Featured
-
-
Article |
 The RNA m6A reader YTHDC1 silences retrotransposons and guards ES cell identity
The RNA m6A reader YTHDC1 silences retrotransposons and guards ES cell identity
N6-methyladenosine RNA and its reader YTHDC1 serve as a bridge to silencing retrotransposons through the RNA derived from these retrotransposons in mouse ES cells.
- Jiadong Liu
- , Mingwei Gao
- & Jiekai Chen
-
Article |
 Cell competition constitutes a barrier for interspecies chimerism
Cell competition constitutes a barrier for interspecies chimerism
Primed pluripotent stem cells from distant species compete with each other, and inactivation of NF-κB signalling in normally outcompeted human cells improves their survival and chimerism in mouse embryos.
- Canbin Zheng
- , Yingying Hu
- & Jun Wu
-
Article |
 Initiation of a conserved trophectoderm program in human, cow and mouse embryos
Initiation of a conserved trophectoderm program in human, cow and mouse embryos
Comparative analysis of human, cow and mouse embryos shows that a mechanism involving atypical protein kinase C initiates the trophectoderm program during the morula stage in these three species.
- Claudia Gerri
- , Afshan McCarthy
- & Kathy K. Niakan
-
Article |
 Dynamic lineage priming is driven via direct enhancer regulation by ERK
Dynamic lineage priming is driven via direct enhancer regulation by ERK
ERK reversibly regulates embryonic stem cell transcription via selective redistribution of co-factors and RNA polymerase from pluripotency to early differentiation enhancers, while leaving transcription factors bound to their enhancers, thus preserving plasticity.
- William B. Hamilton
- , Yaron Mosesson
- & Joshua M. Brickman
-
Letter |
 The SMAD2/3 interactome reveals that TGFβ controls m6A mRNA methylation in pluripotency
The SMAD2/3 interactome reveals that TGFβ controls m6A mRNA methylation in pluripotency
The SMAD2 and SMAD3 protein interactome links TGFβ signalling to diverse effectors including m6A methyltransferase, which has a role in regulating differentiation of human pluripotent stem cells.
- Alessandro Bertero
- , Stephanie Brown
- & Ludovic Vallier
-
Letter |
 Pluripotent state transitions coordinate morphogenesis in mouse and human embryos
Pluripotent state transitions coordinate morphogenesis in mouse and human embryos
Exit of epiblasts from an unrestricted naive pluripotent state is required for epithelialization and generation of the pro-amniotic cavity in mouse embryos and for amniotic cavity formation in human embryos and human embryonic stem cells.
- Marta N. Shahbazi
- , Antonio Scialdone
- & Magdalena Zernicka-Goetz
-
Article |
 Genome editing reveals a role for OCT4 in human embryogenesis
Genome editing reveals a role for OCT4 in human embryogenesis
Genome editing in human zygotes shows that OCT4 is required for normal development at an earlier stage in humans than in mice.
- Norah M. E. Fogarty
- , Afshan McCarthy
- & Kathy K. Niakan
-
Letter |
 Inhibition of mTOR induces a paused pluripotent state
Inhibition of mTOR induces a paused pluripotent state
Inhibition of mechanistic target of rapamycin (mTOR) suspends mouse blastocyst development and the cells remain ‘paused’ in a reversible pluripotent state, allowing prolonged culture.
- Aydan Bulut-Karslioglu
- , Steffen Biechele
- & Miguel Ramalho-Santos
-
Article |
 A developmental coordinate of pluripotency among mice, monkeys and humans
A developmental coordinate of pluripotency among mice, monkeys and humans
Using a single-cell sequencing analysis in monkey embryos, and comparing the genes expressed during early development in this species with those in mice and in human pluripotent stem cells, the authors define characteristics of pluripotency ontogeny across mammalian species.
- Tomonori Nakamura
- , Ikuhiro Okamoto
- & Mitinori Saitou
-
Letter |
 NANOG alone induces germ cells in primed epiblast in vitro by activation of enhancers
NANOG alone induces germ cells in primed epiblast in vitro by activation of enhancers
In mouse embryonic stem cells converted to an epiblast fate in vitro—a state in which the cells can also gain germ cell fate if exposed to the signalling molecule BMP4—the sole expression of the transcription factor NANOG is shown to be sufficient to induce germ cell fate, in the absence of BMP4.
- Kazuhiro Murakami
- , Ufuk Günesdogan
- & M. Azim Surani
-
Article |
 Nanog, Pou5f1 and SoxB1 activate zygotic gene expression during the maternal-to-zygotic transition
Nanog, Pou5f1 and SoxB1 activate zygotic gene expression during the maternal-to-zygotic transition
This study investigates how zygotic transcription is initiated and the maternal transcripts cleared in the zebrafish embryo: using loss-of-function analyses, high-throughput transcriptome sequencing and ribosome footprinting, the important roles of pluripotency factors Nanog, Pou5f1 and SoxB1 during these processes are identified.
- Miler T. Lee
- , Ashley R. Bonneau
- & Antonio J. Giraldez
-
Letter |
 AID stabilizes stem-cell phenotype by removing epigenetic memory of pluripotency genes
AID stabilizes stem-cell phenotype by removing epigenetic memory of pluripotency genes
Fibroblasts deficient in the activation-induced cytidine deaminase (AID) enzyme are shown to fail to stabilize in the pluripotent state, despite initiating the expression of pluripotency genes.
- Ritu Kumar
- , Lauren DiMenna
- & Todd Evans
-
News |
 Cell rewind wins medicine Nobel
Cell rewind wins medicine Nobel
Researchers awarded prestigious prize for their work on reprogramming mature cells to a pluripotent state.
- Alison Abbott
-
News & Views |
 Actors in the cell reprogramming drama
Actors in the cell reprogramming drama
The transformation of skin cells into stem cells is a fascinating but poorly understood process. At last, the molecular characters underlying the initial steps have been revealed. See Letter p.652
- Kyle M. Loh
- & Bing Lim
-
Letter |
 The H3K27 demethylase Utx regulates somatic and germ cell epigenetic reprogramming
The H3K27 demethylase Utx regulates somatic and germ cell epigenetic reprogramming
The H3K27 demethylase Utx is reported to be a critical regulator for the initiation of somatic and germ cell reprogramming.
- Abed AlFatah Mansour
- , Ohad Gafni
- & Jacob H. Hanna
-
Article |
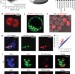 Embryonic stem cell potency fluctuates with endogenous retrovirus activity
Embryonic stem cell potency fluctuates with endogenous retrovirus activity
A rare cell subpopulation within mouse embryonic stem cell cultures is identified that exhibits properties of two-cell (2C) embryos; the interconversion of ES cells to 2C cells correlates with endogenous retroviral activity.
- Todd S. Macfarlan
- , Wesley D. Gifford
- & Samuel L. Pfaff
-
Letter |
 Control of ground-state pluripotency by allelic regulation of Nanog
Control of ground-state pluripotency by allelic regulation of Nanog
Tight regulation of Nanog dose at the chromosome level is important for the acquisition of pluripotency during development.
- Yusuke Miyanari
- & Maria-Elena Torres-Padilla
-
Review Article |
 The promise of induced pluripotent stem cells in research and therapy
The promise of induced pluripotent stem cells in research and therapy
- Daisy A. Robinton
- & George Q. Daley
-
Article |
 lincRNAs act in the circuitry controlling pluripotency and differentiation
lincRNAs act in the circuitry controlling pluripotency and differentiation
- Mitchell Guttman
- , Julie Donaghey
- & Eric S. Lander
-
Letter |
 Molecular coupling of Tsix regulation and pluripotency
Molecular coupling of Tsix regulation and pluripotency
Reprogramming of X-chromosome inactivation during the acquisition of pluripotency is accompanied by repression of Xist, the trigger of X-inactivation, and by upregulation of its antisense counterpart, Tsix. In undifferentiated embryonic stem cells (ESCs), key transcription factors that support pluripotency repress Xist transcription. These authors show that upregulation of Tsix in ESCs depends on a different subset of pluripotency factors. Therefore, two distinct ESC-specific complexes couple reprogramming of X-inactivation to pluripotency.
- Pablo Navarro
- , Andrew Oldfield
- & Philip Avner
-
Brief Communications Arising |
 Can controversies be put to REST?
Can controversies be put to REST?
- Helle F. Jørgensen
- & Amanda G. Fisher
-
Article |
 Epigenetic memory in induced pluripotent stem cells
Epigenetic memory in induced pluripotent stem cells
Pluripotent stem cells can be generated in the laboratory through somatic cell nuclear transfer (generating nuclear transfer embryonic stem cells, ntESCs) or transcription-factor-based reprogramming (producing induced pluripotent stem cells, iPSCs). These methods reset the methylation signature of the genome — but to what extent? Here it is found that mouse iPSCs 'remember' the methylation status of their tissue of origin, but the methylation of ntESCs is more similar to that of naturally produced ES cells.
- K. Kim
- , A. Doi
- & G. Q. Daley
-
-
-
Review Article |
 Nuclear reprogramming to a pluripotent state by three approaches
Nuclear reprogramming to a pluripotent state by three approaches
- Shinya Yamanaka
- & Helen M. Blau
-
Letter |
 Chromatin signature of embryonic pluripotency is established during genome activation
Chromatin signature of embryonic pluripotency is established during genome activation
To study the changes in chromatin structure that accompany zygotic genome activation and pluripotency during the maternal–zygotic transition (MZT), the genomic locations of histone H3 modifications and RNA polymerase II have been mapped during this transition in zebrafish embryos. H3 lysine 27 trimethylation and H3 lysine 4 trimethylation are only detected after MZT; evidence is provided that the bivalent chromatin domains found in cultured embryonic stem cells also exist in embryos.
- Nadine L. Vastenhouw
- , Yong Zhang
- & Alexander F. Schier

 Metabolic regulation of species-specific developmental rates
Metabolic regulation of species-specific developmental rates Stem-cell furore erupts
Stem-cell furore erupts Extrinsic regulation of pluripotent stem cells
Extrinsic regulation of pluripotent stem cells