Abstract
Aim
The goal of this case report is to describe the dermatologic and conjunctival findings in a case of bilateral diffuse uveal melanocytic proliferation (BDUMP), a paraneoplastic syndrome usually associated with gynecologic cancers. There is little information about other dermatologic melanocytic findings in these patients.
Methods
Histologic and fluorescent in situ hybridization (FISH) analysis of three separate skin biopsies, one of which was separated by 21 months from the others, were performed in a 71-year-old patient with BDUMP to assess for histologic and chromosomal abnormality. Conjunctival histologic evaluation was also done.
Results
Dermal melanocytic proliferation was seen in each specimen. The cells were spindle type with mitotic activity. FISH analysis showed a normal copy of chromosomes. The conjunctival sample also showed normal FISH analysis.
Conclusion
BDUMP is associated with multifocal dermal and conjunctival melanocytic proliferation.
Similar content being viewed by others
Introduction
Bilateral diffuse uveal melanocytic proliferation (BDUMP) is a paraneoplastic syndrome usually associated with gynecologic cancers.1, 2, 3, 4, 5, 6, 7, 8 It is associated with thickening of the uvea because of hyperplastic proliferation of benign uveal melanocytes.1, 2, 3, 4, 5, 6, 7, 8 Recent data show that it can be ameliorated by plasma exchange, suggesting that there is a proliferation-inducing factor in the plasma.7, 8 A recent study has shown that plasma from patients with BDUMP contains an IgG that causes cultured melanocytes to proliferate and elongate, which we have termed cultured melanocyte elongation and proliferation factor (CMEP factor).9
There are rare reports about nonuveal melanocytic proliferation in BDUMP, but some of these observations have been interpreted to be melanoma metastases to the dermal layers.6 Others have stated that the condition involves melanophages in the mucosa.5, 10 Here, we describe a patient with classic BDUMP who had benign dermal and conjunctival melanocytic proliferations. In addition, we show that there are no chromosomal changes in the melanocytes, which further lends credence to the fact that the melanocytic proliferation in tissues other than the eye are also benign melanocytic proliferations.
Subjects and methods
A 71-year-old patient presented in 2009 with bilaterally decreased vision and thickening of the uvea with an overlying serous retinal detachment (Figure 1a). The intraocular findings were previously described; and briefly, she fulfilled all the criteria for BDUMP including presenting vision of 20/40 in both eyes, diffuse thickening of the uvea, as well as having circumscribed uveal proliferations, 1–2 + nuclear sclerosis with mild posterior subcapsular changes of the lenses, and intraocular pressures of 8 and 14.7
The patient had a history of biopsy-proven papillary serous adenocarcinoma (FIGO grade 3/3) involving the entire endometrial cavity and invading the myometrial wall, forming an 8.0 × 4.3 × 2.1-cm mass with extension to the lower uterine segment. A total of 4 out of 43 lymph nodes were positive with metastatic disease. She was treated with chemotherapy but had continued lymphadenopathy. A diagnosis of BDUMP was made.
Dermatologic examination showed multiple seborrheic keratoses and a 2-mm bluish pigmented papule on the dorsum of the hand that the patient felt had been growing (Figure 1b). This was biopsied and showed mitotically active melanocytes in the dermis.
The patient underwent plasma exchange followed by chemotherapy. Vision improved and the serous detachment resolved. The patient then did well clinically for almost 2 years on maintenance chemotherapy; but 21 months after her initial presentation, her vision subjectively and objectively decreased in the right eye from 20/40 to 20/80, and there was evidence of recurrence of a serous retinal detachment consistent with recurrence of BDUMP in both eyes. There was also recurrence of metastatic disease, which was noted in the inguinal and hepatoduodenal nodes. In addition, the patient noticed three new pigmented papules, one on the right wrist, one on the right posterior upper arm and one on the left lateral lower leg (Figure 1c). In addition, there was pigmentation of the inferior forniceal conjunctiva on both sides (Figure 1d). Biopsy of each of the skin lesions and of the left forniceal conjunctiva was performed. As a result of a question of whether these could be metastases to the dermis instead of hyperplastic dermal melanocytes, fluorescent in situ hybridization (FISH) was performed on two of the skin biopsies (conjunctival biopsy and skin biopsy from 2009). Plasma exchange and resumption of chemotherapy was again performed, and the patient subsequently expired approximately 3 years following the initial diagnosis of BDUMP.
Methods
Biopsies were placed in 10% buffered formalin, sectioned at 3–5 microns and evaluated with hematoxylin and eosin stain. At least two separate pathologists evaluated each sample. One of the co-authors (TJF) evaluated each one.
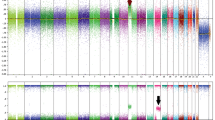
FISH analysis of the nuclei was performed using a Melano-site 4 probe cocktail (Abbott Molecular, Des Plaines, IL, USA and NeoGenomics, Irvine, CA, USA). The probe set was directed to chromosome 6: RREB1, chromosome 6: CEN6, chromosome 6: MYB, and chromosome 11: CCND1 (9–11).
Results
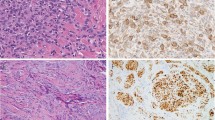
In the skin biopsies from 2009 and 2011, there was a nodular proliferation of melanocytic lineage cells in the mid-dermis. The melanocytic lineage cells had a small-to-moderate amount of cytoplasm, and the nuclei were approximately the size of mid-level keratinocyte nuclei. The nuclei were uniform and had evenly distributed nuclear chromatin. Scattered mitotic figures were identified in the dermal component (Figures 2a and b). The cells appeared to be bland in appearance and uniform in size. The conjunctival biopsy from 2011 showed subepithelial melanocytic proliferation, and the cells had similar morphologic features to the ones seen in the skin lesions (Figures 2c and d).
Magnified view of skin lesion at × 40 and × 100: (a) Scanning magnification of skin from dorsum of left hand showing a nodular proliferation of melanocytic lineage cells (hematoxylin and eosin, × 40). (b) Intermediate magnification of skin showing a proliferation of bland-appearing melanocytic lineage cells (hematoxylin and eosin, × 100). Magnified view of the inferior forniceal conjunctival lesion at × 40 and × 100: (c) Conjunctival biopsy showing relatively circumscribed subepithelial melanocytic proliferation (hematoxylin and eosin, × 40). (d) Conjunctival biopsy done in 2011, showing the uniform population of bland spindle-shaped melanocytes (hematoxylin and eosin, × 100).
FISH studies performed in all four lesions exhibited signal patterns within normal ranges. On the two skin biopsies from 2011, 468 nuclei and 219 nuclei were scored by FISH per probe. On the 2009 cutaneous biopsy, 332 nuclei were scored; and on the conjunctival biopsy from 2011, at least 45 nuclei were scored (Figure 3). These results supported that these lesions were benign melanocytic proliferations rather than malignant melanomas.
Conclusion
Machemer first described BDUMP in 1966.1 Since then, it has been described as a paraneoplastic syndrome associated with distal cancers.1, 2, 3, 4, 5, 6, 7, 8 The uveal melanocytic proliferation is a sine qua non of this entity; and in the past, progressive vision loss has been the rule. Along with others, we have recently shown that plasma exchange improves the prognosis.7, 8
The cause of proliferation of the uveal melanocytes is unknown; however, we recently have shown that there is a substance in the IgG fraction of the serum that causes cultured melanocytes to proliferate. This factor has been termed CMEP factor.9
One also wonders whether or not melanocytes in tissues other than the uvea also proliferate. In a review of the literature, of 28 cases reported by 2003, 25% had evidence of nonocular areas of hyperpigmentation, many of which involved mucosal membranes including the oral mucosa, the vagina, and the penis.11 It has been proposed that the condition includes metastatic melanomas in the dermis.6 Other reports have found similar melanocytic proliferation in the skin in a patient with BDUMP, although no FISH analysis was performed.10, 11 It is difficult to distinguish hyperplastic melanocytes from metastases in the dermis since hyperplasia in the dermis is a rare event.11 However, the fact that there is no atypia is helpful, and the presence of normal FISH decreases the possibility of the cells representing metastases. Although normal chromosomal analysis can still be seen in about 16% of skin melanomas, the fact that we had FISH analysis of two skin biopsies separated by 2 years, and also a conjunctival biopsy decreases the possibility of missing frankly malignant melanomas.12, 13, 14
The cause of proliferation of melanocytes in the dermis and conjunctiva can only be speculated; but it is known that, embryologically, the neural crest cells initially migrate into the dermis where kit ligand released by the keratinocytes cause the vast majority of melanocytes to then migrate into the epidermis.15, 16 The epidermal melanocytes are strongly affected by the adjoining keratinocytes and respond differently to signals than the free uveal and dermal melanocytes. This most likely explains why there is no epidermal melanocytic proliferation in the presence of CMEP in BDUMP patients. There are possibly small rests of dermal melanocytes that can proliferate in the presence of CMEP. What would happen if BDUMP occurred in the presence of a pre-existing extensive dermal nevi is not well-characterized.17
In summary, BDUMP can cause melanocytic proliferation in the dermis and the conjunctiva. If the history of gynecological malignancy is unknown, histologically, these lesions may be mistaken for metastatic melanomas. Careful evaluation of the skin should be performed at the initial diagnosis of BDUMP to determine if there are small foci of dermal melanocytic proliferation as well. Finally, the name should be changed to bilateral diffuse uveal and focal dermal melanocytic proliferation (BDUFDMP) to enlighten the clinician of its dermatologic component as well.

References
Machemer R . [On the pathogenesis of the flat malignant melanoma]. Klin Monbl Augenheilkd 1966; 148 (5): 641–652.
Gass JD, Gieser RG, Wilkinson CP, Beahm DE, Pautler SE . Bilateral diffuse uveal melanocytic proliferation in patients with occult carcinoma. Arch Ophthalmol 1990; 108 (4): 527–533.
Ulrich JN, Garg S, Escaravage GK Jr., Meredith TM . Bilateral diffuse uveal melanocytic proliferation presenting as small choroidal melanoma. Case Rep Ophthalmol Med 2011; 2011: 740640.
Sen J, Clewes AR, Quah SA, Hiscott PS, Bucknall RC, Damato BE . Presymptomatic diagnosis of bronchogenic carcinoma associated with bilateral diffuse uveal melanocytic proliferation. Clin Experiment Ophthalmol 2006; 34 (2): 156–158.
Shinmura M, Hamasaki D, Matsuyama T, Takagi K, Sadamura Y, Momi H et al. [A case of squamous cell carcinoma of the lung with bilateral diffuse uveal melanocytic proliferation (BDUMP) diagnosed by visual disturbance]. Nihon Kokyuki Gakkai Zasshi 2011; 49 (3): 187–191.
Duong HV, McLean IW, Beahm DE . Bilateral diffuse melanocytic proliferation associated with ovarian carcinoma and metastatic malignant amelanotic melanoma. Am J Ophthalmol 2006; 142 (4): 693–695.
Jaben EA, Pulido JS, Pittock S, Markovic S, Winters JL . The potential role of plasma exchange as a treatment for bilateral diffuse uveal melanocytic proliferation: a report of two cases. J Clin Apher 2011; 26 (6): 356–361.
Mets RB, Golchet P, Adamus G, Anitori R, Wilson D, Shaw J et al. Bilateral diffuse uveal melanocytic proliferation with a positive ophthalmoscopic and visual response to plasmapheresis. Arch Ophthalmol 2011; 129 (9): 1235–1238.
Miles SL, Niles RM, Pittock S, Vile R, Davies J, Winters JL et al. A factor found in the IgG fraction of serum of patients with paraneoplastic bilateral diffuse uveal melanocytic proliferation causes proliferation of cultured human melanocytes. Retina 2012; 32 (9): 1959–1966.
Gass JD, Glatzer RJ . Acquired pigmentation simulating Peutz-Jeghers syndrome: initial manifestation of diffuse uveal melanocytic proliferation. Br J Ophthalmol 1991; 75 (11): 693–695.
O'Neal KD, Butnor KJ, Perkinson KR, Proia AD . Bilateral diffuse uveal melanocytic proliferation associated with pancreatic carcinoma: a case report and literature review of this paraneoplastic syndrome. Surv Ophthalmol 2003; 48 (6): 613–625.
Gerami P, Jewell SS, Morrison LE, Blondin B, Schulz J, Ruffalo T et al. Fluorescence in situ hybridization (FISH) as an ancillary diagnostic tool in the diagnosis of melanoma. Am J Surg Pathol 2009; 33 (8): 1146–1156.
Morey AL, Murali R, McCarthy SW, Mann GJ, Scolyer RA . Diagnosis of cutaneous melanocytic tumours by four-colour fluorescence in situ hybridisation. Pathology 2009; 41 (4): 383–387.
Newman MD, Lertsburapa T, Mirzabeigi M, Mafee M, Guitart J, Gerami P . Fluorescence in situ hybridization as a tool for microstaging in malignant melanoma. Mod Pathol 2009; 22 (8): 989–995.
Cramer SF, Fesyuk A . On the development of neurocutaneous units—implications for the histogenesis of congenital, acquired, and dysplastic nevi. Am J Dermatopathol 2012; 34 (1): 60–81.
Hirobe T . How are proliferation and differentiation of melanocytes regulated? Pigment Cell Melanoma Res 2011; 24 (3): 462–478.
Prause JU, Jensen OA, Eisgart F, Hansen U, Kieffer M . Bilateral diffuse malignant melanoma of the uvea associated with large cell carcinoma, giant cell type, of the lung. Case report of a newly described syndrome. Ophthalmologica 1984; 189 (4): 221–228.
Acknowledgements
This study was supported in part by unrestricted grants from Research to Prevent Blindness Inc. and from Terry and Judith Paul.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Pulido, J., Flotte, T., Raja, H. et al. Dermal and conjunctival melanocytic proliferations in diffuse uveal melanocytic proliferation. Eye 27, 1058–1062 (2013). https://doi.org/10.1038/eye.2013.131
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2013.131
Keywords
This article is cited by
-
Diagnostic and therapeutic considerations in patients with bilateral diffuse uveal melanocytic proliferation
International Ophthalmology (2024)