Article
|
Open Access
Featured
-
-
Article
| Open Access Structural evidence for intermediates during O2 formation in photosystem II
Structural evidence for intermediates during O2 formation in photosystem II
Using serial femtosecond X-ray cystallography, we provide structural insights into the final reaction step of Kok’s photosynthetic water oxidation cycle, specifically the S3→[S4]→S0 transition where O2 is formed.
- Asmit Bhowmick
- , Rana Hussein
- & Vittal K. Yachandra
-
Article |
 Structural insights into the inhibition of glycine reuptake
Structural insights into the inhibition of glycine reuptake
Serial synchrotron crystallography reveals the structure of the human glycine transporter GlyT1, showing how a state-specific inhibitor exerts its effects, and potentially informing the design of new GlyT1 inhibitors to treat a range of disorders of the central nervous system.
- Azadeh Shahsavar
- , Peter Stohler
- & Poul Nissen
-
Article |
 Ultrafast structural changes within a photosynthetic reaction centre
Ultrafast structural changes within a photosynthetic reaction centre
Time-resolved serial femtosecond crystallography is used to reveal the structural changes that stabilize the charge-separation steps of electron-transfer reactions in the photosynthetic reaction centre of Blastochloris viridis on a timescale of picoseconds.
- Robert Dods
- , Petra Båth
- & Richard Neutze
-
Article |
 Structures of fungal and plant acetohydroxyacid synthases
Structures of fungal and plant acetohydroxyacid synthases
Structures of the acetohydroxyacid synthase complexes of Saccharomyces cerevisiae and Arabidopsis thaliana provide insights into the biosynthesis of and feedback inhibition by branched-chain amino acids.
- Thierry Lonhienne
- , Yu Shang Low
- & Luke W. Guddat
-
Letter |
 Structures of the intermediates of Kok’s photosynthetic water oxidation clock
Structures of the intermediates of Kok’s photosynthetic water oxidation clock
Crystallography and spectroscopy are used to solve high-resolution structures of the intermediates of Kok’s S-state clock in photosystem II.
- Jan Kern
- , Ruchira Chatterjee
- & Vittal K. Yachandra
-
Letter |
 Human GLP-1 receptor transmembrane domain structure in complex with allosteric modulators
Human GLP-1 receptor transmembrane domain structure in complex with allosteric modulators
Crystal structures of the human GLP-1 receptor in complex with two negative allosteric modulators reveal a common binding pocket, and, together with mutagenesis and modelling studies, further our understanding of the receptor activation mechanism.Author: Please check the wording of the following statement, which will appear online only.
- Gaojie Song
- , Dehua Yang
- & Raymond C. Stevens
-
Letter |
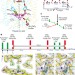 Light-induced structural changes and the site of O=O bond formation in PSII caught by XFEL
Light-induced structural changes and the site of O=O bond formation in PSII caught by XFEL
A new approach, time-resolved serial femtosecond crystallography, is used to view the intermediate states of a photosystem complex following illumination, shedding light on proton transfer and O=O bond formation.
- Michihiro Suga
- , Fusamichi Akita
- & Jian-Ren Shen
-
Letter |
 Structure of photosystem II and substrate binding at room temperature
Structure of photosystem II and substrate binding at room temperature
The structures of three intermediate states of photosystem II, which is crucial for photosynthesis, have been solved at room temperature, shedding new light on this process.
- Iris D. Young
- , Mohamed Ibrahim
- & Junko Yano
-
Letter |
 Structures of riboswitch RNA reaction states by mix-and-inject XFEL serial crystallography
Structures of riboswitch RNA reaction states by mix-and-inject XFEL serial crystallography
Femtosecond XFEL crystallography is used to identify dynamic changes in the adenine riboswitch aptamer domain, with at least four states identified in real time, two in the apo form before binding and two with the ligand bound.
- J. R. Stagno
- , Y. Liu
- & Y.-X. Wang
-
Article |
 De novo phasing with X-ray laser reveals mosquito larvicide BinAB structure
De novo phasing with X-ray laser reveals mosquito larvicide BinAB structure
The structure of the bacterial toxin BinAB, which is used to combat mosquito-borne diseases, reveals pH-sensitive switches and carbohydrate-binding modules that may contribute to the larvicidal function of the toxin.
- Jacques-Philippe Colletier
- , Michael R. Sawaya
- & David S. Eisenberg
-
Letter |
 Macromolecular diffractive imaging using imperfect crystals
Macromolecular diffractive imaging using imperfect crystals
Crystal lattice disorder, which gives rise to a continuous diffraction pattern, is exploited to determine the structure of the integral membrane protein complex photosystem II to a higher resolution than could be achieved using Bragg diffraction alone.
- Kartik Ayyer
- , Oleksandr M. Yefanov
- & Henry N. Chapman
-
Letter |
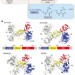 Ribosomal oxygenases are structurally conserved from prokaryotes to humans
Ribosomal oxygenases are structurally conserved from prokaryotes to humans
Crystal structures of human and prokaryotic ribosomal oxygenases reported here, with and without their ribosomal protein substrates, support their assignments as hydroxylases, and provide insights into the evolution of the JmjC-domain-containing hydroxylases and demethylases.
- Rasheduzzaman Chowdhury
- , Rok Sekirnik
- & Christopher J. Schofield

 Influence of pump laser fluence on ultrafast myoglobin structural dynamics
Influence of pump laser fluence on ultrafast myoglobin structural dynamics