Abstract
Background/objectives
To assess the effect of ectopic inner foveal layers (EIFL) based staging scheme, foveal avascular zone (FAZ) alterations and other microstructural optical coherence tomography (OCT) findings on visual function for patients undergoing idiopathic epiretinal membrane (iERM) surgery.
Subjects/methods
In this retrospective study, patients who underwent 27 G pars plana vitrectomy (PPV) for idiopathic ERM with a minimum follow-up of 12 months were included. Preoperative and postoperative OCT scans, FAZ area measurements on en face OCT angiography images and mean retinal sensitivity (MRS) using microperimetry were recorded in all cases. The correlation of FAZ area, EIFL and other OCT parameters with preoperative and postoperative best-corrected visual acuity (BCVA) was analysed.
Results
In all, 112 eyes of 112 patients were included. Visual acuity improvement was statistically significant in all four stages; however, differences between Stages 2, 3 and 4 ERMs remained significant (p < 0.05). The presence and thickness of the EIFL was associated with worse baseline (p = 0.013; p = 0.005, respectively) and final (p < 0.001 for both) BCVA. The presence of cystoid macular oedema was associated with worse BCVA at baseline (p = 0.027) and postoperative month-6 (p = 0.04). The mean FAZ area was significantly reduced in all stages of ERM compared with the fellow eyes (p < 0.05 for all). Postoperative retinal sensitivity improvement was statistically significant in Stage 1 and Stage 2.
Conclusion
The presence of EIFL is an independent predictor of worse postoperative BCVA. Accordingly, despite significant BCVA improvements in all stages of ERM, visual acuity gain remains limited in eyes with Stage 3 and Stage 4 ERM.
Similar content being viewed by others
Introduction
Epiretinal membrane (ERM), which was first described by Iwanoff in 1865, is a common retinal condition characterised by proliferation of a contractile fibrocellular tissue over the internal limiting membrane (ILM) [1,2,3,4,5]. The clinical presentation of ERMs can range from completely asymptomatic to profoundly symptomatic with metamorphopsia, micropsia or macropsia and decreased visual acuity, depending on the localisation (epimacular or extrafoveal) and severity of retinal microstructural damage caused by the membrane [6, 7]. Pars plana vitrectomy (PPV) with membranectomy is the standard treatment in cases of symptomatic ERM. However, improved anatomic success rates do not often correlate well with visual results [8, 9]. Several studies are conducted to identify reliable prognostic biomarkers including the status of ellipsoid zone (EZ) and outer photoreceptor segments, ganglion cell-inner plexiform layer and choroidal thickness [10,11,12,13]. Recently, Govetto et al. suggested a novel optical coherence tomography (OCT)-based staging scheme of ERMs assessing the presence of ectopic inner foveal layers (EIFL). However, there is still limited study evaluating the clinical significance of this novel staging system in the literature. In the current study, we investigated the significance of this new OCT-guided staging system in addition to microstructural OCT findings on postoperative anatomical and functional outcomes. The aim of our study was to answer the following two questions: what is the best timing of surgery in patients with ERM? and what are the reliable prognostic biomarkers that may predict postoperative visual outcome?
Methods
Retrospective, observational and consecutive chart review of patients diagnosed with ERM and seen by 2 retina 23 specialists (AMY and SY) between 1 January 2013 and 1 January 2017 at Bursa Retina Eye Hospital was performed. Each patient was informed about the risks and benefits of the surgery, and written informed consent was obtained. This study adhered to the tenets of the Declaration of Helsinki.
Patient eligibility
Inclusion criteria: patients with idiopathic ERM treated with PPV, ERM and ILM peeling, with a minimum follow-up of 12 months were included.
Exclusion criteria: patients with a history of trauma or previous ocular surgery other than phacoemulsification, intermediate, or advanced age-related macular degeneration, history of choroidal neovascularization of any aetiology or central serous chorioretinopathy, diabetic retinopathy of any stages, macular teleangectasias, macular hole, retinal vascular occlusions of any kind, glaucoma and other causes of optic neuropathy were excluded.
Ocular parameters and OCT imaging
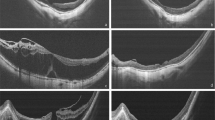
The patients underwent complete ophthalmic examinations, including best-corrected visual acuity (BCVA), intraocular pressure assessment, slit-lamp biomicroscopy, fundus examination, and spectral domain optical coherence tomography (SD-OCT) imaging at baseline and at 6 and 12 months after surgery. Best-corrected 43 visual acuity was recorded at each visit, reported in Snellen fraction, and then converted into logarithm of the 44 minimal angle of resolution (logMAR) values for statistical analysis. In all cases, SD-OCT images were obtained with the Spectralis OCT with eye-tracking dual-beam technology (Heidelberg Engineering GmbH, Heidelberg, Germany) and analysed with the Heidelberg Eye Explorer (version 3.1.0.55) using the HRA/Spectralis Viewing Module (version 6.12.4.0). At each visit, all eyes were imaged with 24-line radial scan of Spectralis which is composed of 24 radial B-scans (5.5 mm in length) over an area of 30 degrees centred at the fovea with 7.5 degrees interscan spacing for qualitative and quantitative assessment. We classified the eyes with ERM according to the OCT-based staging system devised by Govetto et al. [14] (Fig. 1).
The ectopic inner foveal layers were defined as the presence of continuous hyporeflective and hyperreflective bands extending from the inner nuclear layer and inner plexiform layer across the fovea (white arrows) The thickness of the outer nuclear layer is measured as the distance from the external limiting membrane to the internal limiting membrane. However, the upper limit of the outer nuclear layer is defined as the inferior border of the ectopic inner foveal layers in eyes with stage 3 and stage 4 epiretinal membrane (A). The foveal avascular zone (FAZ) assessment on superficial capillary plexus (SCP) en face images of optical coherence tomography angiography. The mean FAZ area was significantly reduced in all stages of ERM compared with the fellow eyes (p < 0.05 for all) and FAZ was nearly absent in eyes with Stage 4 ERM (B). Preoperative (left) and postoperative (right) microperimetric assessment (MAIA) of eyes with Stage 1, Stage 2, Stage 3 and Stage 4 ERM (C).
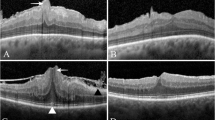
We analysed the microstructural SD-OCT findings of the macula, including the presence of microcystoid retinal changes and the cotton ball sign. The disruption diameter of the EZ and thickness measurements of the outer nuclear layer (ONL) and EIFL was obtained manually with the calliper function of the Spectralis. The central foveal thickness (CFT) was measured with the automated thickness function of the Heidelberg Eye Explorer. The discontinuity of the ellipsoid band in the foveal region was defined as disruption of the EZ. The presence of hyporeflective intraretinal cystoid spaces in the setting of ERMs was considered as cystoid macular oedema (CMO) and the presence of the cotton ball sign was defined as roundish or diffuse highly reflective region located between the EZ and the cone outer segment tip line at the centre of the fovea (Fig. 2) [15]. The thickness of EIFL was measured from the inner border of the ONL to the ILM at the subfoveal area. The ONL thickness was measured from the inner border of the retinal pigment epithelium to the border of the ONL. Two experienced retina specialist masked to the clinical details of the participants assessed the OCT images independently. A third masked retina specialist adjudicated discrepancies.
OCTA imaging and foveal avazcular zone measurement
OCTA images of both eyes of all patients were obtained by Spectralis HRA + OCTA (Heidelberg Engineering, Heidelberg, Germany) at baseline. An OCTA scan pattern of 15 × 5 degrees (44 × 1.5 mm; consisting of 131 B-scans separated by 11 µm) centred on the fovea was acquired. The foveal avascular zone (FAZ) was measured manually using the calliper of the OCT in all cases at en face images of the SCP (Fig. 1).
Microperimetric assessment
Mean retinal sensitivity (MRS) expressed in dB, was assessed using MP (MAIA, Centervue SpA, Padova, Italy) following 20 minutes adaptation in mesopic conditions (light levels below 1 lx) preoperatively and at postoperative month-12. Microperimetry was done with a Goldmann size III target presented against a background of 1.27 cd/m using a 4–2 threshold strategy on a 37-point grid centred on fovea. The average change in mean MRS was recorded for each four stages of ERM.
Surgical procedure
Conventional three-port PPV was performed by one experienced vitreoretinal surgeon (RA) using 27-gauge vitrectomy system DORC (Dutch Ophthalmic Research Center, Zuidland, the Netherlands) and Zeiss microscope with EIBOS 2 (Haag Streit, USA) attachment for non-contact fundus viewing. In the case of advanced cataract as lens opacity of N3, C3, P3 or greater (LOCS III) (Chylack et al. 1993), a combined procedure was performed with simultaneous small-incision phacoemulsification (Alcon Infinity Vision System with Ozil IP). All patients received retrobulbar anaesthesia. Following removal of the core vitreous and posterior vitreous detachment MembraneBlue-Dual® (Dorc, International) was slowly injected over the macula and both the ERM and ILM were peeled in a circumferential pattern for about three disk diameters around the fovea using a 27 G disposable ultra peel forceps (DORC, Dutch Ophthalmic Research Center, Zuidland, the Netherlands). All patients received a standard postoperative protocol of topical antibiotic and anti-inflammatory combination (5 mg moxifloxacin and 1 mg dexamethasone).
Statistical analysis
Descriptive statistics are expressed as mean ± standard deviation or median (minimum–maximum). The normal distribution of the continuous variables was tested using the Kolmogorov–Smirnov test and the homogeneity of variance was tested using the Levene test. The data show a non-normal distribution and the variance between groups of ERM stages was not similar. Therefore, non-parametric tests were used for analysis. The significance of the difference between the independent groups was examined with the Mann–Whitney U and the Kruskal–Wallis test. The analysis of categorical data was performed using the Fischer exact test or Pearson’s Chi-square test. Univariate and multivariate logistic regression was used to evaluate associations of the EIFLs with BCVA. Analysis of the data was performed using IBM SPSS Statistics 21.0 software (IBM Corporation, Armonk, NY, USA). The results were considered statistically significant when p was <0.05.
Results
We reviewed the clinical records of 147 patients who underwent 27 G PPV for idiopathic ERM between January 2014 and January 2018. Of these 147 patients 35 were excluded because of the presence of one or more exclusion criteria and total of 112 patients were included in our study. Baseline characteristics of the patients enroled are summarised in Table 1. Patients had a mean age of 65.6 ± 7.7 (range 38–84), of whom 69 (61%) were female and 43 (39%) were male. Mean follow-up time was 22.2 ± 4.8 (range 12–40) months. There was no significant difference between groups with respect to age (p = 0.372), sex (p = 0.245).
Preoperatively, of the 112 eyes, 16 (14.3%) had stage 1, 20 (17.9%) had stage 2, 56 (50%) had stage 3 and 20 (17.9%) had stage 4 ERM. Our guidelines for surgical intervention were BCVA < 20/30 at diagnosis or moderate/intense metamorphopsia if BCVA is ≥20/30. Combined surgery was performed for 10 eyes (62.5%) in Stage 1, 13 eyes (65%) in Stage 2, 33 eyes (59%) in Stage 3 and 13 eyes (67%) in Stage 4 (p = 0.056). The preoperative BCVA was strongly correlated with postoperative month-6 and month-12 BCVA (p < 0.001).
Preoperative anatomical features on SD-OCT
The presence of EIFL was recorded in 76 of 112 eyes (68.4%), of which 56 were Stage 3 and 20 were Stage 4 ERM. The thickness of the EIFL was significantly higher in Stage 4 (307.4 ± 120 µm) compared with Stage 3 (141 ± 86.1 µm) (p < 0.001). The mean CFT was 461.2 ± 109.5 µm (range 290–939 µm) and increased significantly as the stages of the ERM advanced (p < 0.05 for all, Table 1). Furthermore, the CFT was strongly correlated with the thickness of EIFL (p < 0.001). The cystoid macular oedema was recorded in 43 of 112 patients (38.4%). As the stage of the ERM increased the rate of the cystoid macular oedema increased however the difference between stages did not reach statistical significance (p > 0.05). The cotton ball sign was detected in 18 of 112 patients (21.4%) preoperatively. The eyes with Stage 2 ERM showed significantly higher rates of cotton ball sign compared with other stages (p = 0.01). None of the eyes with Stage 1 and Stage 4 ERM showed cotton ball sign. The incidence of EZ defect was significantly higher in eyes with Stage 3 or 4 ERM than eyes with Stage 1 or 2 ERM (p < 0.001). Accordingly, the mean disruption diameter was significantly greater in eyes with Stage 4 compared with eyes with Stage 1 (p = 0.019) and Stage 2 (p = 0.03).
The ONL thickness of eyes with stage 2 ERM was statistically higher than that of eyes with Stage 1 (p < 0.001) and Stage 3 ERM (p = 0.001). However postoperative month-6 and month-12 mean ONL thicknesses were similar between groups (p > 0.05 for all).
The effect of EIFLs and other microstructural SD-OCT findings on preoperative visual acuity
The mean BCVA was statistically significantly higher in eyes with stage 1 (0.55 ± 0.47 logMAR, 20/63 Snellen equivalent) or stage 2 (0.45 ± 0.25 logMAR, 20/50 Snellen equivalent) ERM than in eyes with stage 3 (0.62 ± 0.45 logMAR, 20/80 Snellen equivalent) or stage 4 ERM (0.76 ± 0.43 logMAR, 20/100 Snellen equivalent) (p = 0.024). The EIFL thickness and CFT were statistically negatively correlated with the baseline visual acuity (p = 0.005, p = 0.032, respectively). Furthermore, the presence of the EIFL was associated with worse baseline BCVA (p = 0.013) (Table 2). However multivariate analysis revealed that the correlation between CFT and preoperative BCVA was statistically insignificant (p = 0.87). The diameter of the EZ disruption (p = 0.01) and presence of CMO (p = 0.027) was negatively correlated with the BCVA, whereas ONL thickness (p = 0.26) had no significant effect on preoperative visual acuity. The presence of cotton ball sign was significantly associated with greater baseline BCVA (p = 0.003).
The effect of EIFLs and other microstructural SD-OCT findings on postoperative visual acuity
Visual acuity improvement was statistically significant in all four stages (p < 0.05) however differences between Stage 2, 3 and 4 ERMs remained significant (p < 0.05). The presence of EIFLs was significantly negatively correlated with month-6 and month-12 BCVA in a univariate linear regression model (p < 0.001 for both). Furthermore, the thickness of the EIFL, disruption diameter of the EZ and CFT were significantly negatively correlated with the final visual acuity (p < 0.001 for all) (Fig. 3). This significance was further confirmed with a multivariate linear regression model, suggesting that the presence of EIFLs may be an independent negative prognostic factor for postoperative visual acuity (p = 0.03). Nevertheless, the correlation between the CFT and final BCVA was statistically insignificant in multivariate analyses (p = 0.72).
A statistically significant positive correlation was observed between postoperative month-12 BCVA and both central foveal thickness (p < 0.001) (A) and ectopic inner foveal layer thickness (p < 0.001) (B). However the correlation between outer nuclear layer thickness and final visual acuity was not statistically significant (p = 0.216) (C).
The ONL thickness had no significant effect on postoperative visual acuity (p = 0.216). The presence of CMO was associated with worse BCVA at month-6 (p = 0.04); however, the correlation was insignificant at month-12 (p = 0.08). The presence of cotton ball sign was significantly associated with greater postoperative BCVA at month-6 (p = 0.02) and month-12 (p = 0.03) (Table 2).
The FAZ area measurement on OCTA
The superficial FAZ area measurement was also performed in 10 eyes with Stage 1; 16 eyes with Stage 2, 21 eyes with Stage 3 and 17 eyes with Stage 4 ERM. The healthy fellow eye of the same patient was matched as a control. The mean FAZ are was significantly reduced in all stages of ERM compared with the fellow eyes (p < 0.05 for all). The FAZ area was statistically significantly smaller in eyes with stage 3 or stage 4 ERM than in eyes with stage 1 or stage 2 ERM (p = 0.012) and the FAZ was nearly absent in eyes with Stage 4 ERM. However, the mean preoperative FAZ area had no significant effect on final BCVA (p = 0.935).
Microperimetric changes with regard to the stage of ERM
Microperimetric assessment was also performed in 14 eyes with Stage 1; 16 eyes with Stage 2, 24 eyes with Stage 3 and 11 eyes with Stage 4 ERM. Preoperative MRS was 25.6 ± 4.2 dB, 21.8 ± 4.0 dB, 20.2 ± 4.1 dB and 17.1 ± 6.2 dB for Stage 1, Stage 2, Stage 3 and Stage 4, respectively. Final MRS was 28.4 ± 2.1 dB, 26.1 ± 3.4 dB, 21.7 ± 4.2 dB and 17.4 ± 6.4 dB for Stage 1, Stage 2, Stage 3 and Stage 4, respectively. Mean retinal sensitivity improvement was statistically significant in eyes with Stage 1 (p = 0.03) and Stage 2 ERM (p = 0.02), whereas no statistically significant change was observed in eyes with Stage 3 and Stage 4 ERM (p > 0.05 for all) (Fig. 1). Furthermore, thicknesses of the ONL (p = 0.038) and EIFL (p = 0.015) were significantly negatively correlated with the preoperative MRS. The disruption diameter of the EZ was significantly negatively correlated with the postoperative MRS values (p < 0.001).
Discussion
Recent advances in instrumentation and vitreoretinal surgical techniques have significantly improved the safety and efficacy of macular surgery [16, 17]. The mainstay of current ERM treatment involves PPV and membrane peeling with or without ILM peeling. However functional outcomes vary between studies [18, 19]. Despite high rates of visual improvement (70–80%) after vitrectomy, some of the patients still have limited visual acuity gain after successful membranectomy without any significant complications [20, 21]. Although the timing of surgery remains controversial, recent evidence suggests that particular microstructural changes on SD-OCT might be useful for the prediction of final visual acuity [12, 22]. Various predictors have been investigated to estimate the postoperative visual prognosis such as disruption of the EZ [23] and cone outer segment tips [12, 24], regional functional changes in the central retina using multifocal electroretinography [25], structural integrity of retinal layers [26] and subfoveal thickness of the inner retinal layers [27]. However, most of the staging systems developed to determine timing of surgery are impractical and time-consuming.
Recently, Govetto and co-workers [14] introduced the novel SD-OCT-based ERM staging scheme based on the presence of EIFLs. EIFL is defined as the presence of a continuous hyporeflective (extension of inner nuclear layer) or hyperreflective (extension of inner plexiform layer) band across the fovea. According to this staging scheme divided ERM classification into four stages. They reported a statistically significant negative correlation between the stage of the ERM and BCVA. Furthermore, the presence of the EIFLs was found to be an independent risk factor for lower BCVA. Subsequently, the same group studied the effect of the EIFL on postoperative functional and anatomical results after PPV with ERM and ILM peeling [28]. They showed that the improvement in BCVA reduces as the stage of the ERM increases and the preoperative presence of EIFL is strongly associated with lower postoperative month-12 BCVA. Priorly Joe et al. first investigated the clinical significance of inner retinal layer thickness (IRLT) in patients with idiopathic ERM. They reported that baseline IRLT was the main determinant of BCVA, which may provide an insight about the optimal surgical timing in patients with ERM [27]. Accordingly, in the current study, the presence of the EIFL was found to be an independent risk factor for lower preoperative and postoperative BCVA in multivariate linear regression analysis. There was a significant negative correlation between the BCVA and stage of the ERM both in the pre- and postoperative period. Furthermore, the thickness of the EIFL was strongly negatively correlated with the preoperative and postoperative BCVA.
Previous studies demonstrated a significant correlation between the CFT and baseline visual acuity [14, 29]. However, in a recent study reported by Govetto et al., the researchers performed mediation analysis to differentiate the sole effects of the EIFL thickness and CFT on BCVA. They found that the baseline CFT had no significant effect on preoperative visual acuity but higher values of baseline CFT was significantly correlated greater improvement in postoperative BCVA [28]. Similarly, in the current study, the mean CFT increased significantly as the stages of the ERM advanced and mean CFT was strongly correlated with the thickness of EIFL. Furthermore, both the preoperative and postoperative BCVA was significantly negatively correlated with the baseline CFT. However, on multivariate linear regression analysis the correlation of CFT with both preoperative and postoperative BCVA was statistically insignificant.
In contrast with the inner retinal biomarkers, which are frequently reversible, outer retinal biomarkers such as defects of the EZ and cone outer segment tip commonly reflect irreversible damage to the photoreceptors and therefore associated with poorer visual outcomes [30, 31]. In the current study, preoperative disruption diameter of the EZ was negatively correlated with both preoperative and postoperative BCVA. The mean ONL thickness of eyes with stage 2 ERM was statistically higher than that of eyes with Stage 1 (owing to prominent stretching of the ONL in Stage 2) and Stage 3 ERM (owing to the development of EIFLs in Stage 3).
The cotton ball sign was first introduced by Tsunoda et al. [15] as a roundish hyperreflective region resembling a cotton ball between the EZ and the cone outer segment tip line. In addition, eyes with the cotton ball sign demonstrated a good visual acuity and therefore the authors hypothesised that the cotton ball sign may indicate the presence of active traction on the fovea and early detection of this sign may be helpful in the management of patients with vitreomacular interface disorders. In our study, eyes with Stage 2 ERM showed higher rates of cotton ball sign and the presence of cotton ball sign was significantly associated with greater preoperative and postoperative BCVA. These results support the hypothesis that cotton ball sign is the result of continuous traction in the early–moderate stages of ERM prior to permanent disorganisation of the IRLs.
The reduction in the FAZ area is suggested to be a result of central displacement of vessels secondary to the tangentional tractional forces caused by the ERM [32]. Our results support this idea because as the stage of ERM and accordingly the tractional forces on the retinal surface increased, the FAZ area significantly decreased. Similarly, Govetto et al. reported significant reduction in the FAZ area in both the superficial and deep capillary plexus [16].
Microperimetry allows simultaneous assessment of the fixation point, retinal sensitivity, and microscotomas in macular diseases [33, 34]. Vecchio et al. reported a study investigating long-term microperimetric changes after idiopathic ERM surgery [35]. They stated that highest values of BCVA was achieved at postoperative month-12 and no significant changes were detected between 1 and 4 years. However, significant improvement was observed in mean retinal sensitivity between 1 and 4 years. In our study, despite significant visual improvement in all stages of ERM, MRS improvement was significant only in early stages of ERM.
To the best of our knowledge, this is the first study assessing the alterations of superficial FAZ area and mean retinal sensitivity with respect to SD-OCT-based ERM staging scheme before and after ERM surgery. In addition, recent study included the results of eyes with Stage 1 ERM.
The main limitation of our study was its retrospective design. Another limitation was that not all patients uniformly received combined cataract and vitreoretinal surgery and this might have affected the visual results. The strengths of our study include a long follow-up of a large study population. Furthermore, all surgeries were performed at the same centre by a single experienced vitreoretinal surgeon, using a standard method with ILM peeling being the only variable and SD-OCT evaluation was performed by two blinded, independent retina specialists. Further prospective, randomised, longitudinal studies with larger number of patients are needed to explore the potential effect of EIFL on functional and anatomical outcomes.
In conclusion, the SD-OCT classification scheme based on the presence of EIFL is a reliable, practical, and reproducible method to estimate the capacity of visual function. The presence of EIFL is an independent predictor of worse postoperative visual acuity. Accordingly, despite significant BCVA improvements in all stages of ERM, visual acuity gain remains limited in eyes with Stage 3 and Stage 4 ERM. In addition, our study revealed that the mean FAZ area has no significant effect on final BCVA and MRS improvement is limited in advanced stages of ERM.
Summary
What was known before
-
The presence and thickness of ectopic inner retinal layers are significantly negatively correlated with postoperative visual function.
What this study adds
-
To the best of our knowledge, this is the first study assessing the alterations of superficial FAZ area and mean retinal sensitivity with respect to SD-OCT-based ERM staging scheme before and after ERM surgery.
-
In addition, recent study included the results of eyes with Stage 1 ERM.
References
Iwanoff A. Beitrage zur normalen und pathologischen Anatomie des Auges. Graefes Arch Clin Exp Ophthalmol. 1865;11:135–70.
Ng CH, Cheung N, Wang JJ, Islam AF, Kawasaki R, Meuer SM., et al. Prevalence and risk factors for epiretinal membranes in a multi-ethnic United States population. Ophthalmology. 2011;118:694–9.
Cheung N, Tan SP, Lee SY, Cheung GCM, Tan G, Kumar N, et al. Prevalence and risk factors for epiretinal membrane: the Singapore Epidemiology of Eye Disease study. Br J Ophthalmol. 2016;101:371–6.
You Q, Xu L, Jonas JB. Prevalence and associations of epiretinal membranes in adult Chinese: the Beijing eye study. Eye(Lond). 2008;22:874–9.
Roth AM, Foos RY. Surface wrinkling retinopathy in eyes enucleated at autopsy. Trans Am Acad Ophthalmol Otolaryngol. 1971;75:1047–59.
Kadonosono K, Itoh N, Ohno S. Perifoveal microcirculation in eyes with epiretinal membranes. Br J Opthalmol. 1999;83:1329–31.
Council MD, Shah GK, Lee HC, Sharma S. Visual outcomes and complications of epiretinal membrane removal secondary to rhegmatogenous retinal detachment. Ophthalmology. 2005;112:1218–21.
Michalewski J, Michalewska Z, Cisiecki S, Nawrocki J. Morphologically functional correlations of macular pathology connected with epiretinal membrane formation in spectral optical coherence tomography (SOCT). Graefes Arch Clin Exp Ophthalmol. 2007;245:1623–31.
Falkner-Radler CI, Glittenberg C, Hagen S, Benesch T, Binder S. Spectral domain optical coherence tomography for monitoring epiretinalmembrane surgery. Ophthalmology. 2010;117:798–805.
Kim JH, Kim YM, Chung EJ, Lee SY, Koh HJ. Structural and functional predictors of visual outcome of epiretinal membranesurgery. Am J Ophthalmol. 2012;153:103–10.
Shimozono M, Oishi A, Hata M, Matsuki T, Ito S, Ishida K, et al. The significance of cone outer segment tips as a prognostic factor in epiretinal membrane surgery. Am J Ophthalmol. 2012;153:698–704.
Itoh Y, Inoue M, Rii T, Hirota K, Hirakata A. Correlation between foveal cone outer segment tips line and visual recovery after epiretinal membrane surgery. Invest Ophthalmol Vis Sci. 2013;54:7302–8.
Watanabe K, Tsunoda K, Mizuno Y, Akiyama K, Noda T. Outer retinal morphology and visual function in patients with idiopathic epiretinal membrane. JAMA Ophthalmol. 2013;131:172–7.
Govetto A, Lalane RA 3rd, Sarraf D, Figueroa MS, Hubschman JP. Insights into epiretinal membranes: presence of ectopic inner foveal layers and a new optical coherence tomography staging scheme. Am J Ophthalmol. 2017;175:99–113.
Tsunoda K, Watanabe K, Akiyama K, Usui T, Noda T. Highly reflective foveal region in optical coherence tomography in eyes with vitreomacular traction or epiretinal membrane. Ophthalmology. 2012;119:581–7.
Parke DW 3rd, Lum F. Return to the operating room afte macular surgery: IRIS registry analysis. Ophthalmology. 2018;125:1273–8.
Naruse S, Shimada H, Mori R. 27-gauge and 25-gauge vitrectomy day surgery for idiopathic epiretinal membrane. BMC Ophthalmol. 2017;17:188.
Kauffmann Y, Ramel JC, Lefebvre A, Isaico R, De Lazzer A, Bonnabel A, et al. “Preoperative prognostic factors and predictive score in patients operated on for combined cataract and idiopathic epiretinal membrane,”. Am J Ophthalmol. 2017;160:185–92.
Laban KG, Scheerlinck LM, van Leeuwen R. Prognostic factors associated with visual outcome after pars plana vitrectomy with internal limiting membrane peeling epiretinal membrane. Ophthalmologica. 2015;234:119–26.
Wong JG, Sachdev N, Beaumont PE, Chang AA. Visual outcomes following vitrectomy and peeling of epiretinal membrane. Clin Exp Ophthalmol. 2005;33:373–8.
Pareja J, Coronado A, Contreras I. Epiretinal membrane surgery in daily clinical practice: results of a proposed management scheme. J Ophthalmol. 2019;2019:8246858.
Miguel AI, Legris A. Prognostic factors of epiretinal membranes: a systematic review. J Fr Ophtalmol. 2017;40:61–79.
Kim JH, Kim YM, Chung EJ, Lee SY, Koh HJ. Structural and functional predictors of visual outcome of epiretinal membrane surgery. Am J Ophthalmol. 2012;153:103–10.
Shimozono M, Oishi A, Hata M, Matsuki T, Ito S, Ishida K, et al. The significance of cone outer segment tips as a prognostic factor in epiretinal membrane surgery. Am J Ophthalmol. 2012;153:698–704.
Hwang JU, Sohn J, Moon BG, Joe SG, Lee JY, Kim JG, et al. Assessment of macular function for idiopathic epiretinal membranes classified by spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2012;53:3562–9.
Uji A, Murakami T, Unoki N, Ogino K, Nishijima K, Yoshitake S, et al. Parallelism as a novel marker for structural integrity of retinal layers in optical coherence tomographic images in eyes with epiretinal membrane. Am J Ophthalmol. 2014;157:227–36.
Joe SG, Lee KS, Lee JY, Hwang JU, Kim JG, Yoon YH. Inner retinal layer thickness is the major determinant of visual acuity in patients with idiopathic epiretinal membrane. Acta Ophthalmol. 2013;91:242–3.
Govetto A, Virgili G, Rodriguez FJ, Figueroa MS, Sarraf D, Hubschman JP. Functional and anatomical significance of the ectopic inner foveal layers in eyes with idiopattic epiretinal membranes: surgical results at 12 months. Retina. 2019;39:347–57.
Doguizi S, Sekeroglu MA, Ozkoyuncu D, Omay AE, Yilmazbas P. Clinical significance of ectopic inner foveal layers in patients with idiopathic epiretinal membranes. Eye (Lond). 2018;132:1652–60.
Jeon S, Jung B, Lee WK. Long-term prognostic factors for visual improvement after epiretinal membrane removal. Retina. 2019;39:1786–93.
González-Saldivar G, Berger A, Wong D, Juncal V, Chow DR. Ectopic inner foveal layer classification scheme predicts visual outcomes after epiretinal membrane surgery. Retina. 2020;40:710–7.
Yoon YS, Woo JM, Woo JE, Min JK. Superficial foveal avascular zone area changes before and after idiopathic epiretinal membrane surgery. Int J Ophthalmol. 2018;11:1711–5.
Tadayoni R, Svorenova I, Erginay A, Gaudric A, Massin P. Decreased retinal sensitivity after internal limiting membrane peeling for macular hole surgery. Br J Ophthalmol. 2012;96:1513–6.
Karacorlu M, Ozdemir H, Senturk F, Karacorlu SA, Uysal O. Correlation of retinal sensitivity with visual acuity and macular thickness in eyes with idiopathic epimacular membrane. Int Ophthalmol. 2010;30:285–90.
Dal Vecchio M, Lavia C, Nassisi M, Grignolo FM, Fea AM. Microperimetric assessment after epiretinal membrane surgery: 4-year follow-up. J Ophthalmol. 2016; 7030791. https://doi.org/10.1155/2016/7030791.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
AMY was responsible for designing the study protocol, writing the protocol and report, conducting the search. SY was responsible for extracting and analysing data, interpreting results and updating reference lists. RA was responsible for analysing data, interpreting results and he contributed to writing the report.
Corresponding author
Ethics declarations
Conflict of interest
The authors whose names are listed immediately below certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical procedure
The research meets all applicable standards with regard to the ethics of experimentation and research integrity, and the following is being certified/declared true. As an expert scientist and along with co-authors of concerned field, the paper has been submitted with full responsibility, following the due ethical procedure, and there is no duplicate publication, fraud, plagiarism, or concerns about animal or human experimentation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mavi Yildiz, A., Avci, R. & Yilmaz, S. The predictive value of ectopic inner retinal layer staging scheme for idiopathic epiretinal membrane: surgical results at 12 months. Eye 35, 2164–2172 (2021). https://doi.org/10.1038/s41433-021-01429-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01429-w