Article
|
Open Access
Featured
-
-
Article
| Open Access Sequential appetite suppression by oral and visceral feedback to the brainstem
Sequential appetite suppression by oral and visceral feedback to the brainstem
Genetically distinct neural circuits in the caudal brainstem receive feedback from the mouth and gut to regulate feeding behaviour on short and long timescales.
- Truong Ly
- , Jun Y. Oh
- & Zachary A. Knight
-
Article |
 Neural basis for fasting activation of the hypothalamic–pituitary–adrenal axis
Neural basis for fasting activation of the hypothalamic–pituitary–adrenal axis
Fasting-activated hypothalamic AgRP-expressing neurons trigger fasting-induced hypothalamic–pituitary–adrenal axis activation through projections to the paraventricular hypothalamus, where they activate CRH neurons by presynaptically inhibiting the terminals of tonically active GABAergic afferents from the bed nucleus of the stria terminalis.
- Amelia M. Douglass
- , Jon M. Resch
- & Bradford B. Lowell
-
Article
| Open Access Dopamine subsystems that track internal states
Dopamine subsystems that track internal states
Distinct dopaminergic neurons in the ventral tegmental area respond to physiological fluid balance and nutrient cues at specific stages of ingestion, driving learning about the physiological effects of ingestion.
- James C. R. Grove
- , Lindsay A. Gray
- & Zachary A. Knight
-
Article |
 Food cue regulation of AGRP hunger neurons guides learning
Food cue regulation of AGRP hunger neurons guides learning
In response to food cues, a hypothalamic circuit in the mouse brain transiently inhibits neurons expressing agouti-related peptide, and this promotes learning of cue-initiated food-seeking tasks.
- Janet Berrios
- , Chia Li
- & Bradford B. Lowell
-
Article |
 REV-ERB in GABAergic neurons controls diurnal hepatic insulin sensitivity
REV-ERB in GABAergic neurons controls diurnal hepatic insulin sensitivity
REV-ERB in GABAergic neurons orchestrates the rhythmic sensitivity of hepatic glucose production to insulin-mediated suppression that peaks at wakening, with implications in the extended dawn phenomenon.
- Guolian Ding
- , Xin Li
- & Zheng Sun
-
Article |
 Hunger enhances food-odour attraction through a neuropeptide Y spotlight
Hunger enhances food-odour attraction through a neuropeptide Y spotlight
Attraction to food odours is enhanced in mice that are experiencing hunger through a mechanism involving neuropeptide Y.
- Nao Horio
- & Stephen D. Liberles
-
Article |
 Violet-light suppression of thermogenesis by opsin 5 hypothalamic neurons
Violet-light suppression of thermogenesis by opsin 5 hypothalamic neurons
Mice possess neurons in the preoptic area of the hypothalamus that are sensitive to violet light; these deep brain neurons sense light via OPN5 and regulate adaptive thermogenesis in brown fat.
- Kevin X. Zhang
- , Shane D’Souza
- & Richard A. Lang
-
Article |
 Neurons that regulate mouse torpor
Neurons that regulate mouse torpor
A specific neuronal population in the medial and lateral preoptic area of the hypothalamus regulates entry into torpor in mice.
- Sinisa Hrvatin
- , Senmiao Sun
- & Michael E. Greenberg
-
Letter |
 A gut-to-brain signal of fluid osmolarity controls thirst satiation
A gut-to-brain signal of fluid osmolarity controls thirst satiation
Drinking behaviour in mice is regulated by a signal derived from the water and salt content of the gastrointestinal tract that is transmitted to forebrain neurons that control thirst via the vagus nerve.
- Christopher A. Zimmerman
- , Erica L. Huey
- & Zachary A. Knight
-
Letter |
 Genetic identification of leptin neural circuits in energy and glucose homeostases
Genetic identification of leptin neural circuits in energy and glucose homeostases
A subset of neurons in the hypothalamus is identified as the primary site of action for regulating energy balance and glucose homeostasis by leptin.
- Jie Xu
- , Christopher L. Bartolome
- & Dong Kong
-
Article |
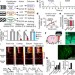 Homeostatic circuits selectively gate food cue responses in insular cortex
Homeostatic circuits selectively gate food cue responses in insular cortex
A combination of microprism-based cellular imaging to monitor insular cortex visual cue responses in behaving mice across hunger states with circuit mapping and manipulations reveals a neural basis for state-specific biased processing of motivationally relevant cues.
- Yoav Livneh
- , Rohan N. Ramesh
- & Mark L. Andermann
-
Article |
 MC4R-dependent suppression of appetite by bone-derived lipocalin 2
MC4R-dependent suppression of appetite by bone-derived lipocalin 2
Osteoblast-derived LCN2 activates the melanocortin 4 receptor in neurons of the paraventricular nucleus of the hypothalamus to suppress appetite, regulates insulin secretion and increases insulin sensitivity and glucose tolerance.
- Ioanna Mosialou
- , Steven Shikhel
- & Stavroula Kousteni
-
Letter |
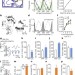 Gamma oscillations organize top-down signalling to hypothalamus and enable food seeking
Gamma oscillations organize top-down signalling to hypothalamus and enable food seeking
Coordinated gamma oscillations in the lateral hypothalamus, lateral septum and medial prefrontal cortex are shown to drive food-seeking behaviour in mice independently of nutritional need and to organize firing of feeding behaviour-related hypothalamic neurons.
- Marta Carus-Cadavieco
- , Maria Gorbati
- & Tatiana Korotkova
-
Letter |
 Thirst neurons anticipate the homeostatic consequences of eating and drinking
Thirst neurons anticipate the homeostatic consequences of eating and drinking
Feedback from the oral cavity to thirst-promoting neurons in the subfornical organ (SFO) during eating and drinking is integrated with information about blood composition, providing a prediction of how oral consumption will affect fluid balance and leading to changes in behaviour.
- Christopher A. Zimmerman
- , Yen-Chu Lin
- & Zachary A. Knight
-
Letter |
 Bidirectional electromagnetic control of the hypothalamus regulates feeding and metabolism
Bidirectional electromagnetic control of the hypothalamus regulates feeding and metabolism
Activation of glucose-sensing neurons in the ventromedial hypothalamic nucleus using radio waves or magnetic fields remotely and non-invasively in vivo increases plasma glucose and glucagon, and suppresses plasma insulin; conversely, remote inhibition of glucose-sensing neurons decreased blood glucose and increased plasma insulin.
- Sarah A. Stanley
- , Leah Kelly
- & Jeffrey M. Friedman
-
Article |
 Neurons for hunger and thirst transmit a negative-valence teaching signal
Neurons for hunger and thirst transmit a negative-valence teaching signal
Cell-type-specific electrical activity manipulations and deep-brain imaging in mice of neuronal populations associated with homeostasis of nutrient or fluid intake reveals that learning is conditioned by a negative-valence signal from the hunger-mediating AGRP neurons and also from the thirst-mediating neurons in the subfornical organ.
- J. Nicholas Betley
- , Shengjin Xu
- & Scott M. Sternson
-
Article |
 Hypothalamic POMC neurons promote cannabinoid-induced feeding
Hypothalamic POMC neurons promote cannabinoid-induced feeding
Cannabinoid-induced feeding signals are shown to enhance pro-opiomelanocortin (POMC) neuronal activity in mice, causing an enhancement of β-endorphin release, which is crucial in causing this cannabinoid-induced response; these results uncover an overlooked role of hypothalamic POMC neurons in the promotion of feeding by cannabinoids.
- Marco Koch
- , Luis Varela
- & Tamas L. Horvath
-
Letter |
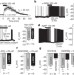 G-protein-independent coupling of MC4R to Kir7.1 in hypothalamic neurons
G-protein-independent coupling of MC4R to Kir7.1 in hypothalamic neurons
α-MSH and AgRP, two hypothalamus-derived peptides with opposing actions on the melanocortin-4 receptor (MC4R), modulate neurons driving feeding behaviour; although previous downstream mechanisms of cellular modulation by these peptides have been determined, here α-MSH and AgRP are shown to regulate neural activity by coupling MC4R to Kir7.1 potassium channels and closing or opening them, respectively.
- Masoud Ghamari-Langroudi
- , Gregory J. Digby
- & Roger D. Cone
-
Letter |
 An excitatory paraventricular nucleus to AgRP neuron circuit that drives hunger
An excitatory paraventricular nucleus to AgRP neuron circuit that drives hunger
The AgRP-expressing neurons in the arcuate nucleus drive food-seeking behaviours during caloric restriction; a mouse study of monosynaptic retrograde rabies spread and optogenetic circuit mapping reveals that these neurons are activated by input from hypothalamic paraventricular nucleus cells and their activation or inhibition can modulate feeding behaviour.
- Michael J. Krashes
- , Bhavik P. Shah
- & Bradford B. Lowell
-
-
Research Highlights |
Hungry cells eat from within
-
Article |
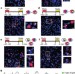 Functional identification of an aggression locus in the mouse hypothalamus
Functional identification of an aggression locus in the mouse hypothalamus
Certain regions of the hypothalamus are important in aggression, but until recently, it has been difficult to specifically stimulate specific cell types within a mixed population of cells. Here, optogenetics is used to solve this specificity problem, finding that optogenetic stimulation of a subdivision within the ventromedial hypothalamus can elicit inappropriate attack behaviours in mice, but electrical stimulation does not produce the same result. Additional analysis of genetic and electrophysiological activity revealed overlapping neuronal subpopulations involved in fighting and mating, with potential competition between these behaviours, as neurons activated during aggression are inhibited during mating.
- Dayu Lin
- , Maureen P. Boyle
- & David J. Anderson

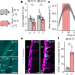 A brainstem–hypothalamus neuronal circuit reduces feeding upon heat exposure
A brainstem–hypothalamus neuronal circuit reduces feeding upon heat exposure Structure: The anatomy of sleep
Structure: The anatomy of sleep