Abstract
Objectives
To compare the visual acuity, contrast sensitivity (CS), spectacle needs, photic phenomena, and quality of life parameters of patients bilaterally implanted with a low add multifocal (MIOL) or an extended depth of focus (EDOF) intraocular lens (IOL), both with intended mini-monovision.
Methods
In this prospective, randomized, comparative study, patients were randomized to receive either Tecnis +2.75 D (ZKB00) (MIOL Group, n = 15) or Tecnis Symfony (ZXR00) (EDOF Group, n = 14) for bilateral implantation with mini-monovision (−0.50 D). Binocular logMAR uncorrected visual acuities (UVA), monocular defocus curves, CS with CSV 1000-E, and Pelli-Robson Test (PRT), spectacle needs and quality of life parameters with NEI RQL-42 questionnaire were evaluated at postoperative 1, 3, and 6 months.
Results
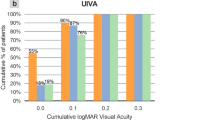
Results of MIOL and EDOF Groups at postoperative month 6 are as follows: distance (6 m) UVA −0.03 ± 0.05 and −0.05 ± 0.06 (p = 0.938), intermediate (60 cm) UVA, 0.04 ± 0.08 and −0.03 ± 0.07 (p = 0.046); near (40 cm) UVA, 0.22 ± 0.08 and 0.15 ± 0.07 (p = 0.046); near spectacle needs, 26.7% and 14.3% (p > 0.05), respectively. Better visual acuity was achieved in the EDOF Group between the defocus range of −0.50 and −1.75 D (p < 0.05). No significant difference was found regarding photic phenomena and CS evaluated with CSV 1000-E between the two IOL groups at 6 months after surgery (otherwise there are differences at 1 and 3 months in favor of EDOF). However, EDOF Group performed better in mesopic CS evaluated with PRT (p < 0.05).
Conclusions
When implanted with mini-monovision better binocular uncorrected visual performance at intermediate and near distances achieved with EDOF than low add MIOL.
Similar content being viewed by others
Introduction
The innovations in phacoemulsification surgery and advances in surgical equipment ensure a safe extraction of the crystalline lens and implantation of intraocular lenses (IOL). As a result of developing IOL technologies and changing patient demands, many multifocal, accommodative, toric, toric-multifocal, and extended depth of focus (EDOF) IOLs have been introduced and the original aim of restoration of far vision with implanted monocular IOLs has been evolved to an objective of improved vision at different distances with total spectacle independence. One of the first IOL designs produced for this purpose, bifocal diffractive multifocal IOLs (MIOL), have been shown to improve near vision without reducing far vision [1]. However, as necessities of our era, the use of computers, tablets, and smartphones increased the visual demands at intermediate distances, and high add MIOLs have been shown to be insufficient for this purpose [1, 2]. Manufacturers found the solution as reducing the add power in bifocal IOL designs and it has been shown to increase intermediate vision with preserved functional near vision [3]. Tecnis ZKB00 is one of the low add MIOL with an addition of +2.75 D near power developed to provide more functional visual restoration in patients with better vision demand at intermediate distance (~50 cm) [4]. It has been shown that it performs better than higher add MIOL at intermediate distance [2, 4, 5] but worse at near vision than other counterparts designed with higher add (Tecnis ZLB00 [+3.25] and ZMB00 [+4.00]) [2].
As an alternative to changing IOL designs, different bilateral implantation techniques have been applied to improve vision at near and intermediate distances, such as pseudophakic monovision and blended (mix-and-match) implantation, and both implantation techniques have been shown to be providing functional vision at different distances [6, 7]. However, new IOL designs with various technological approaches continue to evolve to improve intermediate vision and spectacle independence. Among them, trifocal and EDOF IOL designs are the newest. While trifocal IOLs create three different focal points to provide vision at different distances [8], the basic principle of EDOF IOLs is to create a longitudinal plane of focus to increase the range of vision [9]. Tecnis Symfony (ZXR00) is one of the EDOF IOLs with a unique echelette design showing promising results in obtaining good visual acuity at different distances with its continuous elongated focus while minimizing visual disturbances with the correction of spherical and chromatic aberrations [10].
This study aimed to compare the visual acuity at different distances, contrast sensitivity (CS), spectacle needs, photic phenomena, and quality of life parameters of patients bilaterally implanted with a low add bifocal (Tecnis ZKB00) or an EDOF IOL (Tecnis Symfony), both with planned mini-monovision to improve near vision.
Methods
The study protocol was approved by the Institutional Review Board of Marmara University School of Medicine Hospital (No: 09.2016.309), and the study was financially supported by the Scientific Research Project Commission of Marmara University School of Medicine, Istanbul, Turkey (Project No: SAG-A-131216–0523). The study was performed in accordance with the Declaration of Helsinki principles, and a written informed consent about participating in the study and having their medical information used in the study analysis was provided from all of the patients. The study was registered at ClinicalTrials.gov (NCT04865016).
This prospective, randomized, comparative study included 30 eyes of 15 cataract patients undergoing cataract surgery with implantation of a low add (+2.75 Diopters [D]) bifocal (Tecnis ZKB00; Johnson and Johnson Surgical Vision Inc., Jacksonville, Florida, USA) IOL (MIOL Group) and 30 eyes of 15 patients with implantation of an EDOF (Tecnis Symfony [ZXR00]; Johnson and Johnson Surgical Vision Inc., Jacksonville, Florida, USA) IOL (EDOF Group). Patients were randomized into either one of the groups by an online random integer generator (www.random.org/integers).
Inclusion criteria were patients with bilateral visually significant cataract seeking spectacle independence, especially at far and intermediate distances, age of ≥18 years, preoperative corneal astigmatism <1.00 D, axial length (AL) between 21 and 26.5 mm, and availability to attend all follow-up visits. Patients were excluded from the study if any of the following conditions were present: high visual demand for near vision, amblyopia, glaucoma, corneal opacity, severe/moderate to severe dry eye, pupillary abnormalities and dysfunction, corneal astigmatism ≥1.00 D, AL <21 mm, or ≥26.5 mm, diabetes mellitus with retinal changes, active uveitis or history of uveitis, previous ocular surgery and lack of attendance to any follow-up visit.
Surgical technique
All patients underwent standard small incision phacoemulsification surgery under combined topical-intracameral anesthesia by the same experienced surgeon (ET). A continuous curvilinear capsulorrhexis measuring 5–5.5 mm in diameter was generated with micro-forceps. The IOLs were inserted into the capsular bag using 1MTEC30 model inserter (UNFOLDER® Platinum 1 Series, Johnson and Johnson Surgical Vision Inc., Jacksonville, Florida, USA) through the main incision. IOL centralization was performed with the Purkinje 1 reflex in the central opening of diffractive steps. In all cases, the same phacoemulsification device (Centurion Vision System, Alcon Laboratories, Fort Worth, Texas, USA) was used. Postoperative therapy included topical moxifloxacin, prednisolone, and nepafenac for 1 month.
Intraocular lenses
The Tecnis +2.75 D (ZKB00) IOL is a diffractive multifocal IOL designed with a full diffractive posterior surface with 15 diffractive rings. It has an addition of +2.75 D at the IOL plane. It has an aspheric anterior surface with −0.27 µm spherical aberration to compensate corneal positive spherical aberrations [4]. Tecnis Symfony (ZXR00) EDOF IOL has a diffractive echelette design posterior surface with nine diffractive rings. In addition to the aspheric anterior surface with −0.27 µm spherical aberration, it also has a posterior achromatic design to correct chromatic aberrations [11]. Both IOLs are made of the same hydrophobic acrylic material with UV protection, and both have the same haptic properties.
Patient examinations
Preoperative evaluation
A complete ophthalmological examination was performed in all patients, including measurement of monocular and binocular uncorrected and corrected distance (UDVA, CDVA; at 6 m), intermediate (UIVA, CIVA; at 60 cm), and near (UNVA, CNVA; at 40 cm) logMAR visual acuity, manifest spherical equivalent (SE) refraction, Goldmann applanation tonometry, keratometry, slit-lamp anterior segment examination, photopic pupil size (mm) measurement, fundus examination with pupil dilatation and macular optical coherence tomography (OCT; Spectralis, Heidelberg Engineering, Heidelberg, Germany). Kappa angle and chord µ length (Cartesian displacement between subject fixated coaxially sighted corneal light reflex and center of the entrance pupil, calculated from the Kappa Intercept values) [12] were measured with Orbscan II (Bausch and Lomb, Rochester, NY, USA). AL was measured with the Lenstar Optical Biometer (Haag-Streit AG, Bern, Switzerland), and spherical IOL power was calculated using the SRK-T formula. Ocular dominance was decided with Dolman Method (Hole-in-the-card test). The IOL power was targeted for emmetropia in the dominant eye and approximately −0.50 D myopia in the nondominant eye.
Preoperative and postoperative evaluations
Postoperative examinations were performed 1, 3, and 6 months after surgery. All patients underwent measurements of monocular and binocular UDVA and CDVA, monocular and binocular uncorrected and distance-corrected intermediate (UIVA, DCIVA; at 60 cm) and near (UNVA, DCNVA; at 40 cm) logMAR visual acuity, manifest SE refraction, Goldmann applanation tonometry, slit-lamp anterior segment examination, fundus examination with pupil dilatation and macular OCT.
Distance visual acuity was measured with LCD screen version (CSO Vision Chart, Mod CVC02, version 1.3.0, Florence, Italy) of Snellen Charts under photopic conditions (85 cd/m2). Intermediate and near visual acuities were measured and recorded with a Turkish reading chart prepared with reference to Bailey-Lovie and ETDRS (Early Treatment Diabetic Retinopathy Study) reading charts covering international standards [13]. The logMAR values of the chart designed for 35 cm distance were corrected with the formula “log10(standard distance/new distance [cm])” according to the distance used (40 and 60 cm) [14].
CS was evaluated at 3, 6, 12, and 18 cycle per degree (cpd) spatial frequencies with CSV-1000-E test (VectorVision, Ohio, USA) under mesopic (3.5 candela [cd]/m2) conditions at 2.5 m with distance refractive correction. The test was conducted with and without glare, and as a glare source, two halogen lamps located either side of the test chart and producing 2.5 cd/m2 luminance at eye plane were used. CS at different spatial frequencies was compared with the physiologic CS range for normal subjects of similar age [15]. CS was also evaluated with the LCD screen version (CSO Vision Chart, Mod CVC02, version 1.3.0, Florence, Italy) of Pelli-Robson Test (PRT) under photopic (85 cd/m2) and mesopic (3.5 cd/m2) conditions at 3 m monocularly with distance refractive correction. To the best of our knowledge, PRT CS normal values, performed under similar conditions, are not available in the literature, so the data was used only for intergroup comparisons [16, 17].
Quality of life parameters were evaluated by the validated Turkish version [18] of the National Eye Institute Refractive Error Quality of Life Instrument 42 (NEI RQL-42) questionnaire [19]. It includes 13 subscales of quality of life parameters calculated according to a scoring key, and a “total score” can be obtained by calculating the mean value of all 42 questions [19].
Postoperative evaluation
Patients were evaluated for posterior capsule opacification (PCO), spectacle needs at far, intermediate, and near distances, and photic phenomena at all postoperative visits. For PCO evaluation, the proposed classification system of Abela-Formanek et al. [20] (Grade 0, no observable PCO; grade 1, transparent PCO observable with retroillumination; grade 2, gray-white PCO with retroillumination; grade 3, dense white fibrosis or presence of Elchnig pearls) was used. Spectacle needs of patients were categorized as 0%, 1–24%, 25–49%, 50–74%, and 75–100% of the time for far, intermediate and near distances separately. Photic phenomena were explained to the patients as follows: glare, glows around lights or lighted objects; halo, circular ghost images around lights or lighted objects; and starburst, star-shaped elongations around lights or lighted objects. Then patients were asked to grade the photic phenomena subjectively as grade 0, absent; grade 1, mild; grade 2, moderate; and grade 3, severe.
At postoperative month 6, monocular defocus curves were obtained after correcting the refraction for distance. The curves obtained by measuring distance (6 m) visual acuity between +1.50 D and −4.50 D defocus range with decrements of 0.25 D between +1.50 and 0.00 D, and with increments of 0.25 D between −4.50 and 0.00 D.
Statistical analysis
The sample size was calculated based on the binocular UIVA (60 cm, 0.24 ± 0.11) and UNVA (40 cm, 0.27 ± 0.11) values of the prospective study by Pedrotti et al. [10]. A difference of 0.12 logMAR (6 letters = more than 1 Snellen line) assumed to find a clinically significant difference between two IOL groups. Based on these assumptions and alpha value set to 0.05 with a power of 0.8, 14 patients in each group were decided to be required. With a drop rate assumption of 10%, 15 patients were randomized to each IOL group.
For statistical analysis of the data, SPSS for Windows version 17.0 (SPSS Inc., Chicago, Illinois, USA) was used. The normal distribution of the variables was evaluated by histogram graphs and the Kolmogorov-Smirnov test. While the analysis of descriptive data was presented, mean and standard deviation values were given. Pearson Chi-Square and Fisher’s Exact tests were used in the comparison of 2 × 2 tables. Two independent variables with and without a normal distribution were evaluated with the Independent Samples T test and the Mann–Whitney U test, respectively. Paired T test or Wilcoxon signed-rank tests were used to compare data of two dependent variables with or without a normal distribution, respectively. The Friedman Test was used to compare more than two dependent variables without a normal distributed data, and Dunn–Bonferroni post hoc test was used for pair-wise comparisons. A p value of <0.05 was considered statistically significant.
Results
Sixty eyes of 30 patients were randomized into one of the IOL groups (MIOL or EDOF Group); however, one of the patients in the EDOF Group was excluded from the study analysis because of the inability to attend follow-up visits. Fifty-eight eyes of 29 patients (15 patients in MIOL Group, and 14 patients in EDOF Group) were included in the study analysis. The preoperative characteristics of patients in the groups are summarized in Table 1.
Postoperative binocular UDVA, UIVA, UNVA, and manifest SE values are given in Table 1. There was no statistically significant difference between groups with regard to manifest SE of dominant and nondominant eyes of the patients at all visits after surgery. Binocular UDVA of the groups was comparable; however, binocular UIVA was significantly better in the EDOF Group at 1, 3, and 6 months after surgery (p = 0.004, 0.001, and 0.046, respectively). Binocular UNVA was significantly better in the EDOF Group at 3 and 6 months after surgery (p = 0.001 and p = 0.046, respectively).
Cumulative binocular uncorrected visual acuities (UVA) of the groups at postoperative month 6 are shown in Fig. 1. All patients have 0.00 logMAR or better UDVA (Snellen equivalent: 20/20); 0.20 logMAR or better UIVA (Snellen equivalent: 20/32); and 0.3 logMAR or better UNVA (Snellen equivalent: 20/40) in both groups at postoperative month 6.
The monocular defocus curves of the patients in the MIOL (n = 30) and EDOF (n = 28) Groups are shown in Fig. 2A. Better visual acuities were obtained in the EDOF Group between the defocus range of −0.50 and −1.75 D (p < 0.001 between −0.50 and −1.50 D; p = 0.006 at −1.75 D).
A Mean distance-corrected monocular defocus curves of the patients in the MIOL and EDOF Groups. B Contrast sensitivity with and without glare changes over time in the MIOL and EDOF Groups. D diopters, logMAR logarithm of the minimum angle of the resolution. *p < 0.001, Mann–Whitney U test. †p = 0.006, Mann–Whitney U test.
CS with and without glare evaluated at different spatial frequencies with CSV 1000-E Test is summarized in Table 2. Better CS without glare was obtained at 3, 6, and 12 cpd spatial frequencies in the EDOF Group 1 month after surgery (p = 0.006, 0.005, and 0.016, respectively). In the EDOF Group, better CS with glare was obtained at 6 cpd spatial frequency at 1 month after (p = 0.020), and at 6 and 12 cpd spatial frequencies 3 months after surgery (p = 0.020 and 0.020, respectively). At 6 months after surgery, no statistically significant difference was found in CS with and without glare at any spatial frequency between the two IOL groups.
In the MIOL Group, CS without glare was below the normal range at 3, 6, and 12 cpd spatial frequencies at 1 month after surgery, but it improved to be within the normal range at 3 months. In the MIOL Group, CS with glare at lower spatial frequencies was below the normal range at postoperative month 1 (3 and 6 cpd) and month 3 (6 cpd). However, in the EDOF Group, CS values with and without glare were within the normal range at all time points. The change in CS with and without glare over time is given in Fig. 2B.
Regarding CS with PRT, the only statistically significant difference observed was at 6 months after surgery under mesopic conditions; better CS values were obtained in the EDOF Group (p = 0.022). Table 2 summarizes the postoperative PRT CS values of IOL groups under photopic and mesopic conditions.
The eyes with grade 1 PCO 6 months after surgery were 6 (20%) and 2 (7.14%) in the MIOL and EDOF Groups, respectively (Chi-Square Test; p = 0.156). None of the patients had grade 2 or more PCO in either IOL group.
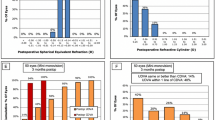
Regarding the patients’ spectacle needs at postoperative month 6, none of the patients needed to use spectacles for far and intermediate distance. For near distances, 26.7% and 14.3% of the patients required spectacles <50% of the time in the MIOL and EDOF Groups, respectively (p = 0.546).
The evaluation of photic phenomena at 6 months after surgery revealed 86.7% and 100% no or mild photic phenomena in the MIOL and EDOF Groups, respectively (p = 0.150) (Supplementary Table 1).
There was no statistically significant difference between the two IOL groups in any subscale and total score values of NEI RQL-42 questionnaire (Supplementary Table 2).
Discussion
Monofocal IOL implantation targeted for distance emmetropia after phacoemulsification is highly dependent on spectacle correction for near and intermediate vision. Many IOL designs have been developed to improve intermediate and near vision, thereby to improve patient satisfaction and quality of life. In this study, we investigated the effects of two IOL options (low add [+2.75 D] diffractive multifocal IOL Tecnis ZKB00 and EDOF IOL Tecnis Symfony, both implanted with −0.50 D mini-monovision to improve near visual acuity) on visual performance at different distances, contrast sensitivities, spectacle needs, photic phenomena and quality of life of patients. When implanted with mini-monovision, we found better intermediate and near visual acuities achieved with Tecnis Symfony than Tecnis ZKB00 without significant difference in distance visual acuity at postoperative month 6.
In the multicentre Concerto Study of Cochener Tecnis Symfony was implanted in both eyes of 411 patients, with intended mini-monovision (mean −0.75 ± 0.52 D in nondominant eyes) in 122 of the patients, and emmetropia was targeted in 299. There was no difference between the two groups in terms of binocular UDVA. In contrast, binocular UIVA (at 70 cm) and UNVA (at 40 cm) were better in the mini-monovision group [21]. Those findings were later supported by another prospective study conducted with −0.50 D intended mini-monovision (UDVA at 5 m, UIVA at 67 cm and UNVA at 33 cm) [22]. In a subanalysis of the Concerto Study, patients implanted with the closest monovision to our study (0.25 < x ≤ 0.50 D) have the highest positive response about choosing the same lens again and recommending the same procedure to their friends and family [23]. In another study where the Tecnis Symfony implanted with −0.75 D monovision, it was stated that excellent visual performance was achieved in binocular far and intermediate (60 and 80 cm) distances and a satisfactory visual performance was achieved in the near distance (40 cm) [24]. In a recent study, the best visual acuity results were achieved with bilateral implantation of Tecnis Symfony or Tecnis Symfony Toric when the dominant eye is targeted at emmetropia and the nondominant eye is targeted between −0.21 D and −0.63 D [25]. Also, in the subset of patients who achieved excellent visual acuity at all distances, mean manifest SE for dominant and nondominant eyes was detected as −0.07 ± 0.14 D and −0.21 ± 0.24 D, respectively [25].
It is difficult to compare data over different studies, in its nature, as the criteria in the assessment of intermediate vision or targeted myopia for implementation of monovision differ. For both IOL designs, reported intermediate visual acuities differ with regard to the accepted viewing distance, e.g., 70 cm [21, 26, 27], 80 cm [4, 28, 29], 100 cm [30]. However, binocular UDVAs, UIVAs at 60 cm, and UNVAs of patients implanted with Tecnis Symfony in our study are comparable with the results of the existing literature (Table 3) [10, 24, 31,32,33].
To our knowledge, there is no study in the literature evaluating Tecnis +2.75 implanted with monovision. Kretz et al. in their multicentre study evaluating Tecnis +2.75 implanted by targeting bilateral emmetropia, found that postoperative binocular UDVA, UIVA at 80 cm and UNVA as 0.06 ± 0.12 (range, 0.00–0.40), 0.20 ± 0.17 (range, 0.10–0.40), and 0.08 ± 0.24 (range, 0.00–0.80) logMAR, respectively [4]. Although UIVA and UNVA seem to be in accordance with our findings, intermediate vision measured at 60 cm with mini-monovision could have favorably affected the UIVA results and slightly improved the UNVA range (0.1–0.3 logMAR) in our study [4].
There is a limited number of studies in the literature comparing the Tecnis Symfony and bifocal IOLs, especially with low add power [28, 30, 31]. Pedrotti et al. compared the clinical performance of Tecnis Symfony, Tecnis Monofocal (ZCB00), and apodized diffractive-refractive MIOL ReSTOR +2.50 and +3.00 (Alcon Laboratories, Inc., Fort Worth, Texas) bilaterally implanted without monovision [31]. For UIVA at 60 cm, although ReSTOR +2.50 performed better than the other three IOLs, the authors stated the magnitude of this difference between Tecnis Symfony and ReSTOR +2.50 was small [31]. In contrast, we found that Tecnis Symfony performed better than Tecnis +2.75 in terms of intermediate vision when both lenses were implanted targeting −0.50 D in the nondominant eye. The results in terms of UNVA in our study were consistent with those in the aforementioned study, reporting Tecnis Symfony (0.18 ± 0.10 logMAR) performing better than ReSTOR +2.50 (0.28 ± 0.11 logMAR) but worse than ReSTOR +3.00 (0.05 ± 0.08 logMAR) [31].
There is only one study in the literature directly comparing Tecnis Symfony and Tecnis +2.75 with four other IOLs (ReSTOR + 2.50, AT LISA 809 M, and AT LISA Tri 839MP [Carl Zeiss Meditec AG, Jena, Germany], and Tecnis ZLB00), which was assessed by distance-corrected monocular visual acuities [34]. The study showed that best DCIVA (60 cm) results were achieved with Tecnis Symfony followed by Tecnis +2.75. Better DCNVA (40 cm) was achieved with Tecnis +2.75 (0.127 ± 0.109 logMAR) than with Tecnis Symfony (0.238 ± 0.097 logMAR) (p < 0.001, reported result of ANOVA test with all IOLs in the study) [34]. However, the study results cannot be directly compared with our results since mini-monovision was applied in our study, and binocular summation might have favored the visual results achieved by Tecnis Symfony.
In the present study, although significantly better binocular UNVA was obtained with Tecnis Symfony compared to Tecnis +2.75 (0.15 ± 0.07 vs. 0.22 ± 0.08 logMAR, respectively, p = 0.046) at postoperative month 6, the cumulative visual acuity of 0.3 logMAR and better was achieved 100% in both IOL groups. Sanders and Sanders [35] examined the levels of near visual acuity required for daily activities, and the commonly read print objects in everyday life were compared with the logMAR near reading acuity scale (ETDRS, Chart “1”; Precision Vision, La Salle, Illinois, USA). They found that the visual acuity required to read the smallest print objects (nutritional values on sweetener packets) for social reading needs to correspond to 0.3–0.4 logMAR [35]. Accordingly, we can interpret that, when implanted with mini-monovision, both IOLs in our study can provide functional near visual acuity.
The results of our study in terms of defocus curves are compatible with in vivo and in vitro studies in the literature [10, 24, 36,37,38]. Two peaks were determined for Tecnis +2.75 in accordance with the bifocality of the IOL, whereas a plateau was formed in accordance with the EDOF feature of the Tecnis Symfony instead of a peak.
In studies with Tecnis Symfony implanted bilaterally with mini-monovision, spectacle need has been reported to be between 0 and 16% for intermediate, and, between 2 and 19.2% for near vision [21, 22, 24, 27, 30]. And, for Tecnis +2.75, only 15.3% of 85 patients after bilateral implantation without monovision were required to use spectacles for some daily activities [4]. In our study, there was no spectacle need for far and intermediate distances, whereas the rate of spectacle need for near distance was 14.3% in the EDOF Group and 26.7% in the MIOL Group (p > 0.05).
Tecnis Symfony and Tecnis +2.75 multifocal IOL have the same negative spherical aberration value (−0.27 µm), and by their aspherical designs, both IOLs are expected to reduce corneal positive spherical aberrations to almost zero and thereby increase CS. Tecnis Symfony also has achromatic technology to reduce chromatic aberrations [39, 40]. Our results show that, although CS at low and intermediate spatial frequencies for Tecnis +2.75 remained below the normal range at the first and third postoperative months, there was no statistically significant difference at any spatial frequency between the two IOL groups at postoperative month 6. This can be interpreted as a possible faster neuroadaptation with Tecnis Symfony which can be an area that needs to be elucidated by further studies. However, at the final follow-up visit, Tecnis Symfony performed better than Tecnis +2.75 only at mesopic letter contrast sensitivity (PRT CS).
In accordance with the results of previous studies [4, 21, 24], the frequency and severity of self-reported visual phenomena in our series were comparable between the two IOL types. In an in vitro study comparing Tecnis Symfony, Tecnis Monofocal (ZCB00), Tecnis +2.75 (ZKB00), and Tecnis +4.00 (ZMB00), halo and glare rates were found to be similar between Tecnis Symfony and Tecnis Monofocal IOLs, which were lower than the other two multifocal/bifocal IOLs, and this has been attributed to the lower numbers of diffractive rings in Tecnis Symfony (9 vs. 15 and 22 respectively) [36]. However, in the study of Pedrotti et al. comparing Tecnis Symfony, Tecnis Monofocal, and ReSTOR MIOL, photic phenomenon complaint evaluated with Glare subscale of NEI RQL-42 was found to be comparable between the four IOLs [31].
When NEI-RQL-42 scores were evaluated, no significant difference was found between the two IOL groups, and overall satisfaction scores were higher than 85 for both IOLs. However, both IOL groups remained below 80 points in the subscales of “dependence on correction”, “glare” and “symptoms”. In the study of Pedrotti et al. scores of “glare” and “dependence of correction” subscales of the patients implanted with Tecnis Symfony were given as 69.34 ± 27.60 and 77.94 ± 25.72, respectively which was comparable with our results [31]. To our knowledge, there are no studies in the literature evaluating NEI RQL-42 results of Tecnis +2.75.
The strengths of our study are its prospective, randomized, and comparative nature. However, several limitations of this study should be mentioned while interpreting the results. Those limitations can be listed as; pupil diameter being measured only preoperatively in photopic conditions, that might have influenced CS and photic phenomena results of the study, subjective evaluation of photic phenomena, lack of a control group in which mini-monovision was not applied, and relatively small number of patients. Also, patients included in this study were subjects who did not require high demands for near vision, which might have positively biased the subjective performance.
In conclusion, when implanted with mini-monovision (−0.50 D), both IOLs can provide functional visual performance at intermediate and near distance without reducing distance visual acuity, with a tendency of better results with Tecnis Symfony EDOF IOL. To confirm our study results, mini-monovision should be evaluated for both IOL groups by forming control groups without monovision in larger sample sizes.
Summary
What was known before
-
Low add MIOL Tecnis +2.75 D (ZKB00) improves intermediate visual acuity with a functional near vision, and EDOF IOL Tecnis Symfony (ZXR00) improves visual acuity at different distances with its continuous elongated focus.
What this study adds
-
When implanted with mini-monovision, both IOLs can provide functional visual performance at intermediate and near distance with a tendency of better results with EDOF Tecnis ZXR00.
-
There might be a possible faster neuroadaptation with EDOF IOL regarding CS.
References
Rosen E, Alio JL, Dick HB, Dell S, Slade S. Efficacy and safety of multifocal intraocular lenses following cataract and refractive lens exchange: metaanalysis of peer-reviewed publications. J Cataract Refract Surg. 2016;42:310–28.
Kim JS, Jung JW, Lee JM, Seo KY, Kim EK, Kim TI. Clinical outcomes following implantation of diffractive multifocal intraocular lenses with varying add powers. Am J Ophthalmol. 2015;160:702–9.e1.
Hayashi K, Manabe S, Hayashi H. Visual acuity from far to near and contrast sensitivity in eyes with a diffractive multifocal intraocular lens with a low addition power. J Cataract Refract Surg. 2009;35:2070–6.
Kretz FT, Gerl M, Gerl R, Muller M, Auffarth GU. Clinical evaluation of a new pupil independent diffractive multifocal intraocular lens with a +2.75 D near addition: a European multicentre study. Br J Ophthalmol. 2015;99:1655–9.
Alfonso JF, Fernandez-Vega L, Puchades C, Montes-Mico R. Intermediate visual function with different multifocal intraocular lens models. J Cataract Refract Surg. 2010;36:733–9.
Finkelman YM, Ng JQ, Barrett GD. Patient satisfaction and visual function after pseudophakic monovision. J Cataract Refract Surg. 2009;35:998–1002.
Jacobi FK, Kammann J, Jacobi KW, Grosskopf U, Walden K. Bilateral implantation of asymmetrical diffractive multifocal intraocular lenses. Arch Ophthalmol. 1999;117:17–23.
Madrid-Costa D, Ruiz-Alcocer J, Ferrer-Blasco T, Garcia-Lazaro S, Montes-Mico R. Optical quality differences between three multifocal intraocular lenses: bifocal low add, bifocal moderate add, and trifocal. J Refract Surg. 2013;29:749–54.
Akella SS, Juthani VV. Extended depth of focus intraocular lenses for presbyopia. Curr Opin Ophthalmol. 2018;29:318–22.
Pedrotti E, Bruni E, Bonacci E, Badalamenti R, Mastropasqua R, Marchini G. Comparative analysis of the clinical outcomes with a monofocal and an extended range of vision intraocular lens. J Refract Surg. 2016;32:436–42.
Gatinel D, Loicq J. Clinically relevant optical properties of bifocal, trifocal, and extended depth of focus intraocular lenses. J Refract Surg. 2016;32:273–80.
Chang DH, Waring GO 4th. The subject-fixated coaxially sighted corneal light reflex: a clinical marker for centration of refractive treatments and devices. Am J Ophthalmol. 2014;158:863–74.
Egrilmez S, Egrilmez ED, Akkin C, Kaskaloglu M, Yagci A. A new turkish near reading chart which covers international standards. Turk J Ophthalmol. 2004;34:404–12.
Bailey IL, Jackson AJ. Changes in the clinical measurement of visual acuity. J Phys Conf Ser. 2016;772:012046.
Pomerance GN, Evans DW. Test-retest reliability of the CSV-1000 contrast test and its relationship to glaucoma therapy. Investig Ophthalmol Vis Sci. 1994;35:3357–61.
Mantyjarvi M, Laitinen T. Normal values for the Pelli-Robson contrast sensitivity test. J Cataract Refract Surg. 2001;27:261–6.
Puell MC, Palomo C, Sanchez-Ramos C, Villena C. Normal values for photopic and mesopic letter contrast sensitivity. J Refract Surg. 2004;20:484–8.
Toker E, Onal S, Eraslan M, Eyriparmak M. The Turkish version of the National Eye Institute Refractive Error Quality of Life Instrument: translation, validity and reliability. Qual Life Res. 2008;17:1269–76.
Hays RD, Spritzer KL. National Eye Institute Refractive Error Quality of Life Instrument (NEI-RQL-42™), version 1.0: a manual for use and scoring. Rand Health Sciences: Los Angeles, CA; 2002.
Abela-Formanek C, Amon M, Schild G, Schauersberger J, Heinze G, Kruger A. Uveal and capsular biocompatibility of hydrophilic acrylic, hydrophobic acrylic, and silicone intraocular lenses. J Cataract Refract Surg. 2002;28:50–61.
Cochener B. Clinical outcomes of a new extended range of vision intraocular lens: International Multicenter Concerto Study. J Cataract Refract Surg. 2016;42:1268–75.
Tan J, Qin Y, Wang C, Yuan S, Ye J. Visual quality and performance following bilateral implantation of TECNIS Symfony intraocular lenses with or without micro-monovision. Clin Ophthalmol. 2019;13:1071–7.
Cochener B. Influence of the level of monovision on visual outcome with an extended range of vision intraocular lens. Clin Ophthalmol. 2018;12:2305–12.
Ganesh S, Brar S, Pawar A, Relekar KJ. Visual and refractive outcomes following bilateral implantation of extended range of vision intraocular lens with micromonovision. J Ophthalmol. 2018;2018:7321794.
Jackson MA, Edmiston AM, Bedi R. Optimum refractive target in patients with bilateral implantation of extended depth of focus intraocular lenses. Clin Ophthalmol. 2020;14:455–62.
Kaymak H, Hohn F, Breyer DR, Hagen P, Klabe K, Gerl RH, et al. Functional results 3 months after implantation of an “extended range of vision” intraocular lens. Klin Monbl Augenheilkd. 2016;233:923–7.
Sachdev GS, Ramamurthy S, Sharma U, Dandapani R. Visual outcomes of patients bilaterally implanted with the extended range of vision intraocular lens: a prospective study. Indian J Ophthalmol. 2018;66:407–10.
Attia MSA, Auffarth GU, Kretz FTA, Tandogan T, Rabsilber TM, Holzer MP, et al. Clinical evaluation of an extended depth of focus intraocular lens with the Salzburg reading desk. J Refract Surg. 2017;33:664–9.
Pilger D, Homburg D, Brockmann T, Torun N, Bertelmann E, von Sonnleithner C. Clinical outcome and higher order aberrations after bilateral implantation of an extended depth of focus intraocular lens. Eur J Ophthalmol. 2018;28:425–32.
Hogarty DT, Russell DJ, Ward BM, Dewhurst N, Burt P. Comparing visual acuity, range of vision and spectacle independence in the extended range of vision and monofocal intraocular lens. Clin Exp Ophthalmol. 2018;46:854–60.
Pedrotti E, Carones F, Aiello F, Mastropasqua R, Bruni E, Bonacci E, et al. Comparative analysis of visual outcomes with 4 intraocular lenses: Monofocal, multifocal, and extended range of vision. J Cataract Refract Surg. 2018;44:156–67.
Ruiz-Mesa R, Abengozar-Vela A, Aramburu A, Ruiz-Santos M. Comparison of visual outcomes after bilateral implantation of extended range of vision and trifocal intraocular lenses. Eur J Ophthalmol. 2017;27:460–5.
Kohnen T, Bohm M, Hemkeppler E, Schonbrunn S, DeLorenzo N, Petermann K, et al. Visual performance of an extended depth of focus intraocular lens for treatment selection. Eye. 2019;33:1556–63.
Gil MA, Varon C, Cardona G, Buil JA. Visual acuity and defocus curves with six multifocal intraocular lenses. Int Ophthalmol. 2020;40:393–401.
Sanders DR, Sanders ML. Near visual acuity for everyday activities with accommodative and monofocal intraocular lenses. J Refract Surg. 2007;23:747–51.
Yoo YS, Whang WJ, Byun YS, Piao JJ, Kim DY, Joo CK, et al. Through-focus optical bench performance of extended depth-of-focus and bifocal intraocular lenses compared to a monofocal lens. J Refract Surg. 2018;34:236–43.
Liu X, Song X, Wang W, Zhu Y, Lyu D, Shentu X, et al. Comparison of the clinical outcomes between echelette extended range of vision and diffractive bifocal intraocular lenses. J Ophthalmol. 2019;2019:5815040.
de Medeiros AL, Jones Saraiva F, Iguma CI, Kniggendorf DV, Alves G, Chaves M, et al. Comparison of visual outcomes after bilateral implantation of two intraocular lenses with distinct diffractive optics. Clin Ophthalmol. 2019;13:1657–63.
Weeber HA, Meijer ST, Piers PA. Extending the range of vision using diffractive intraocular lens technology. J Cataract Refract Surg. 2015;41:2746–54.
Weeber HA, Piers PA. Theoretical performance of intraocular lenses correcting both spherical and chromatic aberration. J Refract Surg. 2012;28:48–52.
Acknowledgements
The authors would like to thank Sezer Hacıağaoğlu, MD, for his valuable contributions to the study’s ethics committee approval.
Funding
The study was financially supported by the Scientific Research Project Commission of Marmara University School of Medicine, Istanbul, Turkey (Project No: SAG-A-131216–0523).
Author information
Authors and Affiliations
Contributions
Acquisition, analysis, and interpretation of data, MOS, SAT, and ET; drafting the work, MOS, SAT, and ET; critical revision, MOS, SAT, and ET; final approval, MOS, SAT, and ET.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Sevik, M.O., Akkaya Turhan, S. & Toker, E. Clinical outcomes with a low add multifocal and an extended depth of focus intraocular lenses both implanted with mini-monovision. Eye 36, 1168–1177 (2022). https://doi.org/10.1038/s41433-021-01600-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01600-3