Featured
-
-
Letter |
 Crystal structure of a bacterial homologue of the bile acid sodium symporter ASBT
Crystal structure of a bacterial homologue of the bile acid sodium symporter ASBT
- Nien-Jen Hu
- , So Iwata
- & David Drew
-
Letter |
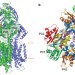 Crystal structures of the CusA efflux pump suggest methionine-mediated metal transport
Crystal structures of the CusA efflux pump suggest methionine-mediated metal transport
Gram-negative bacteria, such as Escherichia coli, use tripartite efflux complexes in the resistance-nodulation-cell division family to expel toxic compounds from the cell. The CusCBA system is responsible for removing biocidal Cu(I) and Ag(I) ions. Here, the X-ray crystal structure is reported of CusA in the absence and presence of bound Cu(I) or Ag(I). The structures reveal that the metal-binding sites are located within the cleft region of the periplasmic domain. A potential pathway for ion export is proposed.
- Feng Long
- , Chih-Chia Su
- & Edward W. Yu
-
Letter |
 Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase
Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase
The Na+/K+-ATPase pumps three sodium ions out of and two potassium ions into the cell while splitting a single molecule of ATP. Here it is found that the carboxy terminus of the ATPase's α-subunit is also a key regulator of a previously unrecognized ion pathway. The data indicate that, in the ATPase's potassium-bound state, a cytoplasmic proton can enter and stabilize site III when empty. When potassium is released, the proton returns to the cytoplasm, thus permitting an overall asymmetric stoichiometry of the transported ions.
- Hanne Poulsen
- , Himanshu Khandelia
- & Poul Nissen
-
News |
 Ageing cells lose protein pumps
Ageing cells lose protein pumps
Longevity of cells could be linked to levels of cellular pumps that get rid of toxic cell products.
- Cassandra Willyard
-
Letter |
 High-performance genetically targetable optical neural silencing by light-driven proton pumps
High-performance genetically targetable optical neural silencing by light-driven proton pumps
If the activity of genetically specified neurons is silenced in a temporally precise fashion, the roles of different cell classes in neural processes can be studied. Members of the class of light-driven outward proton pumps are now shown to mediate powerful, safe, multiple-colour silencing of neural activity. The gene archaerhodopsin-3 (Arch) enables near 100% silencing of neurons in the awake brain when virally expressed in the mouse cortex and illuminated with yellow light.
- Brian Y. Chow
- , Xue Han
- & Edward S. Boyden

 Antiparallel EmrE exports drugs by exchanging between asymmetric structures
Antiparallel EmrE exports drugs by exchanging between asymmetric structures