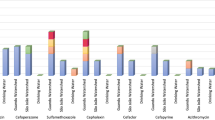
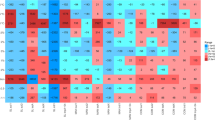
Abstract
This study was conducted to evaluate the abundance of antibiotic resistant bacteria and their resistance genes from agriculture soil supplemented with pig manure. Uncultivable soil sample was supplemented with pig manure samples under microcosm experimental conditions and plated on Luria-Bertani (LB) agar incorporated with commercial antibiotics. The supplementation of soil with 15% pig manure resulted in the highest increase in the population of antibiotic resistant bacteria (ARB)/multiple antibiotic resistant bacteria (MARB). Seven genera that included Pseudomonas, Escherichia, Providencia, Salmonella, Bacillus, Alcaligenes and Paenalcaligenes were the cultivable ARB identified. A total of ten antibiotic resistant bacteria genes (ARGs) frequently used in clinical or veterinary settings and two mobile genetic elements (MGEs) (Class 1 and Class 2 integrons) were detected. Eight heavy metal, copper, cadmium, chromium, manganese, lead, zinc, iron, and cobalt were found in all of the manure samples at different concentrations. Tetracycline resistance genes were widely distributed with prevalence of 50%, while aminoglycoside and quinolone-resistance gene had 16% and 13%, respectively. Eighteen ARB isolates carried more than two ARGs in their genome. Class 1 integron was detected among all the 18 ARB with prevalence of 90–100%, while Class 2 integron was detected among 11 ARB. The two classes of integron were found among 10 ARB. Undoubtedly, pig manure collected from farms in Akure metropolis are rich in ARB and their abundance might play a vital role in the dissemination of resistance genes among clinically-relevant pathogens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Udikovic-Kolic N, Wichmann F, Broderick NA, Handelsman J. Bloom of resident antibiotic-resistant bacteria in soil following manure fertilization. Proc Natl Acad Sci. 2014;111:15202–207.
Marti R, Scott A, Tien YC, Murray R, Sabourin L, Zhang Y, et al. Impact of manure fertilization on the abundance of antibiotic-resistant bacteria and frequency of detection of antibiotic resistance genes in soil and on vegetables at harvest. Appl Environ Microbiol. 2013;79:5701–709.
Zhu YG, Johnson TA, Su JQ, Qiao M, Guo GX, Stedtfeld RD, et al. Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proc Natl Acad Sci. 2013;110:3435–440.
Yang Q, Wang R, Ren S, Szoboszlay M, Moe LA. Practical survey on antibiotic-resistant bacterial communities in livestock manure and manure-amended soil. J Environ Sci Health B 2016;51:14–23.
Liu Y, Zheng L, Cai Q, Xu Y, Xie Z, Liu J, et al. Simultaneous reduction of antibiotics and antibiotic resistance genes in pig manure using a composting process with a novel microbial agent. Ecotoxicol Environ Saf. 2021;208:111724.
Wang FH, Qiao M, Chen Z, Su JQ, Zhu YG. Antibiotic resistance genes in manure-amended soil and vegetables at harvest. J Hazard Mater 2015;299:215221.
Sommer MO. Barriers to the spread of resistance. Nature. 2014;509:567–68.
Humphries RM, Ambler J, Mitchell SL, Castanheira M, Dingle T, Hindler JA, et al. CLSI methods development and standardization working group best practices for evaluation of antimicrobial susceptibility tests. J Clin Microbiol. 2018;56:e01934–17.
Chen J, Li J, Zhang H, Shi W, Liu Y. Bacterial heavy-metal and antibiotic resistance genes in a copper tailing dam area in northern China. Front Microbiol. 2019;10:1916.
APHA. Standards Methods for the Examination of Water and Wastewater. 20th edition. Washington, D.C: American Public Health Association; 1998.
Allen SE, Grimshaw HM, Parkinson JA, Quarmby C. Chemical analysis of ecological materials. Oxford: Blackwell Scientific Publications; 1974.
Yang Q, Tian T, Niu T, Wang P. Molecular characterization of antibiotic resistance in cultivable multidrug-resistant bacteria from livestock manure. Environ Pollut. 2017;229:188–98.
Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH, Morgan DR. Manual of Clinical Microbiology (6th edn). Trends Microbiol. 1995;3:449–49.
Lane DJ. 16S/23S rRNA sequencing. In Stackebrandt E, Goodfellow M, editors. Nucleic acid techniques in bacterial systematics. New York: John Wiley & Sons; 1991, p. 115–75.
Binh CT, Heuer H, Kaupenjohann M, Smalla K. Diverse aadA gene cassettes on class 1 integrons introduced into soil via spread manure. Res Microbiol. 2009;160:427–33.
Heuer H, Schmitt H, Smalla K. Antibiotic resistance gene spread due to manure application on agricultural fields. Curr Opin Microbiol. 2011;14:236–43.
Pruden A, Larsson DJ, Amézquita A, Collignon P, Brandt KK, Graham DW, et al. Management options for reducing the release of antibiotics and antibiotic resistance genes to the environment. Environ Health Perspect. 2013;121:878–85.
Marti R, Tien YC, Murray R, Scott A, Sabourin L, Topp E. Safely coupling livestock and crop production systems: how rapidly do antibiotic resistance genes dissipate in soil following a commercial application of swine or dairy manure? Appl Environ Microbiol. 2014;80:3258–265.
Stockwell VO, Duffy B. Use of antibiotics in plant agriculture. Rev Scientifique Et Tech-Off Int Des Epizooties. 2012;31:199–210.
Chaudhry V, Rehman A, Mishra A, Chauhan PS, Nautiyal CS. Changes in bacterial community structure of agricultural land due to long-term organic and chemical amendments. Microb Ecol. 2012;64:450–60.
Sun HY, Deng SP, Raun WR. Bacterial community structure and diversity in a century-old manure-treated agroecosystem. Appl Environ Microbiol. 2004;70:5868–874.
Cheng W, Chen H, Su C, Yan S. Abundance and persistence of antibiotic resistance genes in livestock farms: A comprehensive investigation in eastern China. Enviro Int. 2013;61:1–7.
Wei B, Yu J, Cao Z, Meng M, Yang L, Chen Q. The availability and accumulation of heavy metals in greenhouse soils associated with intensive fertilizer application. Int J Environ Res Public Health. 2020;17:5359.
Lima T, Domingues S, Da Silva GJ. Manure as a potential hotspot for antibiotic resistance dissemination by horizontal gene transfer events. Vet Sci. 2020;7:110.
Stefanowicz AM, Kapusta P, Zubek S, Stanek M, Woch MW. Soil organic matter prevails over heavy metal pollution and vegetation as a factor shaping soil microbial communities at historical Zn-Pb mining sites. Chemosphere 2020;240:124922.
Rogers SD, Bhave MR, Mercer JF, Camakaris J, Lee BT. Cloning and characterization of cutE, a gene involved in copper transport in Escherichia coli. J Bact. 1991;173:6742–748.
Hemme CL, Deng Y, Gentry TJ, Fields MW, Wu L, Barua S, et al. Metagenomic insights into evolution of a heavy metal-contaminated groundwater microbial community. ISME J. 2010;4:660–72.
Azarbad H, Niklińska M, Laskowski R, van Straalen NM, van Gestel CA, Zhou J, et al. Microbial community composition and functions are resilient to metal pollution along two forest soil gradients. FEMS Microbiol Ecol. 2015;91:1–11.
Lal S, Ratna S, Said OB, Kumar R. Biosurfactant and exopolysaccharide-assisted rhizobacterial technique for the remediation of heavy metal contaminated soil: an advancement in metal phytoremediation technology. Environ Technol Innov. 2018;10:243–63.
Duan M, Zhang Y, Zhou B, Wang Q, Gu J, Liu G, et al. Changes in antibiotic resistance genes and mobile genetic elements during cattle manure composting after inoculation with Bacillus subtilis. Bioresour Technol. 2019;292:122011.
Peng S, Li H, Song D, Lin X, Wang Y. Influence of zeolite and superphosphate as additives on antibiotic resistance genes and bacterial communities during factory-scale chicken manure composting. Bioresour Technol. 2018;263:393–401.
Zhao X, Wei Y, Fan Y, Zhang F, Tan W, He X, et al. Roles of bacterial community in the transformation of dissolved organic matter for the stability and safety of material during sludge composting. Bioresour Technol. 2018;267:378–85.
Zhu L, Zhao Y, Zhang W, Zhou H, Chen X, Li Y, et al. Roles of bacterial community in the transformation of organic nitrogen toward enhanced bioavailability during composting with different wastes. Bioresour Technol. 2019;285:121326.
Zhang J, Lu T, Wang Z, Wang Y, Zhong H, Shen P, et al. Effects of magnetite on anaerobic digestion of swine manure: Attention to methane production and fate of antibiotic resistance genes. Bioresour Technol. 2019;291:121847.
Chen M, Xu J, Dai R, Wu Z, Liu M, Wang Z. Development of a moving-bed electrochemical membrane bioreactor to enhance removal of low-concentration antibiotic from wastewater. Bioresour Technol. 2019;293:122022.
Bahar G, Eraç BA, Mert A, Gülay Z. PER-1 production in a urinary isolate of Providencia rettgeri. J Chemother. 2004;16:343–46.
Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. CIM. 2012;18:268–81.
Bhavsar S, Krilov L. Escherichia coli Infections. Pediatr Rev. 2015;36:167–71.
Wu N, Qiao M, Zhang B, Cheng WD, Zhu YG. Abundance and diversity of tetracycline resistance genes in soils adjacent to representative swine feedlots in China. Environ Sci Technol. 2010;44:6933–939.
Hu T, Wang X, Zhen L, Gu J, Zhang K, Wang Q, et al. Effects of inoculation with lignocellulose-degrading microorganisms on antibiotic resistance genes and the bacterial community during co-composting of swine manure with spent mushroom substrate. Environ Pollut. 2019;252:110–8.
Roberts MC. Mechanisms of bacterial antibiotic resistance and lessons learned from environmental tetracycline-resistant bacteria. Antimicrobial Res Environ. 2011;252:93–121.
Zhang M, He LY, Liu YS, Zhao JL, Liu WR, Zhang JN, et al. Fate of veterinary antibiotics during animal manure composting. Sci Total Environ. 2019;650:1363–70.
Byrne-Bailey KG, Gaze WH, Zhang L, Kay P, Boxall A, Hawkey PM, et al. Integron prevalence and diversity in manured soil. Appl Environ Microbiol. 2011;77:684–87.
Mathew AG, Liamthong S, Lin J, Hong Y. Evidence of class 1 integron transfer between Escherichia coli and Salmonella spp. on livestock farms. Foodborne Pathog Dis. 2009;6:959–64.
Tian Z, Zhang Y, Yu B, Yang M. Changes of resistome, mobilome and potential hosts of antibiotic resistance genes during the transformation of anaerobic digestion from mesophilic to thermophilic. Water Res. 2016;98:261–69.
Stokes HW, Gillings MR. Gene flow, mobile genetic elements and the recruitment of antibiotic resistance genes into Gram-negative pathogens. FEMS Microbiol Rev. 2011;35:790–819.
Acknowledgements
The authors sincerely acknowledge provision of laboratory space and equipment, and technical support by the technical arms of the Department of Medical Microbiology and Parasitology, Obafemi Awolowo University, Ile-Ife, Nigeria.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41429_2023_633_MOESM1_ESM.docx
Supplementary Materials: Impact of soil amended with pig manure on the abundance of antibiotic resistant bacteria and their associated genes
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akinduro, A., Onyekwelu, C.I., Oyelumade, T. et al. Impact of soil supplemented with pig manure on the abundance of antibiotic resistant bacteria and their associated genes. J Antibiot 76, 548–562 (2023). https://doi.org/10.1038/s41429-023-00633-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-023-00633-y