literature
Unsupervised word embeddings capture latent knowledge from materials science literature
Natural language processing algorithms applied to three million materials science abstracts uncover relationships between words, material compositions and properties, and predict potential new thermoelectric materials.
- Vahe Tshitoyan
- , John Dagdelen
- & Anubhav Jain
-
Perspective |
 The digitization of organic synthesis
The digitization of organic synthesis
This Perspective discusses the challenges associated with the prediction of chemical synthesis, in particular the reaction conditions required for organic transformations, and the role of machine-learning approaches in the prediction process.
- Ian W. Davies
-
Article |
 Planning chemical syntheses with deep neural networks and symbolic AI
Planning chemical syntheses with deep neural networks and symbolic AI
Deep neural networks and Monte Carlo tree search can plan chemical syntheses by training models on a huge database of published reactions; their predicted synthetic routes cannot be distinguished from those a human chemist would design.
- Marwin H. S. Segler
- , Mike Preuss
- & Mark P. Waller
-
News |
 Chemical society tried to block business competitor
Chemical society tried to block business competitor
Court ruling raises concerns among some American Chemical Society members.
- Eugenie Samuel Reich

 Identifying general reaction conditions by bandit optimization
Identifying general reaction conditions by bandit optimization