Abstract
Pretreatment classification tools are used in prostate cancer to inform patient management. The effect of cribriform pattern 4 (CC) and intraductal carcinoma (IDC) on such nomograms is still underexplored. We analyzed the Cancer of Prostate Risk Assessment (CAPRA) and National Comprehensive Cancer Network (NCCN) risk scores in cases with and without CC/IDC to assess impact on biochemical recurrence (BCR) and metastases/death of prostate cancer (event free survival-EFS) after prostatectomy. A matched biopsy- prostatectomy cohort (2010–2017) was reviewed for CC/IDC. CAPRA and NCCN scores were calculated. CAPRA score 0–2 were deemed “low”, 3–5 “intermediate” and 6–10 “high”. NCCN scores 1–2 “very low/low”, 3 “favorable intermediate”, 4 “unfavorable intermediate”, 5–6 “high/very high”. Cases were stratified by presence of CC/IDC. BCR and EFS probabilities were estimated using the Kaplan-Meier method. Prognostic performance was evaluated using log-rank tests and Harrell’s concordance index. 612 patients with mean age 63.1 years were included with mean follow up of 5.3 (range 0–10.8) years. CC/IDC was noted in 159/612 (26%) biopsies. There were 101 (17%) BCR and 36 (6%) events. CAPRA discriminated three distinct risk categories for BCR (p < 0.001) while only high risk separated significantly for EFS (p < 0.001). NCCN distinguished two prognostic groups for BCR (p < 0.0001) and three for EFS (p < 0.0001). Addition of CC/IDC to CAPRA impacted scores 3–5 for BCR and scores 3–5 and 6–10 for EFS and improved the overall concordance index (BCR: 0.66 vs. 0.71; EFS: 0.74 vs. 0.80). Addition of CC/IDC to NCCN impacted scores 4 and 5–6 and also improved the concordance index for BCR (0.62 vs. 0.68). Regarding EFS, NCCN scores 4 and 5–6 demonstrated markedly different outcomes with the addition of CC/IDC. The CAPRA nomogram allows better outcome stratification than NCCN. Addition of CC/IDC status particularly improves patient stratification for CAPRA scores 3–5, 6–10, and for NCCN scores 4 and 5–6.
Similar content being viewed by others
Introduction
Prostate cancer is the most commonly diagnosed malignancy in men in North America and accounts for the second most cancer-related deaths1. Treatment plans for newly diagnosed patients involve risk stratification based on biopsy and clinical information to assess the likelihood of local recurrence and distant metastases. Other important factors include comorbidities, functional status, life expectancy and patient preference. There are a variety of prostate cancer-related risk assessment tools to facilitate clinical decision making and patient management. Some of the more well-known methods include the D’Amico classification2 and a few others which are derived from this classification3,4,5. In addition, numerous nomograms are in use to predict disease progression and/or recurrence6,7.
The National Comprehensive Cancer Network (NCCN) is one such commonly utilized pre-treatment risk stratification tool. It uses clinicopathologic variables such as PSA level at the time of diagnosis (ng/ml), Gleason Grade of the biopsy, clinical stage, number of positive core biopsies, and percentage of cancer in each positive cores to stratify patients into 1–2 “very low/low”, 3 “favorable intermediate”, 4 “unfavorable intermediate” and 5–6 “high and very high” risk groups8. Each risk group has its own therapeutic recommendations, which may include active surveillance, radical prostatectomy, and/or radiotherapy and chemotherapy.
The Cancer of Prostate Risk Assessment (CAPRA) score was developed at the University of California San Francisco with the aims of combining the accuracy of nomograms with the practicality and feasibility of the D’Amico method9. It takes into consideration similar clinical and pathological variables as NCCN tool, including age, PSA level at the time of diagnosis (ng/ml), clinical stage (T-stage), Gleason score of the biopsy (both primary and secondary patterns), and the percentage of biopsy cores positive for adenocarcinoma10. It has been widely utilized in predicting prostate cancer metastasis and mortality. Given the natural history of prostate cancer and the toxicities and potential side effects associated with various treatment modalities, it is crucial to use risk stratification tools to choose optimal patient management plans.
Certain pathologic features such as the Gleason score are well established for their prognostic impact. More recently, other morphologic features such as cribriform pattern 4 carcinoma (CC) and intraductal carcinoma (IDC) proved to be independently associated with worse prognosis and nodal metastases11,12,13,14,15,16,17,18,19,20. Despite being strong prognostic indicators on patient outcome, CC and IDC are not specifically included as part of the clinicopathological variables utilized in the aforementioned risk stratification methods. Thus, in the present study, our aim was to determine whether combining the presence or absence of CC and IDC on biopsies with CAPRA or NCCN risk stratification tools would improve their prognostic impact on patient outcome.
Materials and methods
Case selection and review
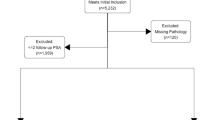
The present study was approved by the research ethics boards of Sunnybrook Health Sciences Centre and the University Health Network (REB 395–2017 and CAPCR 17–5727). A retrospective search was conducted in the laboratory information systems (Sunquest and Cerner CoPath) at both institutions. Consecutive, treatment naïve patients with matched in-house prostate biopsies positive for prostatic adenocarcinoma and subsequent prostatectomy specimens from 2010 to 2017 were included. All relevant clinicopathological information, including PSA, cT stage, global biopsy Gleason score/Grade group, maximum percentage carcinoma, total number of cores, and number of cores positive for carcinoma was retrieved from both electronic patient records and the laboratory information systems. The percentage of biopsy cores positive for adenocarcinoma was then determined by dividing the number of cores positive for adenocarcinoma by the total number of cores present.
The haematoxylin and eosin (H&E)-stained slides were retrieved and reviewed by two genitourinary pathologists (M.R.D. and T.v.D.K.) for presence of CC/IDC as defined in previous publications21,22 and incorporating the recent ISUP definition of CC23: contiguous malignant epithelial cells with multiple glandular lumina visible at 10× with no intervening stroma or mucin which incorporated small and large cribriform glands. Most IDC show a cribriform architectural pattern and its prognostic impact is similar as cribriform pattern 4 carcinoma21. For this work, CC and IDC were combined given the recent consensus recommendations from the International Society of Urological Pathology24. The following information was retrieved from the pathology reports in CoPath: global Gleason score, total number of cores present, and the number of cores positive for adenocarcinoma. The CAPRA score was tabulated using patient age, PSA at biopsy (ng/ml), global Gleason score of the biopsy, percentage of biopsy cores positive for adenocarcinoma, and clinical stage (T-stage). The NCCN risk stratification system was calculated using similar clinicopathological variables: PSA at biopsy (ng/ml), global Gleason score of the biopsy, percentage of biopsy cores positive for adenocarcinoma, number of cores with GG 4 and 5, and clinical stage (T-stage).
Clinical follow-up
The outcome variables were biochemical recurrence (BCR) and metastases and/or deaths of prostate cancer (event-free survival or EFS). Patient follow-up information was retrieved from the laboratory information system. BCR is defined as two consecutive PSA levels >0.2 ng/ml at follow up. Time to BCR was defined as the number of years between radical prostatectomy and BCR. Patients with no BCR were censored at the date of last follow up. For events, time to metastasis (nodal and non-nodal) and death is defined as the number of years between biopsy diagnosis and metastasis or death. For patients with lymph node metastasis, this was defined as the time lapse between the biopsy diagnosis date and radical prostatectomy date. Those patients with no reports of metastasis or death were censored at the time of last follow up.
Statistical analysis
Statistical analyses were performed using R version 4.0.0 (R Core Team, 2020). BCR and EFS probabilities were estimated using the Kaplan-Meier method. Differences in survival curves between strata were assessed using the log-rank test. Cox proportional hazards models were fit, and prognostic performance was evaluated using Harrell’s concordance index, adjusted for optimism using 1000 bootstrap resamples. Additionally, ANOVA tests comparing the log-likelihood statistic of models with and without CC or IDC were conducted, with larger log-likelihood statistics indicating a better model fit. A p value of less than 0.05 was considered statistically significant.
Results
Clinicopathological features
The clinicopathological features of the study cohort from each institution are listed in Table 1. Six hundred and twelve treatment naïve cases were identified (n = 612). Median age was 64 (range 41–79) and median PSA at diagnosis was 6.7 (range 0.5–97.0). CC and/or IDC was noted in 159 (26%) needle biopsies (CC = 103, IDC = 32 and both in 24 cases based on H&E and immunostains when available- Fig. 1). For CAPRA scores, 132 patients were “low risk” 0–2, 388 patients were “intermediate risk” 3–5 and 92 patients were “high risk” 6–10. In terms of NCCN score, 70 patients were “very low/low risk” 1–2, 214 patients were “favorable intermediate risk” 3, 235 “unfavorable intermediate risk” 4, 93 patients were “high/very high risk” 5–6. Median follow-up was 4.4 years and BCR occurred in 101 (17%) patients. In terms of prostatectomy stage, 360 patients were pT2, 161 patients were pT3a, and 90 patients were pT3b at prostatectomy.
Comparison of CAPRA and NCCN on outcome stratification
Figure 2 shows overall Kaplan-Meier curves for recurrence-free survival. CAPRA discriminated three distinct risk categories for BCR (p < 0.001) while only high risk separated significantly for EFS (p < 0.001). NCCN distinguished two prognostic groups for BCR (p < 0.0001) and three for EFS (p < 0.0001). When stratified by CAPRA score, 6 BCR are present in patients with “low risk” 0–2, 59 events in patients with “intermediate risk” 3–5 and 36 events in patients with “high risk” 6–10 (1B). The optimism adjusted c-indices for all models are reported in Table 2; the c index for this model is 0.663. Figure 2C shows stratification by NCCN score with 6 BCR in patients with “very low/low risk” 1–2, 19 occurrences in patients with “favorable intermediate risk” 3, 52 BCR in those with “unfavorable intermediate risk” 4, and 24 BCR in patients with “high/very high risk” 5–6. The optimism-corrected c-index is 0.612.
For (non-BCR) events (metastases and deaths of prostate cancer), there were 64 events in the patient cohort. Figure 2D shows the event-free survival. When stratified by CAPRA score, event-free survival differed significantly between the “low” 0–2, “intermediate” 3–5 and “high risk” 6–10 groups with a log-rank p value < 0.001 and an optimism-corrected c-index of 0.736 (Fig. 2E). Figure 2F shows the event-free survival data stratified by NCCN scores. There were no events (metastases or death) in the “very low/low risk” 0–2 groups, 7 events in the “favorable intermediate” 3 group, 27 events in the “unfavorable intermediate” groups 4, and 30 events in the “high/very high risk” groups 5–6. The optimism-corrected c-index is 0.762.
The Impact of IDC/CC on prognostication
The impact of addition of CC/IDC to CAPRA is shown in Fig. 3 and Supplementary Table 1. The addition of CC/IDC particularly improves the prognostication of the “intermediate” 3–5 group. The adjusted c-index is 0.700, suggesting that the addition of CC/IDC to CAPRA helps improve model performance (ANOVA p value = 0.0038). In terms of NCCN, addition of CC/IDC improves the prognostication for each group for BCR, with the most significant effect seen in groups 4 and 5–6 (Fig. 3B; ANOVA p value = 0.008).
For event-free survival (metastases or death), the addition of CC/IDC significantly influenced the outcome in patients in CAPRA 3–5 and 6–10 groups (Fig. 3C), improving the c-index to 0.790 (ANOVA p value = 0.038). For instance, CAPRA score 3–5 (intermediate risk) patients with CC/IDC in their biopsy had similar outcomes to those with CAPRA score 6–10 (high risk). The addition of CC/IDC to the NCCN score also changed the outcome significantly for patients in groups 4 and 5–6 (Fig. 3D; ANOVA p value < 0.001), though the c-index could not be estimated due to the small number of events in several groups).
Discussion
Pathologic features such as the Gleason score and the 2014 modified International Society of Urological Pathology (ISUP) Grade Groups are well-known predictors of oncologic outcome in prostate cancer patients24,25,26,27. Gleason 4 pattern is composed of cribriform, glomeruloid, poorly formed and fused glands. However, distinct morphologic patterns within Gleason 4 pattern differ with regard to prognostic impact28,29. In particular, the additional prognostic importance of cribriform architecture adenocarcinoma such as CC and IDC in prostatic cancer patients has been previously demonstrated11,12,13,14. A recent publication by our group has shown that either CC or IDC morphologies were present in 95% of prostatectomy specimens that were positive for nodal metastases(11) and these histomorphologies were strong independent predictors(14). Despite their strong prognostic value, the presence of these histomorphologies have yet to be incorporated in any clinical risk stratification tools such as the CAPRA score or the NCCN risk stratification tool. Many studies have looked at whether the addition of histomorphologic features or biomarkers (such as PTEN) can improve the prognostic impact of clinical assessment tools30,31. One recent study by Jeyapala et al. demonstrated that combining CC/IDC with the CAPRA-S score improves the prognostication of BCR after radical prostatectomy30. Another study by Salles et al. showed the addition of CC/IDC to a model using CAPRA preoperative variables only marginally increased the area under the curve with non-organ confined prostate cancer at prostatectomy as outcome parameter32. They also postulated that cribriform histomorphologic features may be associated with MYC and PTEN genomic alterations. To further characterize the prognostic impact of CC/IDC, the present study examined whether the inclusion of these features improves risk stratification of CAPRA and NCCN at biopsy diagnosis.
In the present study, we found that the CAPRA score performed better than the NCCN system in risk stratifying prostatic cancer patients for time to BCR. This is concordant with the results of another study by Zelic et al. in which they compared the prognostic performance of several commonly utilized prostate cancer risk stratification tools33. They discovered that the MSKCC nomograms and CAPRA score outperformed the D’Amico and D’Amico-derived tools (including NCCN) at stratifying prostate cancer deaths. The CAPRA score may also be preferable due to its feasibility and practicality compared to the other risk assessment tools10,34.
We also discovered that the addition of CC/IDC improves the prognostic value for both CAPRA and NCCN for time to BCR and EFS, particularly for the high risk/very high risk groups in both nomograms. Our results are in keeping with two other studies by Kweldam et al. in which CC/IDC was prognostic for BCR as well as cancer specific mortality, including patients with high Gleason scores19,35. Our findings highlight the important role of cribriform architecture adenocarcinoma in choosing the optimal management plan for intermediate risk patients, especially when options may include active surveillance and nodal dissection. Our findings also underscore the importance for pathologists to report these parameters, particularly for GG4 and 5 biopsy cases.
Some of the limitations of the present study include the retrospective design and the inclusion of patients from only two academic institutions. An inherent selection bias may exist as only patients who underwent both biopsy and prostatectomy at our institutions (both academic cancer centers) were included. Particularly, most men with GG1 prostate cancer would be offered active surveillance rather than prostatectomy, resulting in a study population skewed for intermediate risk prostate cancer. Furthermore, the prognostic effect of CC/IDC was only studied in conjunction with the CAPRA and NCCN risk assessment tools. It is unclear whether the presence or absence of CC/IDC features alters the prognostic impact of other risk prognostic tools such as the D’Amico method and other nomograms. It is also worth noting that variations of the CAPRA score exists and they are utilized in different clinical settings and patient populations. For example, the CAPRA-S score is utilized in post-radical prostatectomy patients. In patients who have received primary androgen deprivation therapy for more advanced disease, the Japan CAPRA or J-CAPRA score can be used10. Since these tools utilize different clinicopathological variables, future studies can be conducted to determine how the incorporation of CC/IDC alters their prognostic impact.
In conclusion, we have demonstrated that the CAPRA risk score allows better outcome stratification than NCCN. Addition of CC/IDC status particularly improves patient stratification for CAPRA scores 3–5, 6–10 and NCCN score 4 and 5–6; further confirming their prognostic value and the importance of including these histomorphologic features in pathology reports. Finally, since various risk stratification tools utilize different clinicopathologic variables, it remains to be further elucidated whether the inclusion of CC/IDC on biopsy can improve their prognostic significance.
Data availability
The datasets used in the current study are available from the authors upon reasonable request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin 70, 7-30 (2020)
Thompson I, Thrasher JB, Aus G, Burnett AL, Canby-Hagino ED, Cookson MS, et al. Guideline for the management of clinically localized prostate cancer: 2007 update. J Urol 177, 2106-31 (2007)
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol 71, 618-29 (2017)
Graham J, Kirkbride P, Cann K, Hasler E, Prettyjohns M. Prostate cancer: summary of updated NICE guidance. BMJ 348, 7524 (2014)
Lukka H, Warde P, Pickles T, Morton G, Brundage M, Souhami L, et al. Controversies in prostate cancer radiotherapy: consensus development. Can J Urol 8, 1314-22 (2001)
Cooperberg MR. Prostate cancer risk assessment: choosing the sharpest tool in the shed. Cancer 113, 3062-3066 (2008)
Caulfield S, Menezes G, Marignol L, Poole C. Nomograms are key decision-making tools in prostate cancer radiation therapy. Urol Oncol 36, 283-292 (2018)
Mohler JL, Armstrong AJ, Bahnson RR, D’Amico AV, Davis BJ, Eastham JA, et al. Prostate Cancer, Version 1.2016. J Natl Compr Canc Netw 14, 19-30 (2016)
Cooperberg MR, Hilton JF, Carroll PR. The CAPRA-S score: A straightforward tool for improved prediction of outcomes after radical prostatectomy. Cancer 117, 5039-5046 (2011)
Brajtbord JS, Leapman MS, Cooperberg MR. The CAPRA Score at 10 Years: Contemporary Perspectives and Analysis of Supporting Studies. Eur Urol 71, 705-709 (2017)
Downes MR, Xu B, van der Kwast TH. Gleason grade patterns in nodal metastasis and corresponding prostatectomy specimens: impact on patient outcome. Histopathology 75, 715-722 (2019)
Montironi R, Zhou M, Magi-Galluzzi C, Epstein JI. Features and Prognostic Significance of Intraductal Carcinoma of the Prostate. Eur Urol Oncol 1, 21-28 (2018)
Samaratunga H, Delahunt B, Egevad L, Srigley JR, Billis A, Bostwick DG, et al. Intraductal carcinoma of the prostate is an aggressive form of invasive carcinoma and should be graded. Pathology 52, 192-196 (2020)
Downes MR, Xu B, van der Kwast TH. Cribriform architecture prostatic adenocarcinoma in needle biopsies is a strong independent predictor for lymph node metastases in radical prostatectomy. Eur J Cancer 148, 432-439 (2021)
Guo CC, Epstein JI. Intraductal carcinoma of the prostate on needle biopsy: Histologic features and clinical significance. Mod Pathol 19, 1528-1535 (2006)
Kimura K, Tsuzuki T, Kato M, Saito AM, Sassa N, Ishida R, et al. Prognostic value of intraductal carcinoma of the prostate in radical prostatectomy specimens. Prostate 74, 680-687 (2014)
Ross HM, Kryvenko ON, Cowan JE, Simko JP, Wheeler TM, Epstein JI. Do adenocarcinomas of the prostate with Gleason score (GS) ≤6 have the potential to metastasize to lymph nodes? Am J Surg Pathol 36, 1346-1352 (2012)
Rijstenberg LL, Hansum T, Kweldam CF, Kümmerlin IP, Remmers S, Roobol MJ, et al. Large and small cribriform architecture have similar adverse clinical outcome on prostate cancer biopsies. Histopathology https://doi.org/10.1111/his.14658 (2022)
Kweldam CF, Kümmerlin IP, Nieboer D, Verhoef EI, Steyerberg EW, van der Kwast TH, et al. Disease-specific survival of patients with invasive cribriform and intraductal prostate cancer at diagnostic biopsy. Mod Pathol, 29, 630-636 (2016)
Kweldam CF, Wildhagen MF, Steyerberg EW, Bangma CH, van der Kwast TH, van Leenders GJ. Cribriform growth is highly predictive for postoperative metastasis and disease-specific death in Gleason score 7 prostate cancer. Mod Pathol 28, 457-564 (2015)
Trudel D, Downes MR, Sykes J, Kron KJ, Trachtenberg J, van der Kwast TH. Prognostic impact of intraductal carcinoma and large cribriform carcinoma architecture after prostatectomy in a contemporary cohort. Eur J Cancer 50, 1610-1616 (2014)
Masoomian M, Downes MR, Sweet J, Cheung C, Evans AJ, Fleshner N, et al. Concordance of biopsy and prostatectomy diagnosis of intraductal and cribriform carcinoma in a prospectively collected data set. Histopathology 74, 474-482 (2019)
van der Kwast TH, van Leenders GJ, Berney DM, Delahunt B, Evans AJ, Iczkowski KA, et al. ISUP Consensus Definition of Cribriform Pattern Prostate Cancer. Am J Surg Pathol 45, 1118-1126 (2021)
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA, et al. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am J Surg Pathol 40, 244-252 (2016)
Srigley JR, Delahunt B, Samaratunga H, Billis A, Cheng L, Clouston D, et al. Controversial issues in Gleason and International Society of Urological Pathology (ISUP) prostate cancer grading: proposed recommendations for international implementation. Pathology 51, 463–473 (2019)
Barakzai MA. Prostatic Adenocarcinoma: A Grading from Gleason to the New Grade-Group System: A Historical and Critical Review. Asian Pac J Cancer Prev 20, 661-666 (2019)
Humphrey PA. Histopathology of Prostate Cancer. Cold Spring Harb Perspect Med 7, a030411 (2017)
Montironi R, Cimadamore A, Gasparrini S, Mazzucchelli R, Santoni M, Massari F, et al. Prostate cancer with cribriform morphology: diagnosis, aggressiveness, molecular pathology and possible relationships with intraductal carcinoma. Expert Rev Anticancer Ther 18, 685-693 (2018)
Hassan O, Matoso A. Clinical significance of subtypes of Gleason pattern 4 prostate cancer. Transl Androl Urol 7, S477-S483 (2018)
Jeyapala R, Kamdar S, Olkhov-Mitsel E, Zlotta A, Fleshner N, Visakorpi T, et al. Combining CAPRA-S with Tumor IDC/C Features Improves the Prognostication of Biochemical Recurrence in Prostate Cancer Patients. Clin Genitourin Cancer https://doi.org/10.1016/j.clgc.2022.01.003 (2022)
Jamaspishvili T, Patel PG, Niu Y, Vidotto T, Caven I, Livergant R, et al. Risk stratification of prostate cancer through quantitative assessment of PTEN Loss (qPTEN). J Natl Cancer Inst 112, 1098–1104 (2020)
Salles DC, Vidotto T, Faisal FA, Tosoian JJ, Guedes LB, Muranyi A, et al. Assessment of MYC/PTEN status by gene-protein assay in grade group 2 prostate biopsies. J Mol Diagn 23, 1030–1041 (2021)
Zelic R, Garmo H, Zugna D, Stattin P, Richiardi L, Akre O, et al. Predicting prostate cancer death with different pretreatment risk stratification tools: a head-to-head comparison in a nationwide cohort study. Eur Urol 77, 180–188 (2020)
Cooperberg MR, Pasta DJ, Elkin EP, Litwin MS, Latini DM, Du Chane J, et al. The University of California, San Francisco Cancer of the Prostate Risk Assessment score: a straightforward and reliable preoperative predictor of disease recurrence after radical prostatectomy. J Urol 173, 1938-1942 (2005)
Kweldam CF, Kümmerlin IP, Nieboer D, Steyerberg EW, Bangma CH, Incrocci L, et al. Presence of invasive cribriform or intraductal growth at biopsy outperforms percentage grade 4 in predicting outcome of Gleason score 3 + 4 = 7 prostate cancer. Mod Pathol 30, 1126-1123 (2017)
Acknowledgements
This study was presented, in part, at the 2022 United States and Canadian Academy of Pathology meeting on March 22.
Funding
This work was supported, in part, by Prostate Cancer Canada grant to T.v.D.K. (Movember Discovery grant D2017-1879).
Author information
Authors and Affiliations
Contributions
Y.Y. collected, analyzed data, and drafted the manuscript. M.R.D. and T.v.D.K designed the study, collected and analyzed data, and revised the manuscript. A.F. and N.F. drafted and revised the manuscript. K.L. contributed with statistical analysis. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The present study was approved by the research ethics boards of Sunnybrook Health Sciences Centre and the University Health Network (REB 395-2017 and CAPCR 17-5727).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yu, Y., Lajkosz, K., Finelli, A. et al. Impact of cribriform pattern 4 and intraductal prostatic carcinoma on National Comprehensive Cancer Network (NCCN) and Cancer of Prostate Risk Assessment (CAPRA) patient stratification. Mod Pathol 35, 1695–1701 (2022). https://doi.org/10.1038/s41379-022-01111-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-022-01111-w