Article
|
Open Access
Featured
-
-
Article |
 Structure of the nutrient-sensing hub GATOR2
Structure of the nutrient-sensing hub GATOR2
The 3D structure of the human nutrient-sensing complex GATOR2 is resolved using cryo-electron microscopy.
- Max L. Valenstein
- , Kacper B. Rogala
- & David M. Sabatini
-
Article |
 Excised linear introns regulate growth in yeast
Excised linear introns regulate growth in yeast
A set of 34 excised introns in Saccharomyces cerevisiae, characterized by having a short distance between the lariat branch point and the 3′ splice site, have a biological function within the TOR growth-signalling network.
- Jeffrey T. Morgan
- , Gerald R. Fink
- & David P. Bartel
-
Letter |
 Subcellular transcriptomes and proteomes of developing axon projections in the cerebral cortex
Subcellular transcriptomes and proteomes of developing axon projections in the cerebral cortex
A subcellular sorting approach enables quantitative analysis of subtypes of growth cones in the brain, and reveals subcellular relationships between local mRNA and local proteomes in developing projection neurons.
- Alexandros Poulopoulos
- , Alexander J. Murphy
- & Jeffrey D. Macklis
-
Letter |
 KLHL22 activates amino-acid-dependent mTORC1 signalling to promote tumorigenesis and ageing
KLHL22 activates amino-acid-dependent mTORC1 signalling to promote tumorigenesis and ageing
In response to amino acid stimulation, the ubiquitin E3 ligase CUL3–KLHL22 promotes the activation of mTORC1, which may drive tumour growth in breast cancer.
- Jie Chen
- , Yuhui Ou
- & Ying Liu
-
Letter |
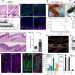 An evolutionarily conserved ribosome-rescue pathway maintains epidermal homeostasis
An evolutionarily conserved ribosome-rescue pathway maintains epidermal homeostasis
Loss of the ribosome-rescue factor Pelo in a subset of mouse epidermal stem cells results in hyperproliferation and altered differentiation of these cells.
- Kifayathullah Liakath-Ali
- , Eric W. Mills
- & Fiona M. Watt
-
Article |
 Mechanisms of mTORC1 activation by RHEB and inhibition by PRAS40
Mechanisms of mTORC1 activation by RHEB and inhibition by PRAS40
The cryo-electron microscopy and crystal structures of several mTORC1 complexes, and accompanying biochemical analyses, shed light on how mTORC1 is regulated and how cancer mutations lead to its hyperactivation.
- Haijuan Yang
- , Xiaolu Jiang
- & Nikola P. Pavletich
-
Letter |
 TRAF2 and OTUD7B govern a ubiquitin-dependent switch that regulates mTORC2 signalling
TRAF2 and OTUD7B govern a ubiquitin-dependent switch that regulates mTORC2 signalling
Ubiquitination of the GβL subunit, a component of both mTORC1 and mTORC2, acts as a regulatory switching mechanism to balance levels of mTORC1 and mTORC2; the failure of this mechanism in some cancers leads to elevated mTORC2 formation and tumorigenesis.
- Bin Wang
- , Zuliang Jie
- & Wenyi Wei
-
Letter |
 KICSTOR recruits GATOR1 to the lysosome and is necessary for nutrients to regulate mTORC1
KICSTOR recruits GATOR1 to the lysosome and is necessary for nutrients to regulate mTORC1
A protein complex composed of KPTN, ITFG2, C12orf66 and SZT2, named KICSTOR, is necessary for lysosomal localization of GATOR1, interaction of GATOR1 with the Rag GTPases and GATOR2, and nutrient-dependent mTORC1 modulation.
- Rachel L. Wolfson
- , Lynne Chantranupong
- & David M. Sabatini
-
Letter |
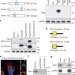 mTORC1 and muscle regeneration are regulated by the LINC00961-encoded SPAR polypeptide
mTORC1 and muscle regeneration are regulated by the LINC00961-encoded SPAR polypeptide
The polypeptide SPAR is encoded by a long non-coding RNA, localizes to the late endosome and lysosome, and regulates muscle regeneration by inhibiting mTORC1.
- Akinobu Matsumoto
- , Alessandra Pasut
- & Pier Paolo Pandolfi
-
Letter |
 Mechanism of arginine sensing by CASTOR1 upstream of mTORC1
Mechanism of arginine sensing by CASTOR1 upstream of mTORC1
Structural data on the protein CASTOR1 reveal how the mTORC1 pathway senses intracellular arginine, suggesting a repurposing of an evolutionarily pre-metazoan mechanism.
- Robert A. Saxton
- , Lynne Chantranupong
- & David M. Sabatini
-
Letter |
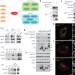 SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1
SLC38A9 is a component of the lysosomal amino acid sensing machinery that controls mTORC1
The mTORC1 protein kinase complex integrates nutrient and growth stimuli to modulate signalling pathways that regulate cellular metabolism and physiology, but the molecular nature of the amino acid sensing mechanism at the lysosome is unknown; here, an orphan member of the human solute carrier group of proteins, SLC38A9, is shown to be an integral component of the lysosomal machinery that can directly sense amino acids and activate mTORC1.
- Manuele Rebsamen
- , Lorena Pochini
- & Giulio Superti-Furga
-
Article |
 RNA G-quadruplexes cause eIF4A-dependent oncogene translation in cancer
RNA G-quadruplexes cause eIF4A-dependent oncogene translation in cancer
The translation of many messenger RNAs that encode important oncogenes and transcription factors depends on the eIF4A RNA helicase to resolve G-quadruplex structures, implying eIF4A inhibition as an effective cancer therapy.
- Andrew L. Wolfe
- , Kamini Singh
- & Hans-Guido Wendel
-
Article |
 mTOR kinase structure, mechanism and regulation
mTOR kinase structure, mechanism and regulation
Co-crystal structures of a number of complexes involving truncated mammalian target of rapamycin, a phosphoinositide 3-kinase-related protein kinase, reveal an intrinsically active kinase conformation and show how rapamycin–FKBP12 directly blocks substrate recruitment to the kinase domain.
- Haijuan Yang
- , Derek G. Rudge
- & Nikola P. Pavletich
-
News & Views |
A healthy diet for stem cells
Understanding how a low calorie intake slows ageing could revolutionize the way that we treat age-related diseases. One potential key to such treatments could be to enhance the local environment of stem cells. See Article p.490
- Fresnida J. Ramos
- & Matt Kaeberlein
-
Article |
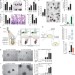 mTORC1 in the Paneth cell niche couples intestinal stem-cell function to calorie intake
mTORC1 in the Paneth cell niche couples intestinal stem-cell function to calorie intake
In the mouse intestine, calorie restriction enhances the regenerative capacity of intestinal stem cells by reducing mTORC1 signalling in their Paneth cell niche.
- Ömer H. Yilmaz
- , Pekka Katajisto
- & David M. Sabatini
-
News & Views |
 The director's cut
The director's cut
A genome-wide characterization of active translation of messenger RNA following inhibition of mTOR will transform our view of this signalling protein's regulatory role in cancer. See Article p.55 & Letter p.109
- Antonio Gentilella
- & George Thomas
-
Letter |
 mTORC1 controls fasting-induced ketogenesis and its modulation by ageing
mTORC1 controls fasting-induced ketogenesis and its modulation by ageing
During periods of fasting the liver produces ketone bodies, which the peripheral tissues can use as a source of energy. Here it is shown that fasting inhibits multi-component mTOR complex 1 (mTORC1) in the liver. Inhibition of mTORC1 is required for activation of PPARα, a master regulator that switches on genes involved in ketogenesis. Livers from aged mice have increased mTORC1 signalling, reduced PPARα activity, and reduced ketone production. The observation that mTORC1 promotes an ageing phenotype in the liver fits well with the observation that inhibition of this pathway increases lifespan in several organisms.
- Shomit Sengupta
- , Timothy R. Peterson
- & David M. Sabatini
-
Research Highlights |
Neuroscience: Quick mood lift
-
Letter |
 Termination of autophagy and reformation of lysosomes regulated by mTOR
Termination of autophagy and reformation of lysosomes regulated by mTOR
When cells are starved, the enzyme TOR is inhibited, inducing autophagy. In this process, autophagosomes sequester intracellular components and then fuse with lysosomes, producing autolysosomes in which cargo is degraded to regenerate nutrients. Now, a mechanism is revealed by which lysosomes are re-formed. When starvation conditions are prolonged, mTOR is re-activated; this attenuates autophagy and results in tubules and vesicles extruding from the autolysosome and maturing into functional lysosomes.
- Li Yu
- , Christina K. McPhee
- & Michael J. Lenardo

 Cryo-EM structure of the SEA complex
Cryo-EM structure of the SEA complex