Abstract
To investigate the risk factors of eosinophilic fasciitis (EF) associated with pleural effusion (PE). A retrospective analysis was performed on 22 patients with EF diagnosed by skin biopsy in our hospital, and they were divided into EF-PE and EF according to chest computed tomography examination. The clinical characteristics, clinical manifestations, comorbidities and laboratory test indicators of the two groups were collected and compared, and the risk factors for occurring PE in patients with EF were determined by multivariate logistic regression analysis. Among 22 patients with EF, 8 had PE. The age, course of disease, incidence of fever, weight loss, cough and shortness of breath, pulmonary infection, hypothyroidism, hydronephrosis and kidney stone, swelling rate of small vascular endothelial cells, consolidation shadows, C-reactive protein and thyroid stimulating hormone in EF-PE group were higher than those in EF group, while free triiodothyronine and thyroxine were lower than those in EF group. Age, fever, shortness of breath, C-reactive protein, ESR, thyroid stimulating hormone, pulmonary infection, hypothyroidism, hydronephrosis, kidney stones, swollen small vascular endothelial cells and chest CT consolidation shadows were identified as risk factors for happening PE in patients with EF, while free triiodothyronine and free thyroxine were identified as protective factors against PE in patients with EF. The incidence of EF-PE was 36.36% in this study. Advanced age, high C-reactive protein, ESR, thyroid stimulating hormone, incidence of fever, shortness of breath, pulmonary infection, hydronephrosis, kidney stones, swollen small vascular endothelial cells, chest CT consolidation shadows, and low free triiodothyronine and thyroxine suggest that patients with EF are significantly at increased risk of PE.
Similar content being viewed by others
Introduction
Eosinophilic Fasciitis (EF), also known as Shulman syndrome, first reported by Shulman in 1974, which is characterized by progressive sclerosclerosis of symmetrical limbs, fascial hyperplasia, peripheral eosinophilia, hypergammaglobulinemia, joint pain, and increased erythrocyte sedimentation rate (ESR) with lymphocytes and plasmocytes infiltration. Approximately 53–67% of patients are sensitive to oral corticosteroid therapy1,2,3. EF can cause haematological diseases such as aplastic anemia, T-cell lymphoma, Hodgkin’s disease, multiple myeloma, myelodysplastic syndrome, and autoimmune diseases such as Hashimoto’s thyroiditis, systemic lupus erythematosus, and glomerulonephritis. It can also cause pneumonia and pleural diseases4,5. Braga et al.6 reported that an EF patient presented symptoms of dyspnea, lumbago and abdominal pain, decreased respiratory sounds in both lungs, and bilateral pleural effusion (PE) was found by chest computed tomography (CT) without hepatosplenomegaly. EF is a rare disease in clinical practice. As it involves multiple departments, it is often missed or misdiagnosed as sjögren’s syndrome, rheumatoid arthritis, scleroderma or systemic sclerosis, etc. PE could cause coughing, shortness of breath, difficulty breathing and even respiratory failure. At present, there are few reports about EF combined with PE, which seriously affects the prognosis and quality of life of patients2. Therefore, early detection and timely and effective treatment of PE are of great significance for the adjustment of diagnosis and treatment of EF and the prognosis of patients. The purpose of this study was to evaluate the clinical characteristics and related risk factors of EF-PE, so as to guide clinical treatment and improve the prognosis of patients with EF.
Patients and methods
Study patients
This study was approved by the Ethics Review Committee of the Second Xiangya Hospital of Central South University (Ethical Code: LYF2022085), all patients signed an informed consent form and all experiments were performed in accordance with the Declaration of Helsinki. We included 22 patients with EF who were hospitalized in the department of Dermatology, Rheumatology and Respiratory Medicine at the Second Xiangya Hospital of Central South University (Hunan, China) between January 2012 and April 2022. The following inclusion criteria were used for patients with EF: (a) EF was diagnosed in accordance with the diagnostic criteria proposed by Pinal-Fernandez et al. in 2014 and Jinnin et al. in 20182,7; (b) patient data were complete. PE was confirmed by chest CT and ultrasound-guided thoracentesis8. And the following patients were excluded: (a) Patients secondary to scleroderma, vascular syndrome, systemic sclerosis and other connective tissue diseases; (b) Diseases with other causes have been confirmed to lead to pleural effusion, such as potential malignancies, congestive heart failure, cirrhosis, tuberculosis and pleurisy, etc.; (c) Chest CT examination was not performed. In this study, patients with EF without PE were used as controls. Patients with PE related symptoms, such as exertive dyspnea or evidence of pleural effusion on chest CT, were excluded. A detailed description of the flow diagram for recruiting voluntary patients can be found in Fig. 1.
Data collection
After the collection of written informed consent, the demographic characteristics (age, gender, height, weight, smoking history, disease duration), clinical manifestations, concomitant diseases, laboratory data, Biopsy data, chest CT data, and (magnetic resonance imaging, MRI) data of affected sites of all enrolled patients with EF were documented.
Statistical analysis
SPSS 26.0 software (IBM Corp.) was used to perform all statistical analyses. Continuous variables were described as the mean ± standard deviation (M ± SD) or median (interquartile range), and categorical variables were expressed as the number (percentage). The comparisons of continuous variables were conducted using Student’s t test or the Mann–Whitney U test. Categorical variables, including the proportions, were compared using the chi-squared test or Fisher’s exact test. Risk factors were identified by multivariate logistic regression analysis. A P value < 0.05 indicated a statistically significant difference.
Ethical approval
This study was approved by the Ethics Review Committee of the Second Xiangya Hospital of Central South University (Ethical Code: LYF2022085), all patients signed an informed consent form and all experiments were performed in accordance with the Declaration of Helsinki.
Results
Demographic characteristics of the two groups
Table 1 shows the demographic characteristics of the 22 patients, including 8 patients with EF-PE (36.36%) and 14 patients with EF (63.63%). Most of the patients with EF-PE were male, with 5 (62.50%).
The age and course of disease in EF-PE group were significantly higher than that in EF group (P < 0.05; Table 1). There were no significant differences in gender, smoking history, smoking duration, history of overwork and body mass index (BMI) between 2 groups (P > 0.05). Detailed information on patient characteristics is shown in Table 1.
Clinical manifestations of the two groups
Table 2 shows the clinical manifestations of the two groups. The incidence of fever, weight loss, cough and shortness of breath in EF-PE group was significantly higher than that in EF group (P < 0.05; Table 2). There was no significant difference in the incidence of other clinical symptoms between the two groups (P > 0.05).
Laboratory indicators of the two groups
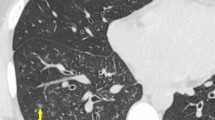
Table 3 shows the laboratory indicators of the two groups. The levels of C-reactive protein, ESR and thyroid stimulating hormone in EF-PE group were higher than those in EF group, while the levels of free triiodothyronine and free thyroxine were lower than those in EF group, the differences were statistically significant (P < 0.05; Table 3 and Fig. 2). There was no significant difference in other laboratory indexes between the two groups (P > 0.05). The pleural effusion was aspirated and laboratory tests were performed. The pleural effusion in the eight patients with EF-PE was found to be positive for Rivalta test, accompanied by elevated levels of total protein and adenosine deaminase (normal reference range: 0–24 μ/L). These results indicated that PE associated with EF is exudate. The pleural effusion showed lymphocyte, histocyte and inflammatory cells, mainly eosinophils, but no malignant tumor cells, and the acid-fast stain, Gram stain and bacterial culture were negative. The level of carcinoembryonic antigen was in the normal range (normal reference range: 0–6.5 ng/mL). These results indicate that PE associated with EF is not caused by malignant tumor cells, bacterial infection or mycobacterium tuberculosis.
The levels of C-reactive protein, ESR, free triiodothyronine, free thyroxine, and thyroid stimulating hormone in EF-PE and EF groups. The levels of C-reactive protein (A), ESR (B), free triiodothyronine (C), free thyroxine (D), and thyroid stimulating hormone (E) were compared between EF-PE and EF groups. Comparisons between the two groups were performed by Student’s t test or the Mann–Whitney U test. P < 0.05 was considered statistically significant.
Concomitant diseases of the two groups
Table 4 shows the concomitant diseases of the two groups. The incidence of pulmonary infection, hypothyroidism, hydronephrosis and kidney stones in EF-PE group was higher than that in EF group (P < 0.05; Table 4). There was no significant difference in the incidence of other complications between the two groups (P > 0.05).
Histopathological examination results of the two groups
Table 5 shows the histopathological examination results of the two groups. The swelling rate of small vascular endothelial cells in EF-PE group was higher than that in EF group (P < 0.05; Table 5). There was no significant difference in the incidence of other histopathological examination results between the two groups (P > 0.05).
MRI of the affected sites and chest CT of the two groups
Table 6 shows the MRI of the affected sites and chest CT of the two groups. The positive rate of chest CT consolidation shadows in EF-PE group was higher than that in EF group (P < 0.05; Table 6). There was no significant difference in the positive rate of other imaging examinations between the two groups (P > 0.05).
Multivariate logistic regression analysis of risk factors for EF happening PE based on clinical parameters and sociodemographic characteristics
Table 7 shows the multivariate logistic regression analysis of risk factors for EF happening PE based on clinical parameters and sociodemographic characteristics of the two groups.
According to the sociodemographic characteristics, patients who were older were more likely to occur PE, with an OR of 1.136 (95%CI = 1.005–1.284, P = 0.042). According to the clinical parameters, fever (OR = 7.500, 95%CI = 1.039–54.116, P = 0.046), shortness of breath (OR = 17.500, 95%CI = 1.596–191.892, P = 0.019), C-reactive protein (OR = 2.885, 95%CI = 1.264–6.589, P = 0.012), ESR (OR = 1.162, 95%CI = 1.007–1.339, P = 0.039), thyroid stimulating hormone (OR = 33.921, 95%CI = 1.747–418.838, P = 0.006), pulmonary infection (OR = 10.000, 95%CI = 1.260–79.339, P = 0.029), hypothyroidism (OR = 21.667, 95% CI = 1.802–260.574, P = 0.015), hydronephrosis (OR = 11.000, 95%CI = 1.420–85.201, P = 0.022), kidney stone (OR = 10.000, 95%CI = 1.260–79.339, P = 0.029), swollen small vascular endothelial cells (OR = 13.000, 95%CI = 1.109–152.351, P = 0.041), and chest CT consolidation shadows (OR = 7.500, 95%CI = 1.039–54.116, P = 0.046) were identified as risk factors for happening PE in patients with EF, while free triiodothyronine (OR = 0.029, 95%CI = 0.001–0.651, P = 0.026) and free thyroxine (OR = 0.553, 95%CI = 0.306–0.999, P = 0.049) were identified as protective factors against PE in patients with EF, with low levels of free triiodothyine and free thyroxine more likely to develop PE (Table 7).
Discussion
EF, first reported in 1974, is a rare clinical disease characterized by diffuse inflammation of the fascia and swelling and hardening of the skin. EF can cause diseases of the blood system, autoimmune diseases, lung and pleural diseases. It has been reported that patients with EF may develop bilateral pleural effusion, causing cough and dyspnea, which seriously affects the prognosis and quality of life of the patients6. However, the early prevalence of EF-PE may be underestimated due to the lack of obvious clinical symptoms in the early stage. Therefore, it is of great clinical significance to investigate the incidence of EF-PE and discuss the risk factors for its occurrence. In this study, 22 patients with EF by chest CT examination found that there are 8 cases (36.36%) complicated with PE, this is the first report of 8 patients with EF-PE, compared with the case before the medical record report, this may be related to sample source, sample size, the extensive examination of chest CT in EF and clinicians in improving the sensitivity of the PE.
At present, studies have found that the onset age of EF is mainly 40–50 years old, and there are also reports of young and old people9. In this study, the age of patients with EF-PE was higher than that of patients with EF, which was consistent with the results of previous case reports6,10. In addition, in this study, the disease course of patients with EF-PE is higher than that of patients with EF, and it has been reported that 10%-20% of patients will automatically resolve after 2–5 years of disease, and if complicated with PE, the disease course may be prolonged11. Multivariate logistic regression analysis showed that advanced age was a risk factor for PE in patients with EF. The possible reason was that elderly patients were vulnerable to infiltration of inflammatory cells due to weakened immune system defense, and inflammatory cells and inflammatory mediators would cause vasoconstriction and increased resistance, leading to the occurrence of PE. It is also possible that patients with EF are not detected early enough, resulting in an older age at diagnosis of EF-PE.
EF-PE is a rare disease that often involves multiple clinical departments. It has a variety of clinical symptoms and is difficult to detect in time. In this study, the incidence of fever, weight loss, cough and shortness of breath was higher in patients with EF-PE than that in patients with EF, which was consistent with the dyspnea reported by Killen et al. in patients with EF complicated with PE10, but fever was not reported in this case. patients with EF complicated with PE often have poor appetite, resulting in weight loss, cough, wheezing, shortness of breath and other symptoms, and even dyspnea. Multivariate logistic regression analysis showed that fever and shortness of breath were risk factors for PE in patients with EF. When patients with EF showed symptoms of fever, wheezing and shortness of breath, they should be aware of the possibility of PE.
Peripheral blood eosinophils, C-reactive protein and ESR are elevated in most patients with EF12,13. In this study, the pleural effusion in the patients with EF-PE was found to be positive for Rivalta test, and was associated with elevated levels of total protein and adenosine deaminase, indicating that the pleural effusion was an exudate in the patients with EP-PE. In addition, inflammatory cells were found in pleural effusion, mainly eosinophils, but no pathogenic bacteria, mycobacterium tuberculosis or malignant tumor cells. This is consistent with Killen et al. ‘s discovery of eosinophilic inflammatory cells in pleural effusion in patients with EF10. In this study, eosinophils in peripheral blood of patients with EF increased, but there was no statistically significant difference between EF-PE and patients with EF. Lebeaux et al. reported that not all patients would have increased eosinophils, 69% of patients in acute stage will be the increase in the number of eosinophils and as the disease development, under the condition of without intervention, eosinophils could descend to reference range, eosinophil levels and disease activity and severity is not parallel9. In this study, the C-reactive protein and ESR of patients with EF-PE were higher than those of patients with EF, and multivariate logistic regression analysis showed that C-reactive protein and ESR were risk factors for happening EF-PE. Interestingly, this study found for the first time that the level of thyroid stimulating hormone in patients with EF-PE was higher than that in patients with EF, while the level of free triiodothyronine and free thyroxine was lower than that in patients with EF. Hemorrhagic pleural effusion and hemorrhagic pericardial effusion have been reported in untreated hypothyroidism caused by pneumonia14. No other conditions causing hypothyroidism, such as severe infection, malignancy, or trauma, were found in the patients with EF and EF-PE in this study. Unfortunately, patients are not willing to undergo thyroid histopathological tests. Meanwhile, multivariate logistic regression analysis found that thyroid stimulating hormone was a risk factor for occurring EF-PE. Free triiodothyronine and free thyroxine were protective factors against EF-PE. When the above laboratory indicators are abnormal, the possible complications of EF should be considered in time, so as to achieve early diagnosis and treatment.
The main clinical features of EF are symmetrical, progressive sclerosclerosis of the limbs, joint pain, limited movement, and orange skin sign. Histopathologic examination of the affected sites may show infiltration of lymphocytes or plasma cells, accompanied by increased eosinophils and fascia hyperplasia and thickening13. In this study, all the 22 patients were examined by skin or muscle biopsy and found infiltration of lymphocytes and plasma cells, with or without eosinophil infiltration, fascia hyperplasia and intima thickening, which were consistent with previous reports. In addition, our study found that the incidence of small vascular endothelial cell swelling in patients with EF-PE was higher than that in patients with EF, and multivariate logistic regression analysis showed that small vascular endothelial cell swelling was a risk factor for happening EF-PE. It may be because inflammatory cells and mediators invade small vascular endothelial cells, resulting in endothelial cell injury, smooth muscle cell proliferation, increased vascular pressure, and promote the occurrence of PE. In this study, the MRI T1 and T2-weighted images of patients with EF at the affected site showed high signals, indicating the thickening of the affected fascia accompanied by mild to moderate strengthening, which was consistent with literature reports15. MRI has been recommended to track the course of disease in treated patients. However, in the absence of prospective studies, there are no rigorous criteria to assess the response to MRI therapy, nor has the optimal follow-up time for MRI therapy in EF been determined16.
EF is associated with systemic diseases such as hematological disease, neoplastic disease, lung and pleural disease4,17. In this study, the incidence of pulmonary infection, hypothyroidism, hydronephrosis, and kidney stones was higher in patients with EF-PE than that in patients with EF, while the incidence of other complications showed no statistical difference between the two groups. In the chest CT results of this study, the positive rate of consolidation shadows was higher in patients with EF-PE than that in patients with EF. Multivariate logistic regression analysis showed that pulmonary infection, hypothyroidism, hydronephrosis, kidney stones and chest CT consolidation shadows were risk factors for PE in patients with EF. Hypothyroidism, kidney seeper, and kidney stones can weaken the immune system’s defenses of patients, chest CT consolidation shadows often hint associated with pulmonary infection, accompanied by infiltration of inflammatory cells and mediators, and at the same time cause the release of a variety of proinflammatory factor, which can cause vasculitis causing vascular endothelial cell damage, make higher blood pressure and permeability increases, eventually occurring PE18,19.
There were some limitations of this study: (a) In the baseline data of this study, the average age of patients with EF-PE was 10 years older and the average course of disease was 1 year longer. Since EF is a rare disease with a small sample size, it may be a confounding factor for the baseline risk of PE. Future studies will strive to include more cases and try to control confounding factors. (b) This is a cross-sectional descriptive study; therefore, we cannot draw conclusions about the direction of causation. (c) As eosinophilic fasciitis with pleural effusion is a rare disease, therefore, additional cases and follow-up of patients need to be collected for further validation. (d) The mechanism of eosinophilic fasciitis associated with pleural effusion remains unexplained and needs further investigation.
Conclusion
This is the first study to report the incidence of EF-PE and to investigate the risk factors associated with it. In this study, the incidence of EF-PE was relatively high (36.36%), and advanced age, high C-reactive protein, high ESR, high thyroid stimulating hormone, low free triiodothyronine, low free thyroxine, the high incidence of fever, shortness of breath, pulmonary infection, hydronephrosis, kidney stones, swollen small vascular endothelial cells and chest CT consolidation shadows suggested that the risk of happening PE in patients with EF was significantly increased. More attention should be paid to patients with EF with these risk factors in clinical diagnosis and treatment.
Data availability
The data used and analyzed in this study are available from the corresponding author on reasonable request; E-mail: ouyangruoyun@csu.edu.cn.
References
Shulman, L. E. Diffuse fasciitis with hypergammaglobulinemia and eosinophilia: A new syndrome?. J. Rheumatol. 11(5), 569–570 (1984).
Pinal-Fernandez, I., Selva-O’ Callaghan, A. & Grau, J. M. Diagnosis and classification of eosinophilic fasciitis. Autoimmun Rev. 13(4–5), 379–382 (2014).
Wright, N. A. et al. Epidemiology and treatment of eosinophilic fasciitis: An analysis of 63 patients from 3 tertiary care centers. JAMA Dermatol. 152(1), 97–99 (2016).
de Masson, A. et al. Severe aplastic anemia associated with eosinophilic fasciitis: Report of 4 cases and review of the literature. Medicine 92(2), 69–81 (2013).
Martin, R. W. et al. The clinical spectrum of the eosinophilia-myalgia syndrome associated with L-tryptophan ingestion. Clinical features in 20 patients and aspects of pathophysiology. Ann. Intern. Med. 113(2), 124–134 (1990).
Braga Neto, M. B., Viscuse, P. V. & Sundsted, K. K. 56-year-old man with abdominal pain and dyspnea. Mayo Clin. Proc. 94(4), 704–708 (2019).
Jinnin, M. et al. Diagnostic criteria, severity classification and guidelines of eosinophilic fasciitis. J. Dermatol. 45(8), 881–890 (2018).
Saguil, A., Wyrick, K. & Hallgren, J. Diagnostic approach to pleural effusion. Am. Fam. Physician 90(2), 99–104 (2014).
Bischoff, L. & Derk, C. T. Eosinophilic fasciitis: Demographics, disease pattern and response to treatment: Report of 12 cases and review of the literature. Int. J. Dermatol. 47(1), 29–35 (2008).
Killen, J. W., Swift, G. L. & White, R. J. Eosinophilic fasciitis with pulmonary and pleural involvement. Postgrad. Med. J. 76(891), 36–37 (2000).
Islam, M. N. et al. Eosinophilic fasciitis: What matters in management in a developing country–a case report with two and a half-year follow-up. J. Health Popul. Nutr. 30(1), 117–120 (2012).
Lakhanpal, S., Ginsburg, W. W., Michet, C. J., Doyle, J. A. & Moore, S. B. Eosinophilic fasciitis: Clinical spectrum and therapeutic response in 52 cases. Semin. Arthritis Rheum. 17(4), 221–231 (1988).
Lebeaux, D. & Sène, D. Eosinophilic fasciitis (Shulman disease). Best Pract. Res. Clin. Rheumatol. 26(4), 449–458 (2012).
Iwamoto, Y. et al. Sudden extensive bloody pleural and pericardial effusion in a subject with untreated known hypothyroidism after total thyroidectomy, triggered by pneumonia. BMC Endocr. Disord. 22(1), 233 (2022).
Kirchgesner, T. et al. Eosinophilic fasciitis: Typical abnormalities, variants and differential diagnosis of fasciae abnormalities using MR imaging. Diagn. Interv. Imaging 96(4), 341–348 (2015).
Moulton, S. J., Kransdorf, M. J., Ginsburg, W. W., Abril, A. & Persellin, S. Eosinophilic fasciitis: Spectrum of MRI findings. AJR Am. J. Roentgenol. 184(3), 975–978 (2005).
Kissin, E. Y. et al. Ultrasound assessment of subcutaneous compressibility: A potential adjunctive diagnostic tool in eosinophilic fasciitis. J. Clin. Rheumatol. 19(7), 382–385 (2013).
Jany, B. & Welte, T. Pleural effusion in adults-etiology, diagnosis, and treatment. Dtsch. Arztebl. Int. 116(21), 377–386 (2019).
Beaudoin, S. & Gonzalez, A. V. Evaluation of the patient with pleural effusion. CMAJ 190(10), E291–E295 (2018).
Acknowledgements
We thank all the voluntary patients involved in the study.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81970086, No. 82170039), the Natural Science Foundation of Hunan Province (No. 2020JJ4802, No. 2020JJ4815) and the General Project of Department of Education of Hunan Province (No.20C1117).
Author information
Authors and Affiliations
Contributions
Conceptualization and design: Z.C., R.O.; Methodology: Z.C., R.O., X.X.; Data management: Z.C., R.O., Y.S.; Statistical analysis and interpretation: Z.C., R.O., B.W., A.J.; All authors contributed to drafting the original manuscript of important intellectual content and final approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Z., Wasti, B., Shang, Y. et al. Clinical characteristics and risk factors of patients with eosinophilic fasciitis associated with pleural effusion. Sci Rep 13, 5452 (2023). https://doi.org/10.1038/s41598-023-32678-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-32678-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.