Article
|
Open Access
Featured
-
-
Article
| Open Access The electron–proton bottleneck of photosynthetic oxygen evolution
The electron–proton bottleneck of photosynthetic oxygen evolution
Microsecond infrared spectroscopy together with quantum chemistry reveal the rate-determining proton and electron movements and identify an oxygen-radical state of the manganese cluster as the S4 state.
- Paul Greife
- , Matthias Schönborn
- & Holger Dau
-
Article |
 Molecular interplay of an assembly machinery for nitrous oxide reductase
Molecular interplay of an assembly machinery for nitrous oxide reductase
Cryo-electron microscopy structures of the bacterial protein machinery that is involved in the production and function of nitrous oxide provide insight into the assembly pathway of this enzyme and the mechanisms of copper transport.
- Christoph Müller
- , Lin Zhang
- & Oliver Einsle
-
Article
| Open Access Discovery, structure and mechanism of a tetraether lipid synthase
Discovery, structure and mechanism of a tetraether lipid synthase
In Methanocaldococcus jannaschii, a radical S-adenosylmethionine enzyme catalyses the formation of the biphytanyl chain.
- Cody T. Lloyd
- , David F. Iwig
- & Squire J. Booker
-
Article |
 Nitrogen reduction by the Fe sites of synthetic [Mo3S4Fe] cubes
Nitrogen reduction by the Fe sites of synthetic [Mo3S4Fe] cubes
Iron atoms in a synthetic metal–sulfur cluster can capture nitrogen and catalyse its silylation, demonstrating successful nitrogen reduction by iron atoms in a sulfur-rich environment.
- Yasuhiro Ohki
- , Kenichiro Munakata
- & Kazuki Tanifuji
-
Article |
 Overcoming universal restrictions on metal selectivity by protein design
Overcoming universal restrictions on metal selectivity by protein design
An alternative approach to metalloprotein design shows that it is possible to overcome the restrictions of the Irving–Williams series and achieve both flexibility and specificity in the binding of metal ions.
- Tae Su Choi
- & F. Akif Tezcan
-
Article |
 Structure of a B12-dependent radical SAM enzyme in carbapenem biosynthesis
Structure of a B12-dependent radical SAM enzyme in carbapenem biosynthesis
X-ray crystal structures of TokK, a cobalamin- or B12-dependent radical SAM methylase, provide insight into how these enzymes use sequential radical-mediated methylations to assemble the C6 side chain of carbapenem antibiotics.
- Hayley L. Knox
- , Erica K. Sinner
- & Squire J. Booker
-
Article |
 Structural basis for tRNA methylthiolation by the radical SAM enzyme MiaB
Structural basis for tRNA methylthiolation by the radical SAM enzyme MiaB
Crystal structures reveal the catalytic mechanism through which the radical S-adenosylmethionine enzyme MiaB adds a methylthio group onto tRNA.
- Olga A. Esakova
- , Tyler L. Grove
- & Squire J. Booker
-
Article |
 Constructing protein polyhedra via orthogonal chemical interactions
Constructing protein polyhedra via orthogonal chemical interactions
An inorganic chemical approach to biomolecular design is used to generate ‘cages’ that can simultaneously promote symmetry and multiple modes of protein interactions.
- Eyal Golub
- , Rohit H. Subramanian
- & F. Akif Tezcan
-
Letter |
 Metal-free ribonucleotide reduction powered by a DOPA radical in Mycoplasma pathogens
Metal-free ribonucleotide reduction powered by a DOPA radical in Mycoplasma pathogens
A subclass of ribonucleotide reductase in Mycoplasma pathogens contains a stable radical formed from a modified tyrosine residue, overturning the presumed requirement for a dinuclear metal site in aerobic ribonucleotide reductase.
- Vivek Srinivas
- , Hugo Lebrette
- & Martin Högbom
-
Letter |
 A naturally occurring antiviral ribonucleotide encoded by the human genome
A naturally occurring antiviral ribonucleotide encoded by the human genome
Viperin inhibits the replication of various viruses by catalysing the conversion of CTP to ddhCTP, which is a unique nucleotide that functions as replication-chain terminator of RNA-dependent RNA polymerases.
- Anthony S. Gizzi
- , Tyler L. Grove
- & Steven C. Almo
-
Article |
 In-crystal reaction cycle of a toluene-bound diiron hydroxylase
In-crystal reaction cycle of a toluene-bound diiron hydroxylase
Crystal structures and DFT calculations suggest a possible mechanism for diiron enzyme arene hydroxylation.
- Justin F. Acheson
- , Lucas J. Bailey
- & Brian G. Fox
-
Article |
 A B12-dependent radical SAM enzyme involved in oxetanocin A biosynthesis
A B12-dependent radical SAM enzyme involved in oxetanocin A biosynthesis
The biosynthesis of oxetanocin A involves OxsB, a B12-dependent S-adenosylmethionine radical enzyme, which catalyses an unusual ring contraction of a 2′-deoxyadenosine phosphate.
- Jennifer Bridwell-Rabb
- , Aoshu Zhong
- & Hung-wen Liu
-
Letter |
 Charge-density analysis of an iron–sulfur protein at an ultra-high resolution of 0.48 Å
Charge-density analysis of an iron–sulfur protein at an ultra-high resolution of 0.48 Å
The ultra-high-resolution structure of the high-potential iron–sulfur protein at 0.48 Å, the highest-resolution X-ray crystal structure of a protein reported so far.
- Yu Hirano
- , Kazuki Takeda
- & Kunio Miki
-
Letter |
 Binding of dinitrogen to an iron–sulfur–carbon site
Binding of dinitrogen to an iron–sulfur–carbon site
A synthetic complex with a sulfur-rich coordination sphere is described that, upon reduction, breaks an Fe–S bond and binds N2, providing a model for the iron–molybdenum cofactor used by nitrogenase enzymes to produce ammonia.
- Ilija Čorić
- , Brandon Q. Mercado
- & Patrick L. Holland
-
Letter |
 Structural insight into magnetochrome-mediated magnetite biomineralization
Structural insight into magnetochrome-mediated magnetite biomineralization
The magnetosome-associated protein mamP is an iron oxidase that reveals a unique arrangement of a self-plugged PDZ domain fused to two magnetochrome domains, defining a new class of c-type cytochrome exclusively found in magnetotactic bacteria.
- Marina I. Siponen
- , Pierre Legrand
- & David Pignol
-
Letter |
 Elucidation of the Fe(iv)=O intermediate in the catalytic cycle of the halogenase SyrB2
Elucidation of the Fe(iv)=O intermediate in the catalytic cycle of the halogenase SyrB2
Synchrotron-based nuclear resonance vibrational spectroscopy is used to characterize the reactive Fe(iv)=O intermediate of the halogenase SyrB2; the substrate directs the orientation of this intermediate, presenting specific frontier molecular orbitals that can activate the selective halogenation.
- Shaun D. Wong
- , Martin Srnec
- & Edward I. Solomon
-
Letter |
 Biomimetic assembly and activation of [FeFe]-hydrogenases
Biomimetic assembly and activation of [FeFe]-hydrogenases
Three synthetic mimics of the di-iron centre in [FeFe]-hydrogenases are loaded onto the HydF protein and then transferred to apo-HydA1; full activation of HydA1 was achieved only with the HydF hybrid protein that contained the mimic with an azadithiolate bridge, confirming the presence of this ligand in the active site of native [FeFe]-hydrogenases.
- G. Berggren
- , A. Adamska
- & M. Fontecave
-
News & Views |
 Bundles of insights into sugar transporters
Bundles of insights into sugar transporters
The structure of a bacterial protein belonging to the 'sugar porter' family guides the building of long-sought molecular models of proteins that transport glucose across cell membranes in humans. See Article p.361
- Peter J. F. Henderson
- & Stephen A. Baldwin
-
Article |
 Antiparallel EmrE exports drugs by exchanging between asymmetric structures
Antiparallel EmrE exports drugs by exchanging between asymmetric structures
NMR and single molecule FRET experiments show that antiparallel EmrE dimers interconvert between two identical but oppositely oriented conformations that are each open only to one side of the membrane.
- Emma A. Morrison
- , Gregory T. DeKoster
- & Katherine A. Henzler-Wildman
-
Letter |
 Structure and reactivity of a mononuclear non-haem iron(III)–peroxo complex
Structure and reactivity of a mononuclear non-haem iron(III)–peroxo complex
- Jaeheung Cho
- , Sujin Jeon
- & Wonwoo Nam
-
Letter |
 Structural basis for a [4Fe-3S] cluster in the oxygen-tolerant membrane-bound [NiFe]-hydrogenase
Structural basis for a [4Fe-3S] cluster in the oxygen-tolerant membrane-bound [NiFe]-hydrogenase
- Yasuhito Shomura
- , Ki-Seok Yoon
- & Yoshiki Higuchi
-
Letter |
 The crystal structure of an oxygen-tolerant hydrogenase uncovers a novel iron-sulphur centre
The crystal structure of an oxygen-tolerant hydrogenase uncovers a novel iron-sulphur centre
- Johannes Fritsch
- , Patrick Scheerer
- & Christian M. T. Spahn
-
Letter |
 Crystal structure of a bacterial homologue of the bile acid sodium symporter ASBT
Crystal structure of a bacterial homologue of the bile acid sodium symporter ASBT
- Nien-Jen Hu
- , So Iwata
- & David Drew
-
Q&A |
 Turning point: Charalampos Kalodimos
Turning point: Charalampos Kalodimos
Rutgers University biophysicist explains how heading overseas has enriched his career.
- Virginia Gewin
-
Letter |
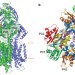 Crystal structures of the CusA efflux pump suggest methionine-mediated metal transport
Crystal structures of the CusA efflux pump suggest methionine-mediated metal transport
Gram-negative bacteria, such as Escherichia coli, use tripartite efflux complexes in the resistance-nodulation-cell division family to expel toxic compounds from the cell. The CusCBA system is responsible for removing biocidal Cu(I) and Ag(I) ions. Here, the X-ray crystal structure is reported of CusA in the absence and presence of bound Cu(I) or Ag(I). The structures reveal that the metal-binding sites are located within the cleft region of the periplasmic domain. A potential pathway for ion export is proposed.
- Feng Long
- , Chih-Chia Su
- & Edward W. Yu
-
Letter |
 Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase
Neurological disease mutations compromise a C-terminal ion pathway in the Na+/K+-ATPase
The Na+/K+-ATPase pumps three sodium ions out of and two potassium ions into the cell while splitting a single molecule of ATP. Here it is found that the carboxy terminus of the ATPase's α-subunit is also a key regulator of a previously unrecognized ion pathway. The data indicate that, in the ATPase's potassium-bound state, a cytoplasmic proton can enter and stabilize site III when empty. When potassium is released, the proton returns to the cytoplasm, thus permitting an overall asymmetric stoichiometry of the transported ions.
- Hanne Poulsen
- , Himanshu Khandelia
- & Poul Nissen
-
News |
 Ageing cells lose protein pumps
Ageing cells lose protein pumps
Longevity of cells could be linked to levels of cellular pumps that get rid of toxic cell products.
- Cassandra Willyard
-
News & Views |
 Model offers intermediate insight
Model offers intermediate insight
Chemical models of enzymes' active sites aid our understanding of biological reactions. Such a model of a reaction intermediate promises to advance our knowledge of the biochemistry of iron-containing haem enzymes.
- Kenneth D. Karlin
-
Letter |
 High-performance genetically targetable optical neural silencing by light-driven proton pumps
High-performance genetically targetable optical neural silencing by light-driven proton pumps
If the activity of genetically specified neurons is silenced in a temporally precise fashion, the roles of different cell classes in neural processes can be studied. Members of the class of light-driven outward proton pumps are now shown to mediate powerful, safe, multiple-colour silencing of neural activity. The gene archaerhodopsin-3 (Arch) enables near 100% silencing of neurons in the awake brain when virally expressed in the mouse cortex and illuminated with yellow light.
- Brian Y. Chow
- , Xue Han
- & Edward S. Boyden

 Enhanced rare-earth separation with a metal-sensitive lanmodulin dimer
Enhanced rare-earth separation with a metal-sensitive lanmodulin dimer