Article
|
Open Access
Featured
-
-
Article
| Open Access TDP-43 forms amyloid filaments with a distinct fold in type A FTLD-TDP
TDP-43 forms amyloid filaments with a distinct fold in type A FTLD-TDP
Cryo-electron microscopy structures and mass spectrometry analyses show that TAR DNA-binding protein 43 (TDP-43) forms amyloid filaments with a distinct fold in type A frontotemporal lobar degeneration with TDP-43 pathology (FTLD-TDP) compared with TDP-43 filaments in type B FTLD-TDP and amyotrophic lateral sclerosis.
- Diana Arseni
- , Renren Chen
- & Benjamin Ryskeldi-Falcon
-
Article
| Open Access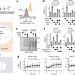 A cytosolic surveillance mechanism activates the mitochondrial UPR
A cytosolic surveillance mechanism activates the mitochondrial UPR
We identify a highly controlled cytosolic surveillance mechanism that integrates independent mitochondrial stress signals to initiate the mitochondrial unfolded protein response (UPR), revealing a link between mitochondrial and cytosolic proteostasis.
- F. X. Reymond Sutandy
- , Ines Gößner
- & Christian Münch
-
Article |
 HSP40 proteins use class-specific regulation to drive HSP70 functional diversity
HSP40 proteins use class-specific regulation to drive HSP70 functional diversity
The binding and activation of HSP70 by class B J-domain proteins is subject to an autoinhibitory regulatory mechanism that controls substrate targeting to HSP70 and is required for the disaggregation of amyloid fibres.
- Ofrah Faust
- , Meital Abayev-Avraham
- & Rina Rosenzweig
-
Article |
 Molecular dissection of amyloid disaggregation by human HSP70
Molecular dissection of amyloid disaggregation by human HSP70
The molecular steps that lead to the disaggregation of amyloid fibrils are shown to involve the synergistic action of HSP70 and its co-chaperones DNAJB1 and HSP110.
- Anne S. Wentink
- , Nadinath B. Nillegoda
- & Bernd Bukau
-
Article |
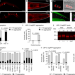 Extracellular proteostasis prevents aggregation during pathogenic attack
Extracellular proteostasis prevents aggregation during pathogenic attack
A systematic analysis of the proteostasis network of secreted proteins in Caenorhabditis elegans identifies numerous regulators of protein homeostasis outside the cell, and highlights the contribution of extracellular proteostasis to host defence.
- Ivan Gallotta
- , Aneet Sandhu
- & Della C. David
-
Article |
 Processive extrusion of polypeptide loops by a Hsp100 disaggregase
Processive extrusion of polypeptide loops by a Hsp100 disaggregase
A combination of optical tweezers and fluorescent-particle tracking is used to dissect the dynamics of the Hsp100 disaggregase ClpB, and show that the processive extrusion of polypeptide loops is the mechanistic basis of its activity.
- Mario J. Avellaneda
- , Kamila B. Franke
- & Sander J. Tans
-
Article |
 Regulation of α-synuclein by chaperones in mammalian cells
Regulation of α-synuclein by chaperones in mammalian cells
Chaperones interact with a canonical motif in α-synuclein, which can be prevented by phosphorylation of α-synuclein at Tyr39, whereas inhibition of this interaction leads to the localization of α-synuclein to the mitochondria and aggregate formation.
- Björn M. Burmann
- , Juan A. Gerez
- & Sebastian Hiller
-
Letter |
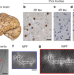 Structures of filaments from Pick’s disease reveal a novel tau protein fold
Structures of filaments from Pick’s disease reveal a novel tau protein fold
The structures of tau filaments from patients with the neurodegenerative disorder Pick’s disease show that the filament fold is different from that of the tau filaments found in Alzheimer’s disease.
- Benjamin Falcon
- , Wenjuan Zhang
- & Michel Goedert
-
Letter |
 Cotranslational assembly of protein complexes in eukaryotes revealed by ribosome profiling
Cotranslational assembly of protein complexes in eukaryotes revealed by ribosome profiling
Cotranslational assembly is a prevalent mechanism for the formation of oligomeric complexes in Saccharomyces cerevisiae, with one subunit serving as scaffold for the translation of partner subunits.
- Ayala Shiber
- , Kristina Döring
- & Bernd Bukau
-
Letter |
 A lysosomal switch triggers proteostasis renewal in the immortal C. elegans germ lineage
A lysosomal switch triggers proteostasis renewal in the immortal C. elegans germ lineage
Sperm-activated lysosomes enhance proteostasis in nematode oocytes just before fertilization; this could prevent transmission of damaged proteins to the next generation and may explain the immortality of the germ-cell lineage.
- K. Adam Bohnert
- & Cynthia Kenyon
-
Letter |
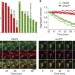 Cytosolic proteostasis through importing of misfolded proteins into mitochondria
Cytosolic proteostasis through importing of misfolded proteins into mitochondria
Proteins prone to aggregation in yeast are imported into mitochondria under stress conditions, suggesting that mitochondrial import and proteolysis may help to disaggregate proteins in the cytoplasm.
- Linhao Ruan
- , Chuankai Zhou
- & Rong Li
-
Letter |
 Alternative modes of client binding enable functional plasticity of Hsp70
Alternative modes of client binding enable functional plasticity of Hsp70
Hsp70 binds unfolded protein segments in its groove, but can also bind and stabilize folded protein structures, owing to its moveable lid, with ATP hydrolysis and co-chaperones allowing control of these contrasting effects.
- Alireza Mashaghi
- , Sergey Bezrukavnikov
- & Sander J. Tans
-
Article |
 Failure of RQC machinery causes protein aggregation and proteotoxic stress
Failure of RQC machinery causes protein aggregation and proteotoxic stress
Defects in the ribosome quality control (RQC) complex, which clears proteins that stalled during translation, can cause neurodegeneration; here it is shown that in RQC-defective cells a peptide tail added by the RQC subunit 2 to stalled polypeptides promotes their aggregation and the sequestration of chaperones in these aggregates, affecting normal protein quality control processes.
- Young-Jun Choe
- , Sae-Hun Park
- & F. Ulrich Hartl
-
Letter |
 Crucial HSP70 co-chaperone complex unlocks metazoan protein disaggregation
Crucial HSP70 co-chaperone complex unlocks metazoan protein disaggregation
An efficient protein disaggregation system uncovered in metazoan cells requires transient interactions between J-protein co-chaperones of classes A and B, which synergistically boost HSP70-dependent disaggregation activity, providing a flexible further level of regulation for metazoan protein quality control, with direct relevance to human diseases such as age-related neurodegeneration.
- Nadinath B. Nillegoda
- , Janine Kirstein
- & Bernd Bukau
-
Letter |
 Excitation-induced ataxin-3 aggregation in neurons from patients with Machado–Joseph disease
Excitation-induced ataxin-3 aggregation in neurons from patients with Machado–Joseph disease
Human neurons derived from induced pluripotent stem cells permit the study of aberrant protein processing and aggregation in Machado–Joseph disease.
- Philipp Koch
- , Peter Breuer
- & Oliver Brüstle
-
Letter |
 α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation
α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation
- Tim Bartels
- , Joanna G. Choi
- & Dennis J. Selkoe
-
Outlook |
 Amyloid: Little proteins, big clues
Amyloid: Little proteins, big clues
After a quarter of a century, the amyloid hypothesis for Alzheimer's disease is reconnecting to its roots in prion research.
- Jim Schnabel
-
Research Highlights |
Toxic clumps trap many proteins
-
Research Highlights |
Ageing: Proteins clump with age
-
Research Highlights |
Pathology: Bent out of shape

 TAF15 amyloid filaments in frontotemporal lobar degeneration
TAF15 amyloid filaments in frontotemporal lobar degeneration