Abstract
The digitally connected body with clinical grade multimodal and multisite biosignal acquisition is a key goal of the wearable device communities, which will enable advanced diagnostics and therapeutics. Recent advances in sensor and biointerfaces have enabled insight into biomarkers and physiological states that far exceed the commercially available technologies. However, they often require intimate contact with the target organ, which is possible acutely or over days but may not translate to continuous monitoring without substantial user engagement. Holistic device ecosystems or standards are therefore required to enable a digitally connected body. Here, we discuss current barriers and highlight potential avenues to enable a seamless, almost imperceptible network, of wearable sensors to capture health comprehensively and chronically.
Similar content being viewed by others
Introduction
Advances in noninvasive sensors for the skin mainly include the introduction of tightly integrated sensing platforms, also known as epidermal electronics1, that provides advanced insight due to either capturing fluids2 of interest or providing physical properties such as ultra-low physical and thermal mass3 that enables sensing properties well beyond the current “brick and strap” approach. This includes biochemical sensors that capture markers such as cortisol, ionic concentrations and pH that enables advanced insight into physiological stresses4. The lamination also enables biophysical sensors such as thermal conductivity5, thermography, mechanoacoustic6 and electrophysiological7 measurements currently not possible with bulky brick formats that physically prohibit these measurements because of resulting slow system response that manifests low fidelity and motion artefacts.
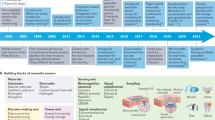
Translation of these epidermal approaches from the laboratory to scaled application in the clinic or at home use however are difficult and challenges are broadly outlined in Fig. 1. The challenges can be classified into 3 categories, Material and Interface, Integration and Adoption. In particular crucial for widespread adoption are materials and engineering schemes compatible with skin renewal, smart interfaces and technologies for seamless integration into healthcare systems to enable potentially significant impacts on life expectancy and population health, particularly in cardiac disease monitoring and treatment.
Materials and interface challenges
The fundamental limitations of the biointerface, if established adhesively via skin mounted electronics, is epidermal turnover, which limits these fixation strategies to several days before inducing irritation and eventual delamination8. This currently results in the need for frequent device replacement which may not be sustainable for chronic monitoring unless a system-level approach that provides sacrificial interfaces that are frequently renewed. However, it should be noted that while this is physically possible it may not yield user acceptance for chronic use as discussed in adoption challenges section9. Figure 2 highlights these most used device classes. Figure 2A is an example of a traditional brick and strap device that localizes all energy storage, computation and sensing in a rigid miniaturized solid body that is then affixed to the body with straps, locations often include the wrist and the chest. This localization of sensors and mass has substantial impact on sensing fidelity often highlighted in motion artefacts and degradation of signal due to sensor placement that follows strap location not the ideal physiological position that would yield highest fidelity signals10. The benefit of this device class is that commercially available materials and fabrication processes can be used enabling a high level of reliability and translation of new sensing technologies and algorithms. Figure 2B highlights thin device classes called epidermal electronics that are adhesively affixed to the skin and omit large components such as screens and electrochemical power supplies11. The approach yields a reduction in mechanical modulus from single-digit GPa for engineering materials to tens of kPa12 for epidermal electronics1. This results in additional sensing opportunities such as mechanoacoustic6, thermal13 and fluidic14 modlities and substantially reduces motion artefacts15 for more conventional biophysical measurements. A downside of this device class is the need for adhesive attachment that is limited by epidermal turnover, and generally a larger footprint than a brick and strap device due to dispersion of components to enable soft and thin mechanics. A compromise between these 2 approaches is shown in Fig. 2C which combines adhesively coupled multimodal epidermal devices with ultra-thin electronics that are attached with magnets. This enables sensing with disposable devices while more complex and expensive electronics can be reused2. A downside of this approach is accidental detachment of electronics and the additional complexity required for self-aligning and low-force connectors.
A Example of a brick and strap style device that confines electronics and sensors in one functional stiff island that gets attached to the body via straps B Example of epidermal electronic device with intimate contact with the skin reproduced with permission from WILEY - V C H VERLAG GMBH& CO. KGAA11 C Hybrid device with adhesive attachment to the skin and reusable electronic component2.
Sensor choice is limited by the biointerface and may also result in a barrier for chronic use. Examples are biochemical sensors which are known to degrade with mechanisms such as biofouling, degradation of recognition elements and electrochemical degradation16. There are also some issues associated with channeling of sweat into devices that on chronic timescales may pose biohazard concerns. These challenges also apply to electrophysiological sensing techniques that are known to suffer from impedance stability issues and efforts to find chronically stable skin electrodes is an active research area17. Because epidermal platforms conform well to the skin, motion artefacts are substantially reduced, however, devices that deform with the skin require special attention towards system-level mechanics to enable chronically stable operation.
Stimulators, especially for chronic time scales, that are noninvasive are difficult to achieve because they often require high levels of power which prohibits chronic use as discussed in the integration challenge section. There are many classes of stimulators for a variety of applications which can be broadly categorized by: haptic stimulation18 such as vibrotactile, pneumatic, acoustic and electrical stimulation such as Transcutaneous Electrical Nerve Stimulation (TENS), Functional Electrical Stimulation (FES), Electrical Muscle Stimulation (EMS)19; and thermal stimulation such as thermoelectric stimulators20. Most of these stimuli are particularly energy-hungry because of the impedance of the skin, electrical mechanical or thermal, to evoke peripheral nervous system responses and typically require tens of mW10,16.
Additionally, encapsulation is also an active research area, which is a consideration for chronic wearable devices that require mechanical strength and fluidic barriers. While this has been largely accomplished for rigid brick and strap devices, the high-fidelity epidermal device types require a different material class that can not rely on metal or ceramic enclosures. System-level mechanical design that enables deformation to absorb stresses are required, which is particularly difficult for computational nodes with analog components that require a rigid island. There are several strategies such as serpentine interconnects, and soft dielectric encapsulations that can overcome these challenges16.
Integration
Integration is an important topic for wearable devices because the way sensors are packaged and integrated in the wearable platform not only affects comfort and chronic stability, however, also directly impacts sensors performance.
The bottleneck for many wearable devices, especially if they require mW levels of power continuously, is the power supply. The biggest electrochemical power supplies such as Li-Ion batteries that are still comfortably wearable on chronic timescales are around 100-200 mAh at an average cell voltage of around 3.7 V which results in 370–740 mWh that translates to run times that are on the order of 1-2 days with high sampling rate multimodal devices8. To capture trends in health holistically run times of weeks or months are desired, hence frequent recharge and device interaction is required which leads to user retention issues, especially if the devices are not embedded in a device ecosystem that serves a dual purpose such as a smartwatch21. Hence new ways to power devices in a seamless and imperceptible way are required. Figure 3 shows examples of current approaches to solve chronic operational times without or with minimal user interaction.
A Biosymbiotic device with a small battery and recharge at distance22, enabling 24/7 operation without user interaction, image credit Gutruf lab. B Photonically powered device via perovskite solar cells enabling operation during light hours without additional infrastructure, Reproduced with permission from ref. 24, copyright (Springer Nature BV, 2023) C Wireless power distribution via textile25 enabling operation utilizing the smartphone as a power source.
Figure 3A is an example of utilizing wireless power transfer to wearables. The devices called biosymbiotics are meshed structures that can be worn 24/7 with epidermal contact that enables high-fidelity biosignal acquisition discussed in the integration section. The devices are recharged using far-field power casting that can transfer tens of mW over distances of up to 2 m which enables a frequent recharge of a small battery at locations of high occupancy to enable uninterrupted operation over weeks or months22. Other modes of wireless power transfer are using photonics, and near-field power. All of which can be used to create chronically wearable devices with high levels of energy requirements. Another approach to solve the energy problem is to use ambient sources of energy to enable continuous operation23. Figure 3B shows a device that is powered by ambient light through perovskite solar cells that harvest energy from inside and outside light sources to recharge a battery to enable operation during daytime hours24. Other approaches to extracting power from the body and the environment include the use of electrochemical power from sweat, motion via tribo- and piezoelectric effects, as well as thermal gradients via thermoelectrics. Using power supplies that we already carry with us is also an approach to enable distributed sensing with wearables, as well as implantable devices. An example of this is shown in Fig. 3C, here a shirt with embroidered metamaterials is used as a guide to capture the electromagnetic radiation emitted by a smartphone to power and communicate with nodes distributed on the body25. This enables the distribution of sensor nodes harnessing higher sensing fidelity and at the same time only requires the maintenance of one device reducing user fatigue.
An important topic that is not often discussed in academic work is the material platforms used for wearable devices. While replication on the lab scale may be straightforward, the translation to medium and large-scale products can be very challenging and may result in compromises that increase device stiffness and epidermal conformality to comply with scalable fabrication technologies. This may have several fundamental reasons, some can be materials driven, such as the availability of reel-to-reel processing of new functional materials, or some may be dictated by difficulties handling ultra-thin substrates required for epidermal sensor formats. Currently, successful translational activities use processes such as TPU for soft materials that can be injection molded and die cut26 as well as flexible electronic substrates such as polyimide which can be readily processed by printed circuit manufacturers enabling a scalability suitable at the consumer product scale27. New materials can also be successfully scaled by high-throughput printing methods such as screen printing28.
Communication with the wearable device are an important aspect, especially for devices that require 24/7 data streams. The most prevalent solution is blue tooth low energy (BLE) which provides the ability to send fairly high data rates (1–24 Mbps) at low power consumption of ~2-5 mW. However, devices require a smartphone or other BLE-enabled device in the vicinity. Other approaches are to store information locally on the device with subsequent offloading via NFC (Near Field Communication), which does not require any power because devices are powered by the reader, and are often chosen when low sampling rate sensing is sufficient27. Solutions for resource-constrained environments however are not widely investigated, very recent work leverages long-distance protocols alongside wearable antennas to transmit biosignals over 15 miles, suitable for areas where cell reception and smartphones are available29.
Computation of the acquired signals is currently a highly investigated topic. For chronic wearable devices, this is especially important because the data volume generated by multimodal high sampling rate biosignal acquisition can quickly amount to datasets that are not analyzable by humans. Here machine learning and artificial intelligence can enable fantastic capabilities especially when large and continuous datasets are available for training and efficient computation is available with wirelessly connected infrastructure30. This capability is especially useful for small and rarely occurring biomarkers that would be difficult or impossible to discover in contemporary daily clinical activity31. Fundamentally the question of where computation is performed is interesting because power consumption for communication makes up a substantial portion of a wearable energy budget so with proper analysis of the clinical need there may be a benefit of analyzing data on the device vs sending out all data to the cloud for analysis. With modern AI models model size and resulting computation time important parameters range from simple LSTM (Long Short-Term Memory) models occupying kb32 to complex models with multimodal data occupying several GB33.There is also the question of data security which may result in advanced energy costs with the need for encryption on the wearable node to comply with HIPA(Health Insurance Portability and Accountability Act) rules34.
Adoption challenges
A critical aspect of wearable device impact is user acceptance. There is some work on the psychology of using a wearable device, however, very few studies look at the link of new technological solutions and the associated impact on user acceptance. On average devices to capture health status only have a 50% user retention rate of only 6 month8. There are several factors that play into this decision, however leading drivers are wearability, privacy, ease of use, data accuracy and relevancy and perceived value for the wearer. Often the signal fidelity and clinical insight that a device can gather is only an aspect of its impact and many factors play into retention that go well beyond the hardware itself.
A device that can satisfy many of the above-noted challenges is still not guaranteed to commercially succeed. Because of the complicated commercial landscape especially for medical devices. Critical factors are interoperability with electronic health records, insurance coverage and medical community acceptance that ultimately drive clinical outcomes, adoption rate and user retention. This results in a high barrier to entry26.
Another important factor that slows innovation, however, is critical to maintaining trust in medical devices, are regulatory certifications through the FDA, CE or similar frameworks. Especially for diagnostic and therapeutic devices, which are likely to benefit most from multimodal and multisite chronically wearable hardware require substantial efforts to demonstrate efficacy and safety that can take many years and substantial funding to complete. Often the initial technological choices are tested rigorously in this process and knowledge of the benchmarks is useful when creating fundamental experiments on new technologies.
Because of this long process, market realities are only probed after substantial investment, the development of substantially new technology therefore represents a high risk, reducing the number of new translational activities. Examples of new device classes that failed are smartpills such as Proteus Digital Health Ingestible Sensors35 that despite exciting technology and regulatory approvals did not develop into widespread adoption because of slow adoption of the medical community, insurers, and patients.
Examples of multimodal chronic wearables
An example of a wearable device class that provides the ability to capture high-fidelity biosignals with high sampling rates continuously without interruption is shown in Fig. 4. The devices called biosymbiotics are attached epidermally via soft mesh structures that conform to the body without the need for adhesives (Fig. 4A). The devices retain the advantage of epidermal electronics, however, expand usage time over many weeks of operation because they provide the ability of normal epidermal turnover and transepidermal water loss. The energy supply is enabled via far-field power casting that recharges a small battery that supplies 24 h of autonomous operation. The devices are recharged when in 2 m proximity of a 915 MHz power caster which provides sufficient energy when placed in high occupancy areas such as a working desk or next to the bed (Fig. 4B). Operation can continue throughout the day with a variety of clothing and even during showers and aquatic activities (Fig. 4C). Data streams including battery state of chart over 2 weeks demonstrate operation at high sampling rates without data stream interruption and sensing modalities such as diametric biceps contractions and mK resolution skin temperature measurements, data that would not be attainable through traditional wearable devices (Fig. 4D)36.
A Biosymbiotic device overview detailing device architecture and sensing modalities. B Photographic image of recharge at distance. C Device operation with a variety of clothing. D Example of device operation over 2 weeks with uninterrupted datastreams and continuous high level of state of charge through contactless at distance recharge events Reproduced with permission from ref. 36, copyright (Elsevier, 2023).
Outlook
Wearable high-fidelity skin interfaces across critical sensing targets have developed rapidly, individually enabling insights beyond current clinical standards. However, approaches differ widely in engineering methods and successful deployment will hinge on key advances in materials and schemes for chronic use. Specifically, material strategies enable epidermal turnover and seamless epidermal attachment with smart soft interfaces such as hydrogels or ultra-thin flexible substrates with low impedance interfaces to the skin to enable uninterrupted high-fidelity interfaces over weeks of operation. Additionally, widespread adoption of these soft and imperceptible wearables will only succeed if seamless system-level approaches are created that connect interface types across modalities and application timeframes. This integration also applies to communication, computation, and powering strategies. A convergence of these core technologies such as the widespread availability of wireless power casting technologies, wireless low-power mesh network infrastructure hardware and low-power neural computation in commercially available systems on a chip, may be enabling technologies to facilitate rapid dissemination of this new device class. In the near future this device class likely has the highest impact on life expectancy and population health by detection and treatment of cardiac disease, where good solutions for treatment exist however chronic monitoring capabilities lack fidelity and capability. Key sensing technologies are continuous blood pressure and high-fidelity electrocardiograms with automated AI analysis methods in a form factor that is easily accepted by the patient, the medical treatment team, the hospital system and the insurer.
References
Kim, D.-H. et al. Epidermal electronics. Science (1979) 333, 838 LP–838843 (2011).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Krishnan, S. R. et al. Wireless, battery-free epidermal electronics for continuous, quantitative, multimodal thermal characterization of skin. Small vol. 14 Preprint at https://doi.org/10.1002/smll.201803192 (2018).
Sempionatto, J. R., Lasalde-Ramírez, J. A., Mahato, K., Wang, J. & Gao, W. Wearable chemical sensors for biomarker discovery in the omics era. Nat. Rev. Chem. 6, 899–915 (2022).
Krishnan, S. R. et al. Epidermal electronics for noninvasive, wireless, quantitative assessment of ventricular shunt function in patients with hydrocephalus. Sci. Transl. Med. 10, eaat8437 (2018).
Lee, K. et al. Mechano-acoustic sensing of physiological processes and body motions via a soft wireless device placed at the suprasternal notch. Nat. Biomed. Eng. 4, 148–158 (2020).
Driscoll, N. et al. MXene-infused bioelectronic interfaces for multiscale electrophysiology and stimulation. Sci. Transl. Med. 13, eabf8629 (2021).
Stuart, T., Hanna, J. & Gutruf, P. Wearable devices for continuous monitoring of biosignals: Challenges and opportunities. APL Bioeng. 6, 21502 (2022).
Yin, Z., Yan, J., Fang, S., Wang, D. & Han, D. User acceptance of wearable intelligent medical devices through a modified unified theory of acceptance and use of technology. Ann. Transl. Med. 10, 629 (2022).
Heikenfeld, J. et al. Wearable sensors: modalities, challenges, and prospects. Lab Chip 18, 217–248 (2018).
Kim, J. et al. Miniaturized flexible electronic systems with wireless power and near-field communication capabilities. Adv. Funct. Mater. 25, 4761–4767 (2015).
Raj, P. & Kumar, R. A brief review: a study on mechanical properties of polycarbonate with different nanofiller materials. Advances in Materials and Manufacturing Engineering: Select Proceedings of ICMME 2019 285–291 (2021).
Krishnan, S. R. et al. Continuous, noninvasive wireless monitoring of flow of cerebrospinal fluid through shunts in patients with hydrocephalus. NPJ Digit. Med. 3, 1–11 (2020).
Reeder, J. T. et al. Waterproof, electronics-enabled, epidermal microfluidic devices for sweat collection, biomarker analysis, and thermography in aquatic settings. Sci. Adv. 5, eaau6356 (2019).
Kim, J. et al. Miniaturized battery-free wireless systems for wearable pulse oximetry. Adv. Funct. Mater. 27, 1604373 (2017).
Ray, T. R. et al. Bio-integrated wearable systems: a comprehensive review. Chem. Rev. 119, 5461–5533 (2019).
Kim, H., Kim, E., Choi, C. & Yeo, W.-H. Advances in soft and dry electrodes for wearable health monitoring devices. Micromachines 13, 629 (2022).
Pacchierotti, C. et al. Wearable haptic systems for the fingertip and the hand: taxonomy, review, and perspectives. IEEE Trans. Haptics 10, 580–600 (2017).
Karamesinis, A., Sillitoe, R. V. & Kouzani, A. Z. Wearable peripheral electrical stimulation devices for the reduction of essential tremor: a review. IEEE Access 9, 80066–80076 (2021).
Yin, J., Hinchet, R., Shea, H. & Majidi, C. Wearable soft technologies for haptic sensing and feedback. Adv. Funct. Mater. 31, 2007428 (2021).
Tamilmani, K., Rana, N. P., Wamba, S. F. & Dwivedi, R. The extended Unified Theory of Acceptance and Use of Technology (UTAUT2): A systematic literature review and theory evaluation. Int J Inf Manage 57, 102269 (2021).
Tucker, S. et al. Biosymbiotic, personalized, and digitally manufactured wireless devices for indefinite collection of high-fidelity biosignals. Sci Adv 7, eabj3269 (2021).
Stuart, T., Cai, L., Burton, A. & Gutruf, P. Wireless and Battery-free Platforms for Collection of Biosignals. Biosens. Bioelectron. 178, 113007 (2021).
Min, J. et al. An autonomous wearable biosensor powered by a perovskite solar cell. Nat. Electron. 6, 630–641 (2023).
Tian, X. et al. Implant-to-implant wireless networking with metamaterial textiles. Nat. Commun. 14, 4335 (2023).
Xu, S., Kim, J., Walter, J. R., Ghaffari, R. & Rogers, J. A. Translational gaps and opportunities for medical wearables in digital health. Sci. Transl. Med. 14, eabn6036 (2023).
Heo, S. Y. et al. Wireless, battery-free, flexible, miniaturized dosimeters monitor exposure to solar radiation and to light for phototherapy. Sci. Transl. Med. 10, eaau1643 (2018).
Kim, J., Kumar, R., Bandodkar, A. J. & Wang, J. Advanced materials for printed wearable electrochemical devices: a review. Adv. Electron. Mater. 3, 1600260 (2017).
Stuart, T. et al. Biosymbiotic platform for chronic long-range monitoring of biosignals in limited resource settings. PNAS 120, e2307952120 (2023).
Song, Y. et al. 3D-printed epifluidic electronic skin for machine learning–powered multimodal health surveillance. Sci. Adv. 9, eadi6492 (2023).
Shcherbak, A., Kovalenko, E. & Somov, A. Detection and classification of early stages of Parkinson’s disease through wearable sensors and machine learning. IEEE Trans. Instrum. Meas. 72, 1–9 (2023).
Zihlmann, M., Perekrestenko, D. & Tschannen, M. Convolutional recurrent neural networks for electrocardiogram classification. in 2017 Computing in Cardiology (CinC) 1–4 https://doi.org/10.22489/CinC.2017.070-060 (2017).
Qiu, S. et al. Multimodal deep learning for Alzheimer’s disease dementia assessment. Nat. Commun. 13, 3404 (2022).
D’Aniello, G., Gravina, R., Gaeta, M. & Fortino, G. Situation-aware sensor-based wearable computing systems: a reference architecture-driven review. IEEE Sens. J 22, 13853–13863 (2022).
Litvinova, O. et al. Digital pills with ingestible sensors: patent landscape analysis. Pharmaceuticals 15, 1025 (2022).
Stuart, T. et al. Context-aware electromagnetic design for continuously wearable biosymbiotic devices. Biosens Bioelectron 228, 115218 (2023).
Acknowledgements
P.G. acknowledges support from the Department of Biomedical Engineering at the University of Arizona, support from the Flinn Foundation Translational Bioscience Seed Grant Pilot Program, the National Science Foundation I-Corp program, the Technology and Research Initiative Fund (TRIF) and the Gordon and Betty Moore Foundation.
Author information
Authors and Affiliations
Contributions
P.G. conceptualized and wrote the article and created figures.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Peer review
Peer review information
Communications Materials thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Ahyeon Koh and John Plummer.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gutruf, P. Towards a digitally connected body for holistic and continuous health insight. Commun Mater 5, 2 (2024). https://doi.org/10.1038/s43246-023-00443-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43246-023-00443-7